Diabetic polyneuropathy (DPN) is the most common microvascular complication of diabetes, affecting approximately 50% of all people with diabetes (type 1 diabetes [T1D] and type 2 diabetes [T2D]).1 DPN is a major cause of disability due to sensory loss, gait instability and fall‐related injuries,1–5 and is the strongest risk factor for diabetic foot ulceration and amputation.1–4 Up to half of patients with DPN develop neuropathic pain, which is often very severe. DPN-related neuropathic pain has a major impact on patients’ quality of life, sleep and mood, and the related healthcare costs are substantial.2,6–8 Moreover, the healthcare expenditure for diabetic foot care in England is greater than the combined cost of three out of the four most common cancers.9 Unfortunately, there are no effective treatment strategies for DPN, and current diabetic foot screening diagnoses the condition late, when sensory loss is established and nerve damage is already severe.2 DPN has a significant impact on the individual, family and society.10 The management of DPN is in stark contrast to the other diabetes-related microvascular complications such as retinopathy and nephropathy, where both conditions can be diagnosed early and have effective disease-modifying treatments. The incidence of DPN is associated with potentially modifiable cardiovascular risk factors, including raised triglyceride levels, body-mass index, smoking and hypertension.11 Early identification of DPN could offer an opportunity for patients with diabetes to actively alter their lifestyles and to implement improved foot care before the onset of significant morbidity. The importance of early diagnosis has been demonstrated in diabetic retinopathy, which is no longer the leading cause of working age blindness in the UK as a result of the implemented national screening programme.12 However, leg amputations as a result of diabetic foot disease are increasing annually;13 a recent report stated that 20 amputations are performed per day in the UK.14 Furthermore, it has been estimated that 80% of amputations in England could be prevented through improved healthcare and management of diabetes.14
This article aims to discuss the challenges of diagnosing and managing DPN, and reviews current and emerging lifestyle interventions and therapeutic options.
Challenges of diagnosing diabetic polyneuropathy
A growing body of literature has shown that the onset of DPN occurs in patients in prediabetic states (those with impaired fasting glucose and impaired glucose tolerance).15,16 It is currently impossible to instigate a screening programme at this stage due to lack of resources, but it is likely that millions of people with prediabetes have polyneuropathy. This presents a major challenge, since early diagnosis may be key to improving disease outcomes, as advanced disease is impossible to reverse.4 Once DPN can be detected via clinical examination, it may be too advanced for any intervention to reverse or halt the process as nerves take years to mature and specialise, and once damaged or lost, they cannot regenerate.4
In clinical practice, the diagnosis of DPN is made on the basis of questionnaires and clinical examination, the latter involving the use of a tuning fork, patella hammer, pinprick sensation and monofilament examination.17–9 These tests are simple, cheap and widely available, but are not reproducible and cannot diagnose milder, ‘subclinical’ DPN.3 However, the landmark Toronto consensus meeting in 2009 recommended the use of objective measures in order to make a confirmed diagnosis of DPN, such as nerve conduction testing, which is necessary for clinical research or in the event of diagnostic uncertainty.17 Additionally, the modified Toronto Clinical Neuropathy Score (TCNS) has been shown to be a valid and reliable measure of polyneuropathy.18 Moreover, other clinical scoring questionnaires can be helpful; the DN4 questionnaire developed by the French Neuropathic Pain Group is administered by a clinician, takes less than 5 minutes to complete and is an effective way of screening for painful DPN.19
