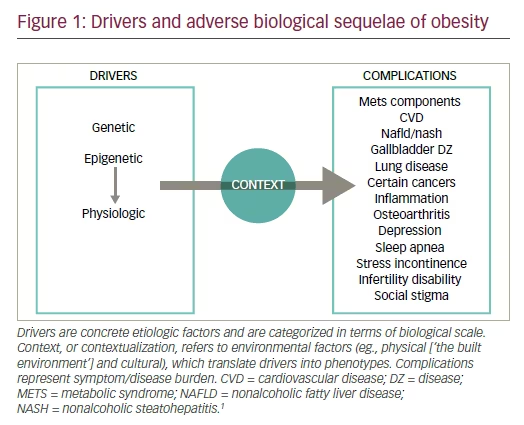
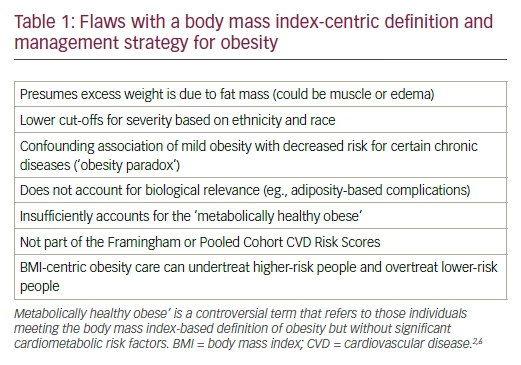
Obesity has been declared a chronic disease by many organizations, with multiple complex drivers and adverse biological sequelae, commonly regarded as complications (see Figure 1).1,2 For most clinicians and the informed public, overweight and obesity are defined exclusively by a body mass index (BMI; weight [kg]/height [m]2) ≥25 and 30, respectively. However, there are many flaws in this definition (see Table 1). The prevalence rate for obesity in the US remains essentially unchanged, with troublesome increases in certain population subsets, such as children, several ethnicities (particularly those in lower socio-economic strata), women over 60 years old, and more severe forms of obesity (for example, those with a BMI ≥35).3,4 However, with all the knowledge that has been learned, and advances in obesity research, education, and clinical practice, why is the problem, at least from an epidemiological standpoint, not really improving? One answer may be the need for greater governmental intervention (for example, behavioral economics and obesity-related legislation).5 But even this may be quite unrealistic. A better option for now may be to use a new term and approach. The word ‘obesity’ is antiquated, does not reflect the basic underlying pathophysiology, and carries a stigma that resists any conciliatory political correctness. On the other hand, ‘adiposity’ and ‘chronic disease’ are two medical concepts that, when joined together as ‘adiposity-based chronic disease’ (ABCD) transmit a clear message.1,6
What is adiposity and how does it lead to chronic disease?
There are many terms in the medical literature referring to body fat and related clinical conditions, but these terms generally lack specificity and detail. Though obesity is the most popular, others include ‘adiposopathy’, ‘fatness’, and ‘adiposity’. Adiposopathy refers to the premise that the key pathological mediators of abnormal body fat that lead to adiposity-based complications (particularly cardiometabolic) are secreted factors: hormones, cytokines, adipokines, etc.7 Fatness is a non-specific, generic term typically used to describe an increased amount of fat, but this term also confers stigma, as does any word with the root ‘fat’.8
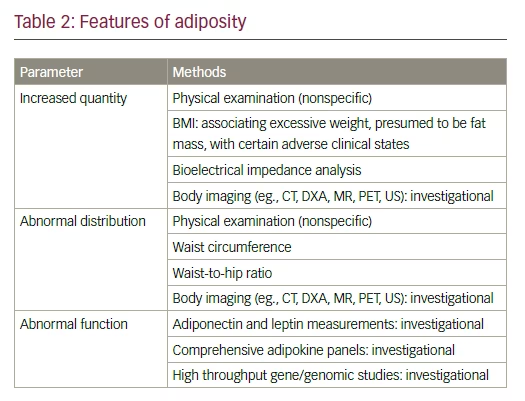
Adiposity refers to body fat in general terms but includes, not only increased quantity and/or abnormal function, but also the impact of various distributions of fat (see Table 2).9 For instance, visceral (intra-abdominal) adipose tissue is associated with higher cardiometabolic risks than subcutaneous abdominal adipose tissue.9 Intracellular fat can also occur in bone marrow, perivascular, epi/pericardial, and subcutaneous gluteal/femoral adipose tissue, as well as ectopic lipids in non-adipose tissue, such as skeletal muscle, cardiac muscle, kidney, pancreas, and liver.10,11 In fact, it has been postulated that ectopic lipid deposition occurs when two necessary pre-conditions exist: first, over-nutrition and second, inability of eutopic adipose tissue to accommodate excess energy storage needs.12 It is further hypothesized that it is the ectopic lipid that principally contributes to cardiometabolic risk, or the metabolic syndrome (MetS), explaining in a somewhat simplistic fashion why many patients with obesity (but minimal ectopic fat) can be healthy (with no or minimal MetS
components).12 Additionally, in some ethnicities, it is the relatively low amounts of fat and muscle mass that contribute to cardiometabolic risk: Asian-Indians have a specific diabetes and obesity phenotype described by Mohan et al.13 that is associated with sarcopenia (low muscle mass and therefore relatively high fat-to-muscle mass even with normal adjusted BMI). Long regarded as a medical curiosity, ‘lipodystrophy’ (complete or partial loss or absence of adipose tissue) has found new relevance in the context of increased cardiometabolic risk.14 The decreased capacity of lipodystrophic adipose tissue stores can promote increased ectopic lipid stores.15 For the sake of clarity, adiposity and ABCD refer to intracellular fat molecules, whereas extracellular fat molecules are relevant in the context of dyslipidemias and lipid-based chronic disease.
