Growth hormone deficiency (GHD) is an endocrine cause of short stature. GHD may be idiopathic, due to brain tumours affecting the pituitary gland, morphologic pituitary abnormalities, or genetic defects.1,2 The diagnosis of GHD is based on auxologic criteria and laboratory studies including tests of growth hormone (GH) secretion by stimulating GH release.3 The identification of central nervous system (CNS) tumours remains the primary purpose of neuroradiological imaging in the evaluation of children with GHD, but brain magnetic resonance imaging (MRI) also has a secondary role identifying abnormalities in the pituitary anatomy that aid the clinician in the diagnosis and prognosis.2,4,5
The Growth Hormone Research Society currently recommends that an MRI of the brain, with particular attention to the hypothalamic–pituitary region, should be carried out in any child diagnosed with GHD.3 However, there is no clear evidence to support these recommendations in patients with mild GHD (peak stimulated GH value between 5 and <10 ng/mL).6 The prevalence of pituitary abnormalities and brain tumours seems to be higher in patients with lower GH values in provocative testing.7 However, peak GH value as predictor of the presence or absence of brain MRI abnormalities has not yet been clearly defined.
Previous studies have been inconclusive, showing a wide variability in the prevalence of MRI abnormalities in patients with GHD, ranging from 25.9–100.0%.8–12 Due to this variability patients with mild GH values (peak stimulated GH value between 5 and <10 ng/mL) are unlikely to undergo a brain MRI. This is evidenced by the fact that in the clinical settings some healthcare providers will only obtain brain MRI if the patient has severe GHD (GH level <5 ng/mL) or if there are other risk factors present (such as other pituitary hormone deficiencies, severe headache, vision problems). Therefore, despite the recommendations from the Growth Hormone Research Society, when to obtain a brain MRI in patients with GHD remains controversial.8–12
The aim of this study was to evaluate peak GH levels, determined by provocative testing, as a predictor of the presence or absence of brain MRI abnormalities and to determine if a brain MRI should be performed in all patients with GHD. A secondary aim was to identify whether symptoms suggestive of brain abnormalities influenced this prediction.
Methods
Study population
This retrospective study included all paediatric patients diagnosed with GHD at a tertiary paediatric referral center, Children’s Mercy Hospital, Missouri, United States, from January 2008 to January 2015. The Institutional Review Board approved the research protocol. Informed consent was waived because of less than minimal risk. The conducted research is not related to either human or animal use. Chart reviews were based on International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes: 253.3 (GHD), 259.4 (dwarfism), 781.91 (loss of height), 783.41 (failure to thrive), and 783.43 (short stature). The diagnosis of GHD was based on growth failure and peak GH level on stimulation test of <10 ng/mL with two stimulants (either clonidine, arginine and/or glucagon). All patients with GHD underwent brain MRI with detailed examination of the hypothalamic–pituitary area at diagnosis.
Patients with GHD that did not have a brain MRI after the diagnosis of GHD was made were excluded from the study. Patients with known CNS anatomical abnormalities, history of cranial radiation and history of CNS trauma before the diagnosis of GHD were also excluded from the study. These particular exclusion criteria were applied as these parameters represent acquired causes of pituitary dysfunction in which GHD was expected to develop, and in which GHD was diagnosed after MRI and the lesion was treated.
Diagnosis of growth hormone deficiency
Prior to inclusion in this chart review, standing height was measured with a Harpenden stadiometer (QukckMedical, Issaquah, WA, USA). Height was calculated using the World Health Organization (WHO) child growth standards.3 Height velocity was calculated using Tanner’s growth charts.3 The following clinical and auxological assessment were considered characteristics of growth failure: height >3.0 standard deviation score (SDS) below the mean; height >1.5 SDS below the mid-parental height; height >2.0 SDS below the mean with a height velocity over 1 year >1.0 SDS below the mean for chronological age; or a decrease in height SDS of >0.5 over 1 year in children >2 years of age. In the absence of short stature, a height velocity >2.0 SDS below the mean over 1 year or >1.5 SDS sustained over 2 years.3 Chronic diseases, malnutrition and other endocrine disorders were excluded as the cause of the short stature.