There are a number of established and emerging markers of DPN, including skin biopsy and corneal confocal microscopy.17 The latter provides a non-invasive quantitative method of detecting DPN and has been found to be more sensitive in assessing nerve repair than other standard measures.20 Corneal confocal microscopy and skin biopsy both detect nerve fibre loss in recently diagnosed T2D, but in different patients, suggesting a patchy pattern of small fibre neuropathy.21
If a patient is diagnosed with DPN, regular foot examinations should be performed to determine the person’s risk of developing diabetic foot ulceration. A large body of clinical trial evidence supports the effectiveness of screening and structured healthcare strategies in reducing amputations and ulcerations.22–5 Diabetic foot screening is recommended in many clinical practice guidelines, but there is considerable heterogeneity in terms of levels of evidence and grades of recommendation.26,27 As a result, there is currently no robust screening system in the UK; fewer than half of National Health Service local commissioners provide all three care structures recommended by the National Institute for Health and Care Excellence for patients with active diabetic foot disease.28 A one-stop microvascular assessment with combined foot, eye and renal screening may improve
foot-screening uptake.29
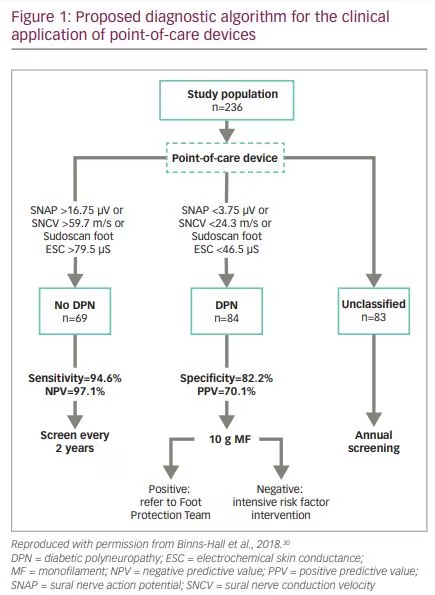
A recent study in Sheffield noted that retinal screening has a 90% uptake, but uptake of foot screening was poor at around 50%.30 Researchers therefore examined the feasibility and patient acceptability of a combined eye, foot and renal screening clinic (Figure 1).30 A total of 244 consecutive patients with either T1D or T2D attending retinal screening in hospital and community settings had their feet examined by a podiatrist before undergoing retinal photography whilst the mydriatic was taking effect. Assessments included dorsalis pedis and posterior tibial pulses, TCNS and 10-g monofilament testing. In addition, large‐fibre function was assessed using point-of-care devices such as Sudoscan™ (Impeto Medical, Paris, France) and DPNCheck® (Neurometrix Inc., Waltham, MA, USA).31,32 Results showed that the one‐stop microvascular screening clinic was feasible and had high patient acceptability and uptake. Moreover, the prevalence of distal symmetrical polyneuropathy, was underestimated by 10-g monofilament test (14.4%), compared with DPNCheck (51.5%). Combined assessment using DPNCheck and Sudoscan correctly classified 73.0% of patients, with a sensitivity of 93.2%. In addition, previously undiagnosed painful DPN was diagnosed in 25% of patients.30 Combined large- and small-nerve-fibre assessment using non-invasive, quantitative and quick point-of-care devices may be an effective model for the early diagnosis of distal
symmetrical polyneuropathy.
Lifestyle interventions for diabetic polyneuropathy
Optimal diabetes control is generally considered an essential first step in the prevention and management of DPN.33–6 In the 2003 Steno-2 study, a multifactorial intervention targeting glucose, cholesterol, weight and blood pressure in patients with T2D, was able to reverse retinopathy, nephropathy and autonomic neuropathy, but not polyneuropathy.37 This study was limited by the fact that the vibration perception threshold used in this study is not very sensitive and participants had advanced neuropathy. However, findings from the landmark Diabetes Control and Complications Trial Research Group (DCCT) study in 1993 showed that the risk of DPN was significantly reduced in patients with T1D using intensive glycaemic control.36 Unfortunately, even in people with a glycated haemoglobin (HbA1c) <8%, the incidence of DPN remained at 20% 8 years after study completion.33
Studies of patients with T2D have had inconsistent findings, perhaps because of inappropriate endpoints and recruitment of patients with advanced disease. A 2012 Cochrane review concluded that enhanced glucose control reduces the incidence of clinical neuropathy, although this was not statistically significant (p=0.06). However, enhanced glucose control has been shown to significantly reduce nerve conduction and vibration threshold abnormalities.38 Early intervention is likely to be key to successful outcomes in patients with T2D and DPN. A study published in The Lancet in 1971 showed that glucose control intervention in patients with newly diagnosed T2D shows improvement in nerve conduction within 6 months.39 More recently, the Look AHEAD (Action for Health in Diabetes) trial (n=5,145), a long-term intensive lifestyle intervention programme designed to achieve and maintain weight loss, resulted in a significant decrease in questionnaire-based DPN in obese and overweight adults with T2D.40 Furthermore, in the Bypass Angioplasty Revascularization Investigation 2 Diabetes (BARI 2D)
