Polycystic ovary syndrome (PCOS) is a heterogeneous syndrome, considered the most common endocrinopathy in women of reproductive age, with a prevalence of 6–8% in premenopausal women.1,2 However, long-term sequelae are extended beyond the reproductive axis, being present from birth to senescence. Consequently, early detection and treatment options are in high demand to prevent long-term complications. This review aims to summarize the evidence published regarding the cardiovascular risk factors that accompany PCOS.
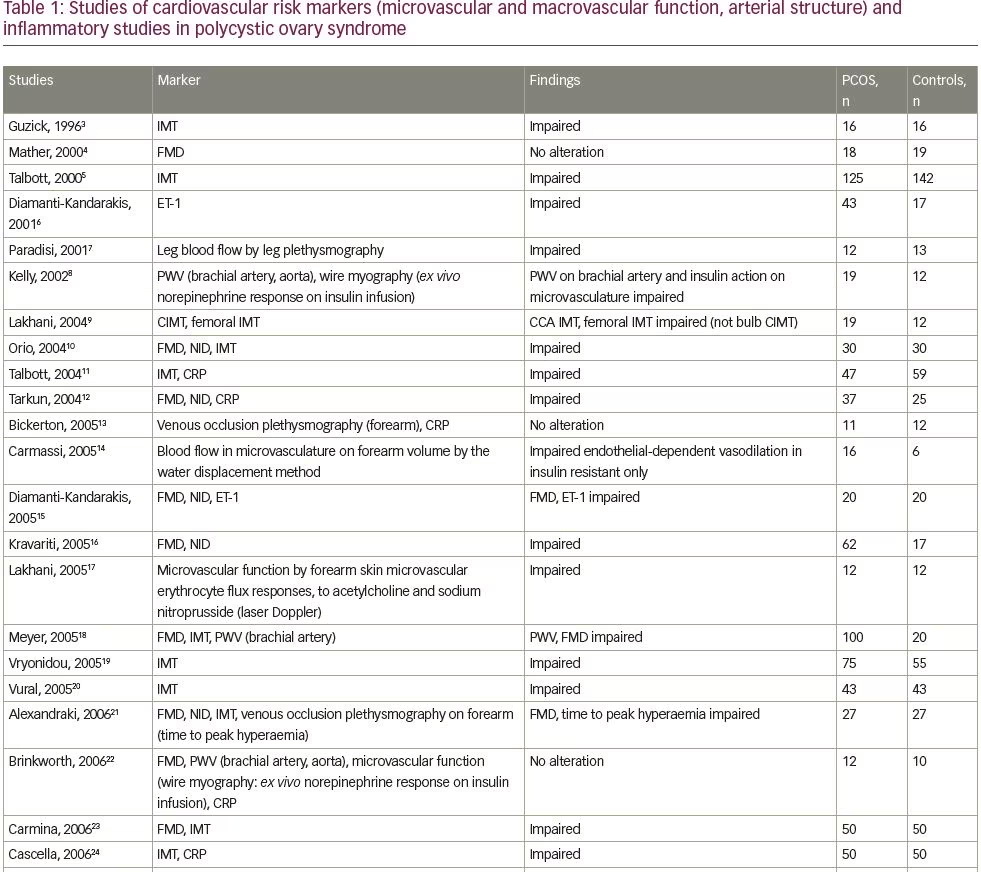
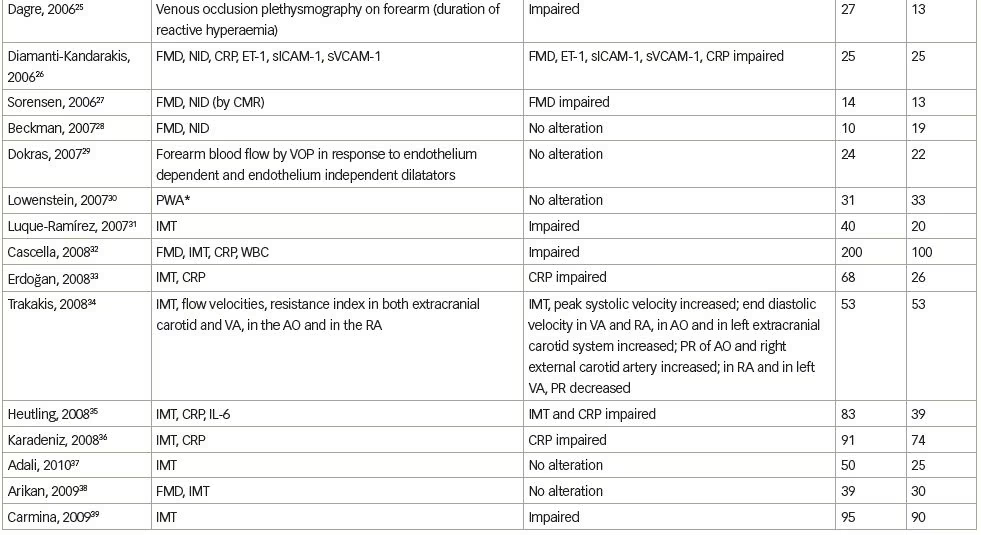
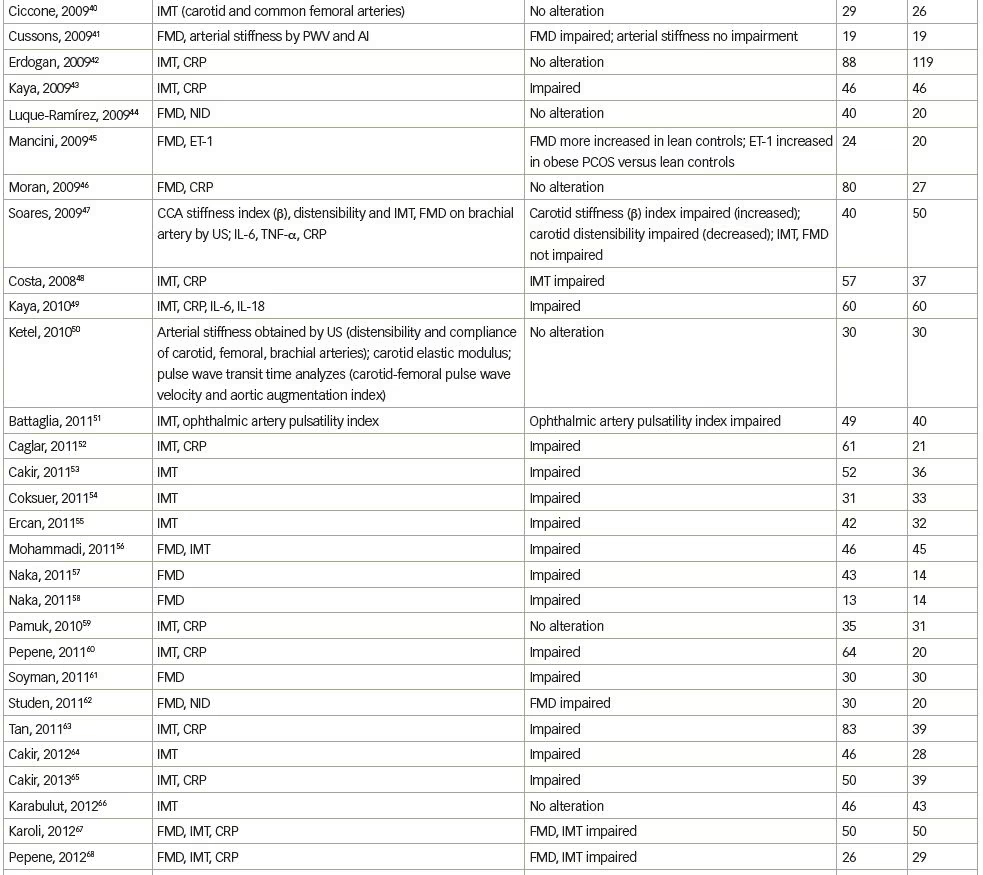
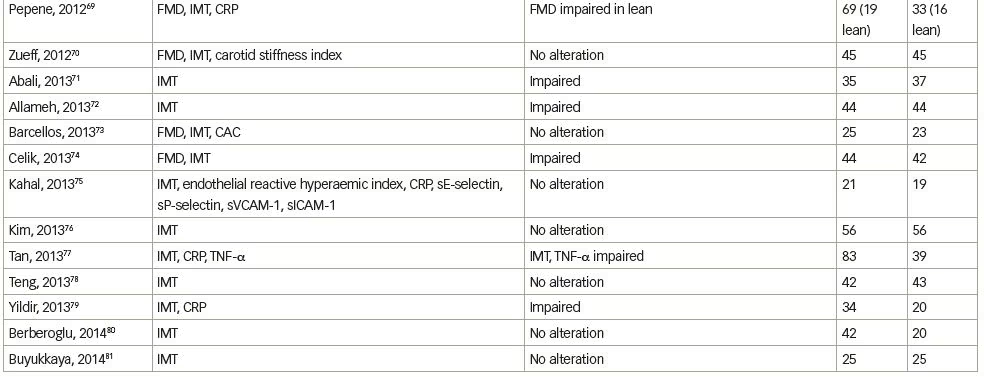
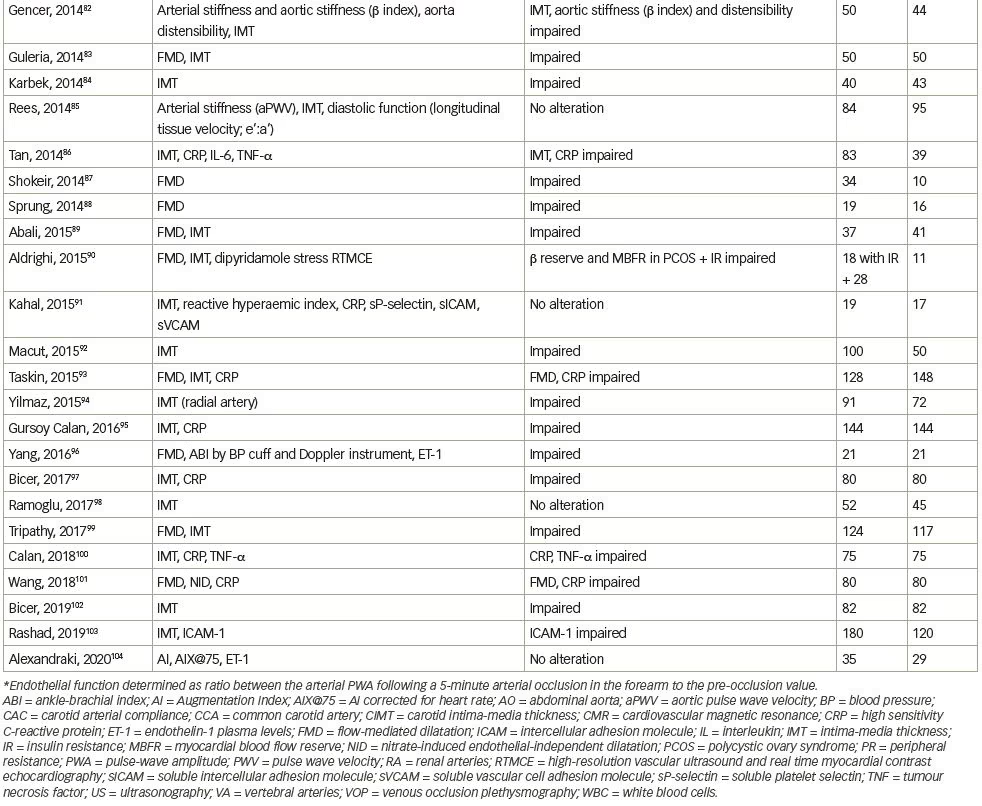
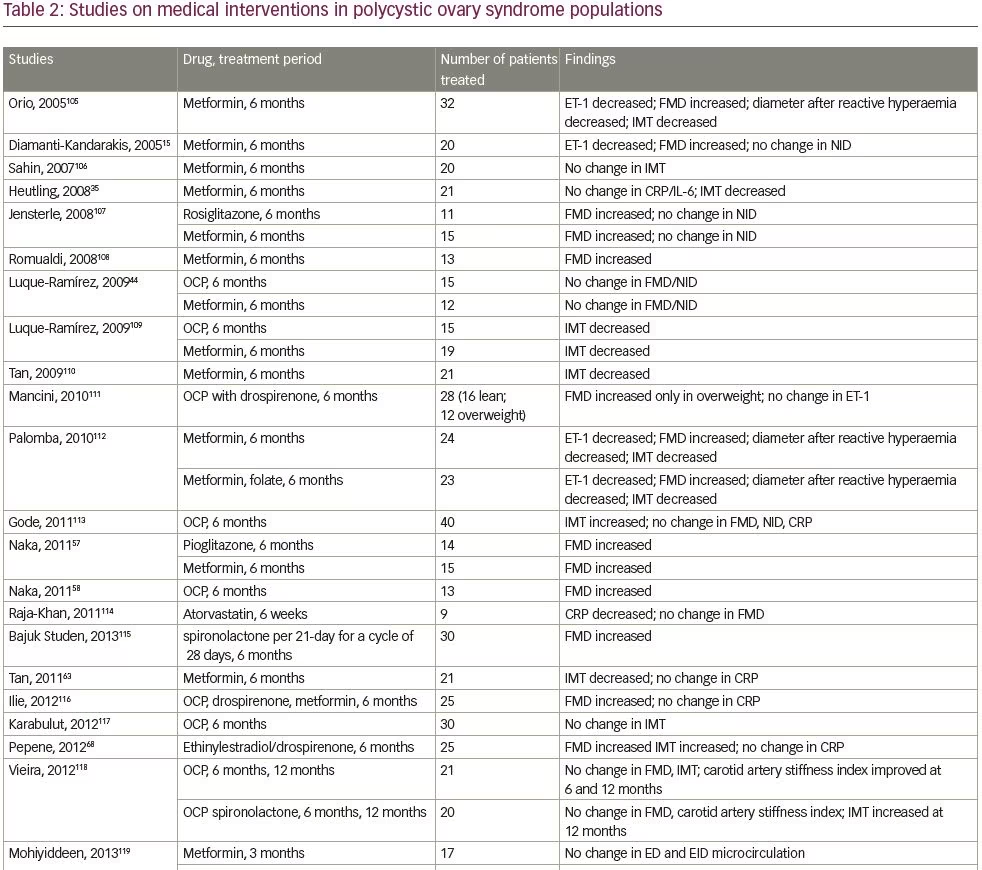
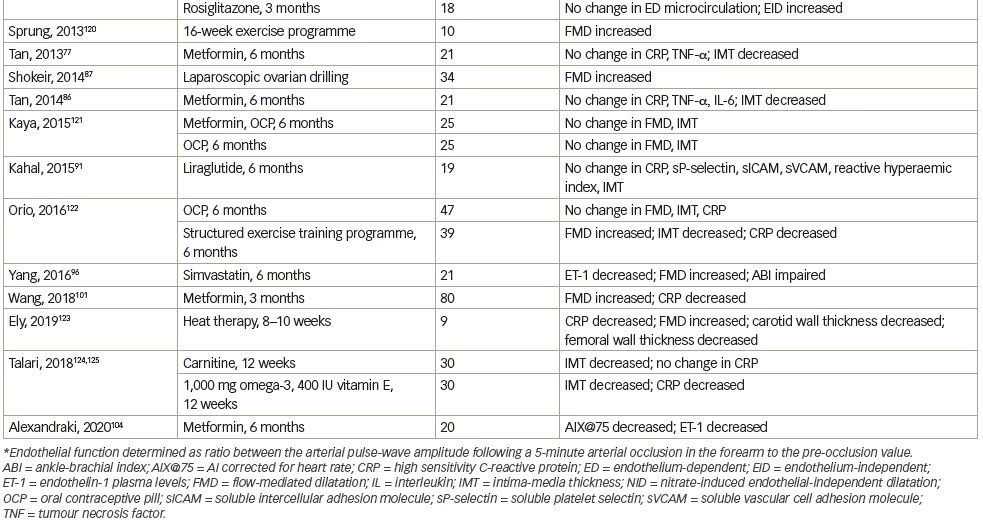
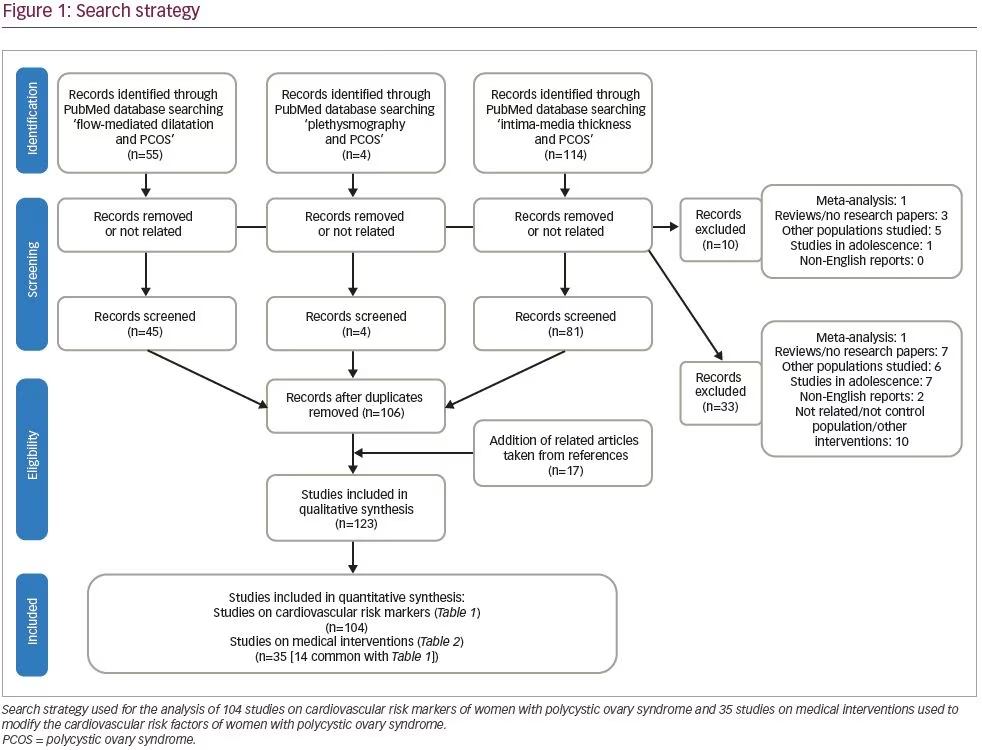
We systematically searched and reviewed all relevant publications in PubMed up to February 2020 using the terms ‘flow-mediated dilatation and PCOS’ (55 results), ‘plethysmography and PCOS’ (4 results), and ‘intima-media