The diagnosis of GHD was based on growth failure and peak GH levels of <10 ng/mL in GH stimulation test with two stimulants. GHD was divided in mild (peak GH level between 5–10 ng/mL) and severe (peak GH level <5 ng/mL).6 In patients that failed the GH stimulation test with one stimulant, but passed with a second stimulant and there was strong suspicion of GHD, a third stimulant was used. If these patients failed with two stimulants they were included in the study. Stimulants used were clonidine, arginine and glucagon. Stimulants were administered with the following doses: clonidine 0.1 mg/m2, oral; arginine 0.5 gm/kg, administered intravenously over 30 minutes; and glucagon 0.03 mg/kg, subcutaneous. GH levels were measured at 0, 30, 60, 90, 120, 150 and 180 minutes after the administration of stimulants.
All patients with diagnosis of GHD underwent a brain MRI, performing pre- and post-gadolinium per radiologist discretion, with enhanced T1- and T2-weighted images in axial, coronal and sagittal sections. All the scans were evaluated by the same method by one observer, and were later validated by two different blinded radiologists, and the consensus achieved. The size and signal intensities of the anterior pituitary were evaluated.
Pituitary gland height was measured in a plane perpendicular to the floor of the sella turcica to the highest point of the superior gland surface, usually at the point of insertion of the pituitary stalk. Pituitary hypoplasia was defined as a pituitary height SDS <-2. The pituitary stalk was classified as normal, thick, thin (hypoplastic), truncated or absent. The stalk was defined as hypoplastic if there was a significant reduction at any level in its diameter or irregularity along its length, but it remained in continuity with the pituitary. The neurohypophyisis was defined as normal or ectopically placed. Ectopia was diagnosed on the basis of absence of the normal posterior lobe high signal on T1 sequences in the sella turcica, and the presence of high signal in the infundibular recess of the third ventricle.3–5,7,9
Classification of brain abnormalities on magnetic resonance imaging
Brain MRI was classified as normal versus abnormal, depending on whether CNS anatomy was disrupted or not. If CNS anatomy was disrupted, this was further classified as associated versus not associated with pituitary gland. When associated with the pituitary gland, documentation was provided regarding pituitary hypoplasia/aplasia, anatomy of the stalk, and position of the posterior pituitary. Brain MRI abnormalities were divided in mild and severe. Mild abnormalities were classified as ones that were less likely associated with pituitary hormone deficiencies, and severe abnormalities were classified as those that are more likely associated with pituitary hormone deficiencies.2,4,8,9
Signs/symptoms suggestive of intracranial pathology included headache, vision problems (blurred vision, double vision, loss of peripheral vision), symptoms of pituitary hormone deficiency, central hypothyroidism, central diabetes insipidus, central adrenal insufficiency and hypogonadotropic hypogonadism.
Statistical analysis
Comparisons were made based on gender (male versus female), age (2–4 years, 5–9 years, 10–14 years and 15–18 years), peak GH value on GH stimulation test (mild versus severe GHD) and brain MRI results (normal versus abnormal, and mild versus severe abnormality). The comparison between groups was evaluated using independent samples t-test, Chi-square and Fisher’s exact test. This data is presented as mean ±SDS. The statistical significance was determined with an alpha of p<0.05. The analysis was done using SAS (version 9.4) and SPSS (version 22.0).
Results
Six hundred and twenty-eight charts were reviewed, and a total of 386 patients with GHD that underwent brain MRI, were included in the study. Two hundred and ninety patients were male (75.1%) and 96 patients were female (24.9%). Age of patients ranged from 2–18 years old.
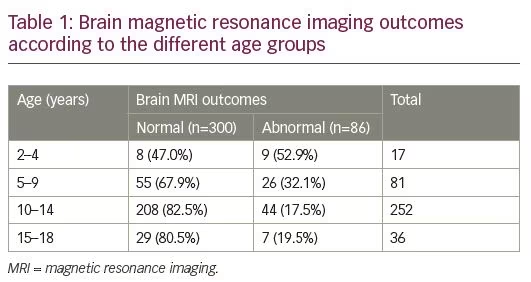
Of the 386 patients, 300 patients had a normal brain MRI (77.7%) and 86 patients had an abnormal brain MRI (22.3%). There was no difference in the prevalence of abnormal MRI between male and female patients (21.3% of males and 25.0% of females had an abnormal brain MRI; p=0.45). Brain MRI abnormalities were more prevalent between 2–9 years old (Table 1).
