Rare catecholamine-secreting tumours that arise from the adrenal medulla and sympathetic ganglia are referred to as pheochromocytomas (PHEO) and paragangliomas, respectively.1 The annual incidence of PHEO in general population is approximately 2–8 cases per million.2 The age onset is typically the fourth decade of life.3 It occurs in less than 0.2% of patients with hypertension.4–7 Symptoms are present in 50% of patients, result from excessive catecholamine secretion and are typically paroxysmal.1 The classic triad is headache, sweating and tachycardia. However, hypertension is the most common sign of PHEO, with only 5–15% of patients presenting with normal blood pressure.1
This catecholamine-secreting tumour is, in the majority of cases, sporadic; however, 40% are hereditary and 15 different susceptibility genes have been reported since 1990.8,9 The presence of a genetic syndrome may raise the suspicion of a hereditary PHEO, associated to von Hippel–Lindau Syndrome (VHL), multiple endocrine neoplasia type 2 (MEN2) or neurofibromatosis type 1 (NF1). The frequency of PHEO in VHL is 10–20%, in MEN2 is 50%, and in NF1 is 0.1–5.7%.3,10–19 However, the absence of a genetic syndrome should not exclude a hereditary PHEO, since it can result from mutation of several other genes, such as SDHA, SDHB, SDHC, SDHD, SDHAF2, FH, HIF–2a, EGLN-1, EGLN-2, KIF1B, MAX and TMEM127.8,9,20,21
NF1 is an autosomal dominant disease, present in 1 in 2,600 to 1 in 3,000 individuals.17,22 One-half of cases are inherited, and the other half results from de novo mutations of the NF1 gene.23 The main clinical manifestations are café-au-lait macules, axillary/inguinal freckling, Lisch nodules and neurofibromas.23 In patients with NF1 and hypertension the incidence of PHEO increases to 20–50%. In fact, 60% of cases of NF1 with PHEO have persistent hypertension.3 Recent prospective studies by Zinnamosca et al.24 and Képénékian et al.25 suggested that PHEO prevalence in NF1 was underestimated, since they obtained a prevalence of 14.6% and 7.7%, respectively.
Case presentation
We present a 31-year-old female patient with NF1 diagnosed upon clinical criteria before age 10; depression–anxiety syndrome and left ovarian dysgerminoma were diagnosed at age 26. The patient underwent multiple surgeries for excision of cutaneous and brachial plexus neurofibromas and a hysterectomy, as well as salpingo-oophorectomy in September 2015. She was medicated with fluoxetine, valerian and diazepam. She is a single mother of a boy, who was 6 years old at the time and presumably healthy. Her father had clinically diagnosed NF1 and two aunts had thyroid nodules under surveillance.
In July 2016, the patient was referred for genetic analysis due to the detection of a bilateral tubal epithelial atypical lesion in the histology of the salpingo-oophorectomy specimen, which raised the suspicion of a hereditary syndrome associated with BRCA mutation. In the same appointment, because of family history, the patient brought a thyroid ultrasound done in the context of primary care, which revealed a 4 mm incidental nodule. Upon this result she was referred to the endocrinology department to further evaluation.
The first endocrinology appointment was in August 2017. The patient described episodes of palpitations, headaches, paleness, tremor, nausea, dizziness, hypertensive spikes and loss of consciousness within the last 6 months. She also reported surgical complications during excision of a neurofibroma with intra-operative hypertensive spikes. At physical examination, blood pressure was 120/80 mmHg, multiple café-au-lait spots were noticeable through all the skin and she had a subcutaneous nodule in the right abdominal flank. The initial study (October 2017) revealed thyroid-stimulating hormone 1.570 µIU/mL (reference range 0.358–3.740 µIU/mL), free T4 1.11 ng/dL (0.76–1.46 ng/dL), total urinary metanephrines 7,436 µg/24 hours (<785 µg/24 hours), fractionated urinary metanephrines 4,039 µg/24 hours (<341 µg/24 hours), and urinary normetanephrine 3,397 µg/24 hours (<444 µg/24 hours).