trial (n=2,159), the group of patients treated with insulin-sensitising therapies had a significantly reduced incidence of DPN compared with those receiving insulin-providing treatments. There was a significant association between changes in the Michigan Neuropathy Screening Instrument (MNSI) questionnaire and changes in body weight, HbA1c and serum lipids.41 Moreover, a study by the Diabetes Prevention Program Research Group (n=2,890) showed that lifestyle interventions improved autonomic nervous system function.42
Current and evolving treatments for diabetic polyneurophathy
Increased understanding of the pathophysiology of DPN has elucidated many potential therapeutic targets. It is known that prolonged hyperglycaemia results in a downstream metabolic cascade that causes peripheral nerve injury through an increased flux of the polyol pathway, enhances production of advanced glycation
end‐products, release of cytokines, activation of protein kinase C and increased oxidative stress.43 Despite this, there are no approved
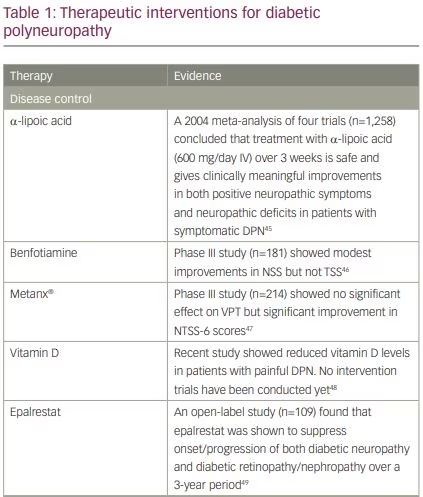
disease-modifying treatments for the management of DPN. Many clinical trials of pathogenic treatments have been performed but without success.44 The treatments supported by the strongest evidence are summarised in Table 1.45–60 A 2016 systematic review concluded that α-lipoic acid, opioids, botulinum toxin A, mexidol, reflexology and Thai foot massage had significant beneficial effects on DPN of the feet.61 However, of these, only α-lipoic acid, whose benefits are believed to derive from its multiple antioxidant properties,62 is supported by a robust body of evidence. A 2004 meta-analysis of four trials comprising n=1,258 patients (α-lipoic acid n=716; placebo n=542) concluded that treatment with α-lipoic acid (600 mg/day, intravenous) over 3 weeks is safe and resulted in clinically meaningful improvements in both positive neuropathic symptoms and in neuropathic deficits, in patients with symptomatic DPN.45 Further studies are needed to determine the effectiveness of α-lipoic acid in reducing pain and/or preventing the progression of DPN. At present, α-lipoic acid is not approved by the US Food and Drug Administration (FDA) or European Medicines Agency (EMA) for the treatment of DPN.
Vitamin B12 deficiency has been associated with significant neurological pathology, especially polyneuropathy, but oral vitamin B12 supplements have not been found to be beneficial in DPN.63 Some studies have suggested that benfotiamine – a lipid-soluble derivative of vitamin B1 (thiamine)46,64 – and Metanx® (L-methylfolate + pyridoxal 5-phosphate + methylcobalamin)47 may be effective for the alleviation of polyneuropathy symptoms, but these agents are not approved by the FDA/EMA as the studies are not conclusive due to issues with trial design and reproducibility. The 2017 American Diabetes Association position statement does not advocate their use.2
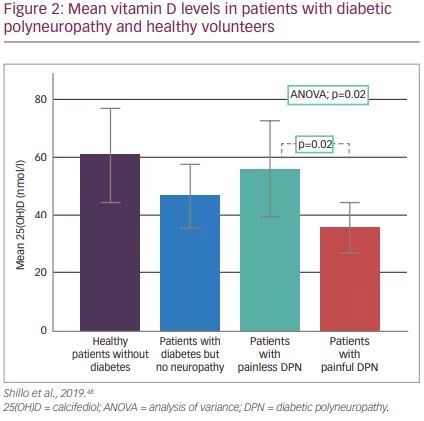
A 2015 systematic review and meta-analysis concluded vitamin D deficiency appears to be associated with DPN in T2D patients.65 However, included studies did not account for potential confounding factors such as seasonal sunlight exposure and daily activity. More recently, a study that accounted for these confounding factors divided patients into four groups: patients with diabetes but no neuropathy, healthy patients without diabetes, patients with painless neuropathy and patients with painful neuropathy. Vitamin D levels were significantly lower in those with painful neuropathy (Figure 2).48 Randomised, placebo-controlled intervention studies are now needed to determine whether vitamin D replacement improves neuropathic pain in vitamin D-deficient patients and can lead to nerve repair.