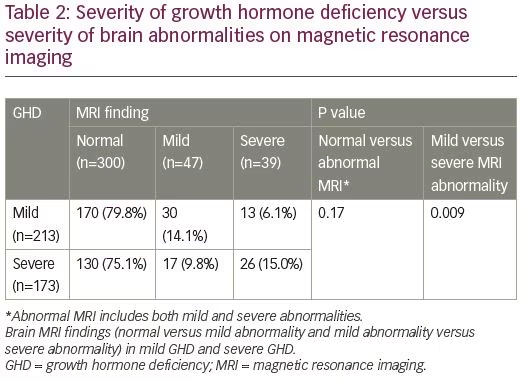
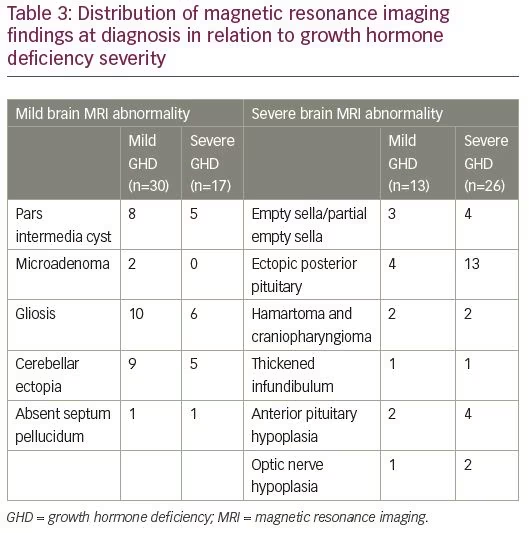
Of the 213 patients with mild GHD, 170 had a normal brain MRI (79.8%), 30 had mild brain MRI abnormality (14.1%) and 13 had severe brain MRI abnormality (6.1%). Of the 173 patients with severe GHD, 130 had a normal brain MRI (75.1%), 17 had mild brain MRI abnormality (9.8%) and 26 had severe brain MRI abnormality (15.0%) (Table 2). There was no difference in prevalence of MRI abnormalities between patients with mild GHD and severe GHD (p=0.17). Severe brain MRI abnormalities were more prevalent in patients with severe GHD compared to mild GHD (p=0.009). The distribution of brain MRI findings according to severity of GHD is depicted in Table 3.
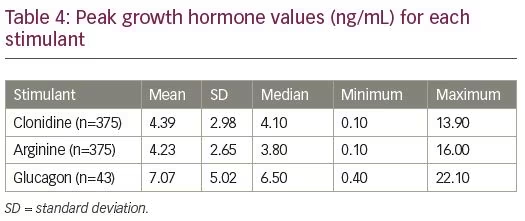
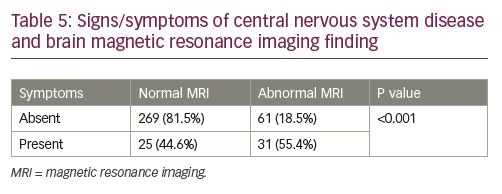
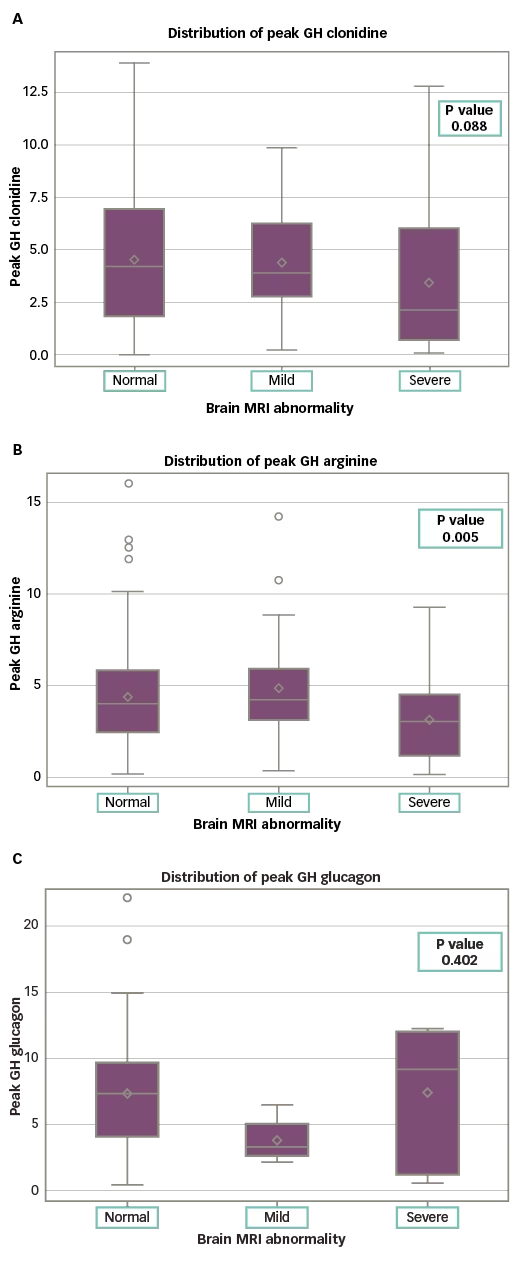
The difference in peak GH values was not statistically significant for patients with normal versus abnormal MRI, with any agent (clonidine: p=0.07; arginine: p=0.17; glucagon: p=0.42) (Table 4 and Figure 1). The presence of signs/symptoms concerning for CNS disease was predictive of MRI abnormality (55.0% of symptomatic patients had an abnormal MRI; p<0.001) (Table 5). Controlling for symptoms did not provide statistical significance when comparing the mild GHD versus the severe GHD groups. In the absence of symptoms, 81.9% of the patients with mild GHD had a normal MRI and 80.1% of the patients with severe GHD had a normal MRI (p=0.80). In the presence of symptoms, 37.0% of the patients with mild GHD versus 49.0% of the patients with severe GHD had an abnormal MRI (p=0.4).
Discussion
The results in our study population show that peak stimulated GH levels did not predict the presence or absence of brain MRI abnormalities. The prevalence of severe brain MRI abnormalities was higher in patients with severe GHD, but patients with mild GHD also had severe brain MRI abnormalities that are considered clinically significant. Even when controlling for symptoms and signs of CNS disease, there was no difference in prevalence of brain MRI abnormalities between patients with mild versus severe GHD. Previous studies have shown discrepant results regarding the prevalence of brain MRI abnormalities in patients with GHD and therefore recommendations as to when to obtain a brain MRI in these patients remains controversial.