Overall, a re-working of obesity into an ABCD model provides the granularity and robustness to better account for the human phenotype and expression of disease. Consequently, one can foresee a shift in the diagnostic work-up from conventional anthropometrics (BMI, waist circumference [WC], etc.) to advanced body imaging and molecular biology studies. High-resolution imaging can provide not only anatomic description of the disease process, but also information about function, such as inflammation and metabolism. Interestingly, Lee et al.16 demonstrated that adipose tissue volume and attenuation by computed tomography (CT) was associated with cardiovascular disease (CVD) risk, independent of anthropometrics (BMI and WC).
An accurate analysis of the adipocyte secretome, as well as secretory products in ectopic lipid, can provide important information to guide decision-making, including emergent properties from network analysis for research hypothesis generation. For example, a preliminary network analysis of an adipokine-cardiovascular-lifestyle network is presented by Mechanick et al.17
The second part of ABCD is chronic disease. This is defined as a pathophysiological state persisting for at least three months and characterized by adaptive and maladaptive processes that may or may not achieve a steady state, but nevertheless evolve and develop biological complexity.18 The implication is that, unlike relatively direct and expeditious treatments for acute or sub-acute illnesses that target
the most life-threatening components, treatments for chronic disease need to target many components at different time points, and might even miss the underlying root cause(s). James et al.19 have postulated that chronic disease results from some as-yet ill-defined disequilibrium among evolution, adaption, resultant allostatic genetic traits, and the environment. Hence, a representation of genetic load may be able to predict not only the potential for a chronic disease, but the type of interventions best suited for prevention and health promotion in a particular individual.
The role of lifestyle medicine in adiposity-based chronic disease
Lifestyle medicine is the non-pharmacological and non-surgical management of chronic disease.18 Conceptually, the multiple facets intrinsic to lifestyle medicine, particularly the networked molecules in a healthy eating pattern, are perfectly suited to address the network-based complexities of chronic disease.18,20 Empirically, lifestyle medicine needs to be comprehensive. For instance, in an observational study of monozygotic twin pairs with discordant BMI, Nordstrom et al.21 found that high BMI was more associated with T2D risk than CVD or mortality risk; this implies how efforts to simply lose weight in patients with obesity would be insufficient to reduce CVD risk as it depends on where they lose the weight from and how they lose the weight.
The goal of lifestyle medicine is to reduce disease-oriented risk in a preventive care paradigm. But it is also the goal, perhaps even more importantly, to promote a state of health regardless of the pre-existing state or severity of disease. Health not only applies to physical or biological health, but also to behavioral health that includes quality of life and an improved body image. This approach to ABCD not only removes the stigma of referring to ‘obesity’, but also changes the conversation to one about ‘health’ (positive) instead of ‘disease’ (negative). Whether this newer approach in a behavioral medicine context is more motivational and effective remains to be seen and merits scientific study. Pragmatically, ABCD is managed as part of a comprehensive lifestyle intervention, from childhood to advanced age, and not in isolation addressing one component at the brief time of a patient encounter. More specifically, each of the major cardiometabolic risks and their related chronic disease states (adiposity-, hypertension-, dysglycemia-, dyslipidemia-, tobacco-, nutrition-, and physical activity-based chronic disease)22 receives focus, with the other components of lifestyle medicine (such as behavior, sleep hygiene, community engagement, and alcohol moderation)23 also being addressed. The success or failure of lifestyle medicine in patients with ABCD can be measured in terms of adipositybased complications (improvements in specific metrics [A1C, low-density lipoprotein, blood pressure, C-reactive protein, estimated glomerular filtration rate, etc.]) or related symptoms independent of the use of pharmacotherapy or procedures.
The trajectory for ABCD management and health promotion through lifestyle medicine is building an evidence base with high-impact scientific research, development of comprehensive education and training programs, refinement of structured approaches for clinical practice, and lastly, collaboration with government and policy-makers to create a receptive and synergistic health care system. Simply put, this plan must be more than aspirational. Let’s start.