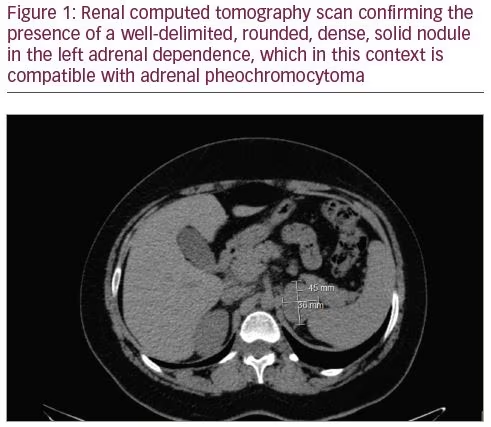
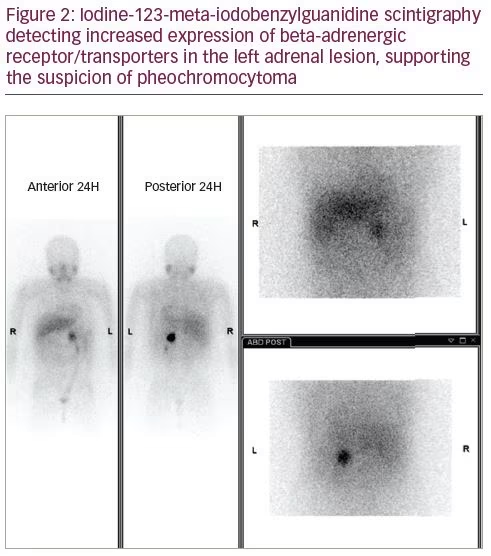
Under the suspicion of a PHEO, we asked for urinary and plasma metanephrines, a renal computed tomography (CT) scan and an iodine-123-meta-iodobenzylguanidine scintigraphy (MIBG). The analytic results of January 2018 were: plasma metanephrines 728 µg/mL (<65 µg/mL), plasma normetanephrine of 1,741 µg/mL (196 µg/mL), total urinary metanephrines 6,082 µg/24 hours (<785 µg/24 hours), fractionated urinary metanephrines 3,572 µg/24 hours (<341 µg/24 hours), urinary normetanephrine 2,510 µg/24 hours (<444 µg/24 hours). Renal CT scan (Figure 1) showed a left adrenal nodule and MIBG (Figure 2) detected intense uptake by a left adrenal lesion, compatible with PHEO. A posterior evaluation by cardiology concluded that there were no electrocardiographic or echocardiographic alterations.
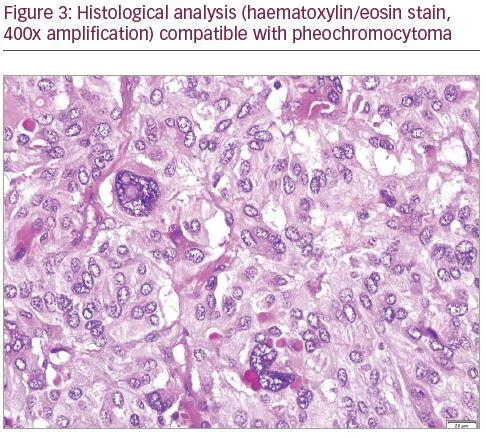
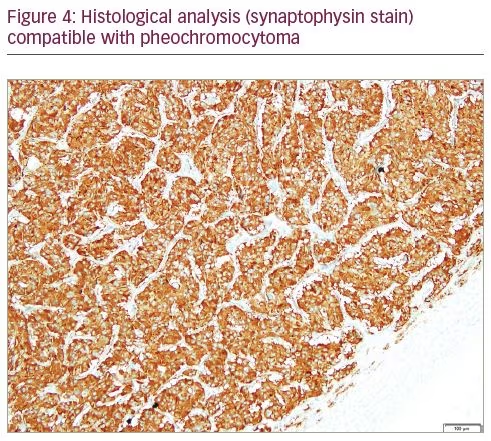
In March 2018, the patient underwent left adrenalectomy, after treatment with phenoxybenzamine for 7 days, until 24 hours before surgery. In the last 24 hours, salt consumption was liberalized and the patient was given a 3 L supply of intravenous saline solution. After an uneventful surgery, she was admitted at the intermediate care unit of the emergency room. Histological analysis (Figures 3 and 4) of the 48 g, 5 x 4.5 x 2.5 cm specimen showed a 4 x 2 x 1 cm adrenal gland with a 5 cm nodule consistent with PHEO, with a Pheochromocytoma of the Adrenal Gland Scoring Scale of 1 (pT2 Nx).