Aldose reductase is an important enzyme in the polyol pathway, which is involved in glucose metabolism.66 Consequently, aldose reductase inhibitors have been investigated as treatments for patients with DPN. Epalrestat, an aldose reductase inhibitor, is marketed in Japan, China and India but not approved by the FDA or EMA because the only large multicentre study investigating its efficacy and safety was an open-label study.49 A recent Japanese phase III study (n=557) investigated another aldose reductase inhibitor, ranirestat.67 While nerve conduction velocity was improved in the ranirestat group, no improvement in DPN signs and symptoms compared with placebo were observed during the 52 weeks of treatment.67
Pain management in diabetic polyneuropathy
The optimum management for neuropathic pain in DPN is uncertain, which is reflected in the variation in clinical guidelines and recommendations. Most guidelines recommend tricyclic agents,50 serotonin-norepinephrine reuptake inhibitors (duloxetine was the first medication to be approved specifically for the treatment of DPN)51–3 or α-2-delta ligands (gabapentin or pregabalin) as first-line agents, followed by opioids and topical treatments.57,68 However, these treatments have limitations, including discontinuation due to adverse events; lengthy dose-titration periods; drug–drug interactions; the need for frequent administration; and the risk of addiction, abuse and withdrawal symptoms.69,70 A recent systematic review and meta-analysis concluded that modest efficacy, large placebo responses, heterogeneous diagnostic criteria, and poor phenotypic profiling has led to disappointing outcomes in clinical trials, and that inadequate response to drug treatments constitutes a substantial unmet need in all patients with neuropathic pain.71
Antiepileptic drugs have been used in pain management since the 1960s and some seem to be particularly effective for neuropathic pain. As noted previously, gabapentin and pregabalin are approved by some regulatory bodies (only pregabalin is approved by the FDA and EMA) for use in DPN, but are limited by side effects.54,55 Mirogabalin is a novel
α-2-delta ligand, and may be associated with fewer central nervous system side effects. It has recently received approval in Japan after causing significant reductions in DPN pain in a phase III trial.72 Lacosamide, an anticonvulsant that enhances the slow inactivation of sodium channels, has been investigated for use in painful DPN but failed to obtain regulatory approval because it has limited efficacy.73 Oxcarbazepine, which acts upon voltage-gated sodium channels, has also been investigated for the treatment of painful DPN, but initial evidence in support of its use is of very low quality.74 It is worth noting however, that a 2014 study suggested that the efficacy of oxcarbazepine depended on pain phenotype.58 The study showed that oxacarbazepine was not beneficial in patients with neuropathic pain and the
non-irritable nociceptor phenotype but was effective for those with the irritable-nociceptor phenotype.58
Lidocaine 5% patches (Lidoderm®, Endo Pharmaceuticals, Dublin, Ireland) are used in patients with painful DPN, although their use is approved only for patients with postherpetic neuralgia.59 The capsaicin 8% patch (Qutenza®, Averitas Pharma Inc., NJ, USA) is an adhesive patch that was approved in 2009 for the treatment of non-diabetic adults with polyneuropathy. It contains a high concentration of synthetic capsaicin, a selective agonist of transient receptor potential vanilloid 1 channel.75 A phase III, 52-week, open-label, safety study showed that capsaicin 8% patch treatment over 52 weeks was well tolerated with no negative functional or neurological effects.76 In a phase III study (n=369), one 30-minute capsaicin treatment led to modest improvements in pain and sleep quality that were sustained over 3 months.77 As a result of these data, in 2015, Qutenza’s licence was extended to include patients with diabetes.60 An ongoing study funded by Diabetes UK is investigating whether repeated applications of the patch can not only reduce pain but also prevent or modify the underlying nerve damage.78