Tillmann et al. investigated the relationship between pituitary appearance and the diagnosis of GHD. They assessed MRI scans along with GH status on stimulation tests in 110 patients. GHD was established with a peak GH level of <5.8 ng/mL, a lower cut off than the one used in our study. In Tillmann’s study, 79.2% of patients with GHD had hypothalamic–pituitary axis abnormality, which is much higher than in the population presented here. Even when comparing patients with severe GHD and severe brain MRI abnormalities, the prevalence in our study was 15.0%, compared to a prevalence of 54% in the study Tillmann et al. They also found that 46% of patients that were GH sufficient (GH level >5.8 ng/mL) had a brain MRI abnormality. They recommended obtaining brain MRI in all patients with GHD as an aid to diagnosis.13
Acharya et al.14 and Maghnie et al.15 had different findings from Tillmann’s study and showed that all patients with pituitary abnormalities had a peak GH level, determined by stimulation tests, of <3 ng/mL (prevalence 53%). These results suggest that only patients with peak GH level of <3 ng/mL should undergo brain MRI. Another study from Maghnie et al. showed similar results, with data suggesting that GHD is more severe in patients with ectopic posterior pituitary, indicating that an abnormal brain MRI is highly predictive of severe GHD.16 Based on these studies, brain MRI can contribute to the prediction of the pattern and severity of GHD.14–16
Lo et al. found no significant clinical difference in the severity of brain MRI abnormalities in patients with severe GHD and mild GHD. In the severe GHD group there were 23% of patients with pituitary hypoplasia, compared to 11% in the mild GHD group. They also found one interrupted stalk and one ectopic posterior pituitary in the severe GHD group, but none in the mild GHD group. As there were no significant differences between these groups, it was recommended to obtain a brain MRI in all patients with GHD.17 This outcome is similar to our results, showing no difference in the prevalence of brain MRI abnormalities in patients with mild versus severe GHD (patients with severe GHD had a prevalence of brain MRI abnormalities of 24.9%, compared to 20.2% for the patients with mild GHD; p=0.17).