One month after surgery, urinary catecholamines and metanephrines normalized: adrenaline 3 µg/24 hours (<18 µg/24 hours), noradrenaline 24 µg/24 hours (<76 µg/24 hours), dopamine 120 µg/24 hours (<390 µg/24 hours), total metanephrines 222 µg/24 hours (<785 µg/24 hours), fractionated metanephrines 20 µg/24 hours (<341 µg/24 hours) and normetanephrine 202 µg/24 hours (<444 µg/24 hours). Meanwhile, the genetic study from July 2018 was negative for BRCA1 and BRCA2 mutations. Nevertheless, testing did identify a mutation in exon 18 of the NF1 gene (c.2033dup [p.lle679Aspfs*21]), further confirming the clinical diagnosis and now allowing predictive testing in her family, including her son. There is a 50% chance of transmission of this genetic variant to offspring and, for this reason, the patient’s son is waiting for genetic testing. The geneticist considered the ovarian dysgerminoma as a clinical manifestation of NF1. The patient’s father was not screened for PHEO.
One year after surgery, urinary catecholamines and metanephrines remained normal (adrenaline 4 µg/24 hours [<18 µg/24 hours], noradrenaline 31 µg/24 hours [<76 µg/24 hours], dopamine 123 µg/24 hours [<390 µg/24 hours], total metanephrines 261 µg/24 hours [<785 µg/24 hours], fractionated metanephrines 24 µg/24 hours [<341 µg/24 hours] and urinary normetanephrine 237 µg/24 hours [<444 µg/24 hours]). Currently, the patient is under long-term follow-up, with annual metanephrines.
Discussion
NF1 diagnosis is based upon clinical criteria developed by the US National Institutes of Health (two or more of the following: six or more café-au-lait macules, two or more neurofibromas of any kind or one plexiform neurofibroma, axillar or inguinal freckles, optic glioma, two or more Lisch nodules, bone lesions and first-generation familiar history of NF1).25 Genetic testing can be performed to confirm the diagnosis in cases that do not meet clinical criteria or for direct screening in family members. Hypertension, very common in these patients, is in the majority of cases of reno-vascular aetiology as a result of chronic vascular lesions. However, PHEO can be a not so rare cause of hypertension in these patients.3,23–25 Retrospective studies, dating back to 1999, described a prevalence of PHEO in NF1 of 0.1–5.7%, which increased to 20–50% if hypertension was present.3,17–19 In fact, persistent hypertension is seen in about 60% of patients with NF1 and PHEO.26 The incidence in autopsy series reached 3.3–13%.3,27 Recently, prospective studies found a prevalence of PHEO of 14.6%24 and 7.7%25 in patients with NF1.