NaV1.7 is a voltage-gated sodium channel that is expressed in nociceptors and plays an important role in pain signalling in humans.79 Gain of function abnormality of this channel can cause congenital pain conditions such as congenital erythromelalgia, which is characterised by severe, continuous pain.80 Moreover, nonsense polymorphisms of these channels can cause congenital insensitivity to pain. NaV1.7 may also play a role in painful peripheral neuropathies, including painful DPN. In a recent study, 12 rare variants of NaV1.7 were found in patients with painful DPN, whereas none were seen in patients with painless DPN.81 Selective blockers of NaV1.7 are currently in clinical development as novel analgesics, although this quest is proving challenging.82
Various forms of electrical stimulation have been used to manage pain in DPN, including transcutaneous electrical nerve stimulation, percutaneous electrical nerve stimulation and frequency-modulated electromagnetic neural stimulation. A 2013 review of eight studies concluded that stimulation may be an effective alternative and adjunctive therapy to current interventions for DPN.83
Currently, there is great interest in the field of pain research in identifying different stratification methods for patients with neuropathic pain. Brain imaging may enhance our understanding of the different pain phenotypes associated with painful DPN and may offer a means to stratify pain treatment strategies. A recent Japanese study found that patients with painful DPN had increased anterior cingulate cortex perfusion.84 Moreover, they found that greater baseline activation in this brain region was predictive of treatment responsiveness to duloxetine treatment. Another recent study examined the sensory cortex of the brain using functional magnetic resonance imaging response to thermal pain. The study found that patients with insensate painful DPN
(i.e. lower-limb sensory loss and neuropathic pain) had the lowest somatosensory cortical thickness, compared with all other patients, including those with sensate painful DPN (i.e. relatively preserved lower limb nerve function with features of neuropathic pain).85 Furthermore, there was functional reorganisation of the somatosensory cortex in the insensate painful DPN patient group. This study was of great interest as it demonstrated a relationship between clinical painful DPN phenotype and structural and functional abnormalities within the
somatosensory cortex.
It is clear that the treatment of painful DPN is an important unmet need. Given the complexity of DPN, inhibition of single pathways may be insufficient and combination therapy may be needed. The
OPTION-DM study (optimal pathway for treating neuropathic pain in diabetes mellitus; trial registration identifier: ISRCTN17545443) is a head-to head, double-blind, cross-over trial that aims to determine the most clinically beneficial, cost-effective and best tolerated treatment pathway for painful DPN. Three pathways will be evaluated: amitriptyline supplemented with pregabalin, pregabalin supplemented with amitriptyline, and duloxetine supplemented with pregabalin.86 The study is due to complete in July 2020.
Summary and concluding remarks
The rising global burden of diabetes is responsible for an increase in the prevalence of DPN and painful DPN. Despite this, DPN continues to be underdiagnosed and undertreated, and thus imposes a substantial health and economic burden. Major public health initiatives may be needed to target modifiable risk factors and educate patients with diabetes on the importance of foot screening. The recent success of the combined eye, foot and renal screening clinic in Sheffield, England, is an example of how foot screening can be integrated with other microvascular assessments.30
The pathogenesis of DPN is complex, and investigations of potential disease-modifying therapies have proved frustrating. Inhibition of single metabolic factors may be insufficient for the treatment of DPN and combined therapeutic approaches may be needed. While there are no new disease-modifying therapies on the horizon, management of patients with DPN must depend on individual requirements and on the presence of other comorbidities. Pain resolution is of paramount importance for patients with painful DPN, and there is a clear unmet need for new therapeutic options to improve current standard of care. The available treatments such as antidepressants, antiepileptic drugs and opioids are often limited by contraindications and safety issues, and often fail to achieve adequate pain relief. The finding that therapeutic effect may depend on pain phenotype may increase the effectiveness of emerging medications. In addition, innovative approaches such as the topical capsaicin patch, new methods of electrical stimulation and novel therapeutic targets such as NaV1.7 appear promising.