Another study, from Canada, showed a high prevalence of brain MRI abnormalities in patients with GHD (80%). The higher prevalence of MRI abnormalities is likely due to the more stringent criteria used to diagnose GHD in this institution, including peak GH level of <8 ng/mL on three tests – two pharmacologic and one physiological.18 These criteria are different from the ones used in our study which likely explains the difference in prevalence. Their data suggest that brain MRI should be done in all patients with GHD, but it is not clear if the prevalence of brain MRI abnormality is inversely proportional to the peak GH level on stimulation test.
Rosenfeld et al. investigated which patients with GHD are most likely to benefit from MRI. Their study included 100 patients who failed GH screening tests.6 They found a significant relationship between the presence of risk factors, maximal GH level of <5 ng/mL, and sellar defects. With no risk factors, MRI scans showed normal findings in 15 of 17 patients with maximal GH of <5 ng/mL, in 33 of 34 patients with GH between 5–10 ng/mL, and in all 35 patients with GH of >10 ng/mL. They concluded that MRI scans should be obtained in any child with risk factors (such as multiple pituitary hormone deficiency, hypoglycaemia, ophthalmologic anomalies, low-stimulated GH or acquired growth failure). Otherwise, asymptomatic children with growth delay and maximal GH of >10 ng/mL do not need routine MRI screening. Patients with mild GHD and risk factors should undergo an MRI. Patients with mild GHD and no risk factors, are unlikely to benefit from MRI.6 Our results are similar to those of Rosenfeld et al., as we found that symptoms of CNS disease are predictive of brain MRI abnormality (55% of symptomatic patients had an abnormal MRI; p<0.001) (Table 5). However, in our population, the prevalence of MRI abnormalities was similar in the mild and severe GHD groups, independently of the symptoms, which differs from Rosenfeld’s recommendations regarding obtaining brain MRI in patients with mild GHD only if they are having symptoms concerning of CNS disease.
In our study, 77.7% of patients with GHD had a normal brain MRI. This is similar to other studies that have found that a normal MRI is the most common finding in children with GHD.19,20 As with our study, the abnormalities found in other studies include empty sella/partial empty sella, posterior pituitary ectopy, decreased pituitary size, thickened infundibulum, optic nerve hypoplasia and tumours like hamartoma and craniopharyngioma.2,4,7,19,21,22
One of the limitations of this study is that the analysis was not confined to GH levels of <10 ng/mL. Our data show some GH levels of >10 ng/mL as a result of patients who failed a GH stimulation test with one stimulant but passed with a second stimulant, in which case, if there was strong suspicion of GHD, a third stimulant was used. However, this serves to show that GH levels on stimulation tests are not a good predictor of brain MRI abnormalities.
Conclusion
Based on conflicting findings in previous research studies, the topic of conducting brain MRI on all paediatric patients with GHD remains controversial. The identification of CNS tumours remains the primary purpose of neuroradiological imaging in the evaluation of children with GHD but brain MRI also has a secondary role identifying abnormalities in the pituitary anatomy such as irregularities in pituitary size, location and stalk connection. These structural abnormalities help validate the diagnosis of GHD. Finding structural abnormalities also aids in the prognosis and follow-up of patients as they have higher risk of developing other pituitary hormone deficiencies and adult GHD.2,3,12
The Growth Hormone Research Society published a consensus guideline for the diagnosis and treatment of GHD in paediatric patients in 2000. They recommended that an MRI of the brain with particular attention to the hypothalamic–pituitary region should be carried out in any child diagnosed with GHD’.3 Until the present study there was no clear evidence to support these recommendations. Previous series have reported an incidence of MRI abnormalities between 25.9–100% in patients with GHD.8–10,12
With the discrepancy of the results in previous studies, some investigators have advocated MRI studies for any patient with GHD,13,17,18 while others recommend imaging studies after confirmation of a severe defect in GH secretion,6,14–16 or after clinical and biochemical data suggests intracranial pathology.
Our results show that the severity of GHD based on peak GH levels determined by stimulation tests did not predict the presence or absence of MRI abnormalities in our study population. The prevalence of severe brain MRI abnormalities was higher in patients with severe GHD, but patients with mild GHD also had severe brain MRI abnormalities that are considered clinically significant. Even when controlling for symptoms and signs of CNS disease, there was no difference in prevalence of brain MRI abnormalities between patients with mild versus severe GHD. In conclusion, based on these results we recommend obtaining brain MRI in all patients with clinical and axiological data suggestive of GHD regardless of the peak GH level on stimulation test.