The classical triad of this tumour of the adrenal medulla is headaches, diaphoresis and palpitations. Nevertheless, the clinical manifestations may vary as a result of variable levels of catecholamines in circulation and includes paleness, anxiety, dyspnoea, fever, orthostatic hypotension, syncope, constipation, nausea, vomiting, epigastric pain, polyuria, anorexia and weight loss.1 Approximately 13% of patients are normotensive, leaving the other 87% with hypertension that can be persistent in 50–60% of the cases.3,28 However, 22% of patients with PHEO are asymptomatic.3,28
Hereditary PHEO may be associated with VHL, MEN2 or NF1; with the association with NF1 the least prevalent.8,9,21 The characteristics between sporadic PHEO and associated with NF1 are similar, namely the age of onset and the prevalence of malignant disease (10–12%).9,11,19,27 Compared with VHL and MEN2, PHEO in NF1 is more frequently unilateral (90% of cases), diagnosed incidentally, later in life and in patients with no familial history.9,11,19,27,29,30 These differences might be related to the fact that current guidelines do not recommend routine biochemical or imaging screening of PHEO in asymptomatic/normotensive patients with NF1, despite the recommendation of doing it for patients with VHL or MEN2.31
Recent studies reinforce the idea that PHEO prevalence in NF1 is underestimated and it may have atypical presentation (asymptomatic/normotensive or associated with non-specific symptoms). Patients with undiagnosed PHEO are at risk of developing encephalopathy, cerebrovascular disease, cardiac arrhythmias, congestive heart failure, acute myocardial infarction, myocarditis, cardiomyopathy, pulmonary oedema and shock. Most of these are life-threatening cardiovascular complications that result from PHEO crisis triggered by tumour manipulation, pregnancy, drugs or metastatic disease.11,27,30,32–34 Furthermore, patients with PHEO associated with NF1 have more intraoperative hypertension variability and perioperative complications than patients with other genetic syndromes.35 As a preventive measure, authors highlight the important role of routine screening in the absence of hypertension or classical symptoms, particularly prior to elective surgeries, during preconception planning or early in pregnancy.11,27,29,30,32,34
For biochemical diagnosis it is necessary to confirm the inappropriately elevated secretion of catecholamines and its metabolites through free plasma metanephrines or fractionated urinary metanephrines. Localization studies should be done with imaging, such as CT scan or magnetic resonance imaging; or functional exams, such as iodine-123-meta-iodobenzylguanidine scintigraphy, octreotide scan or fluorodeoxyglucose-positron emission tomography.21,28,33 The Endocrine Society recommends genetic testing for patients presenting with
PHEO/paraganglioma and family history or clinical features suggesting an hereditary syndrome.33 Despite this, it is considered that sequencing NF1 gene is complex, expensive and, as there is a high incidence of de novo mutations (around 50%), clinical diagnosis of NF1 is sufficient.21,33
The treatment consists in adrenalectomy. Preoperative preparation includes alpha-adrenergic blockade initiated 7–14 days before. Beta-blockers can be used if reflux tachycardia results from treatment with alpha-blockade. Liberalization of salt consumption and fluid intake are advised to prevent post-operative hypotension. Blood pressure, heart rate and plasma glucose levels should be monitored. Normal plasma or urinary metanephrines 2–4 weeks after surgery document therapeutic success.21,28,33
In terms of follow-up, the recommendations for patients with a prior diagnosis of PHEO advocate lifelong annual biochemical surveillance.11,28,33 Regarding previously published cases, we found two Portuguese cases highlighting the rarity and importance of high levels of suspicion of PHEO in NF1 in patients with36 and without37 hypertension or classical symptoms. In English-language literature there are 73 case reports from the last 18 years.30 In these cases, PHEO was more prevalent in women, with incidental presentation and unilateral. From the 73 patients, 49% did not report classical symptoms; 37% were normotensive; and 42% had major complications, such as myocardial infarction/myocarditis, metastatic PHEO, hypertensive crisis, heart/or multi-organ failure, cardiomyopathy, bleeding/vascular complications and death.30
Conclusion
Patients with NF1 have a genetic susceptibility to the development of PHEO and a high level of suspicion for PHEO is needed in symptomatic and hypertensive individuals with NF1. Biochemical Screening for PHEO by checking plasma or urinary metanephrines should be performed in all patients with NF1 who are hypertensive. Furthermore, systematic screening for PHEO might be considered in all patients with NF1, irrespective of their blood pressure, given the atypical presentation. Early identifications of PHEO could reduce the mortality and morbidity associated with PHEO crisis in patients with NF1; patients with NF1 can develop life threatening complications if PHEO diagnosis is missed.