thickness and PCOS’ (116 results). Reviews and studies without a control group to be compared with the PCOS group were excluded. Finally, we selected 104 research studies investigating cardiovascular risk factors in PCOS compared to controls, published between 1996 and 2020, and 35 studies (14 common) investigating cardiovascular risk factors in women with PCOS before and after therapeutic interventions, published between 2005 and 2020 (Tables 1 and 2, Figure 1).3–125
Definition of polycystic ovary syndrome
PCOS is a heterogeneous syndrome that has remained a syndrome of unknown aetiology since its first description by Stein and Leventhal 85 years ago, and is probably one of the least understood endocrine disorders in women.126 The clinical phenotype is determined by genetic and environmental factors.127 In 2009, after a systematic review of the published literature, the Androgen Excess Society suggested that PCOS should be defined by the presence of hyperandrogenism (clinical and/or biochemical), ovarian dysfunction (oligo-anovulation and/or polycystic ovaries), and the exclusion of related disorders. Nevertheless, some endocrinologists have also addressed the possibility that there may be forms of PCOS without overt evidence of hyperandrogenism, but more data were required before validating this supposition.128 Therefore, PCOS remains a diagnosis of exclusion, and it is evident that PCOS is a polyprismatic condition. The prevalence of different phenotypes and the actual clinical significance of different laboratory and imaging variables remain under continuous investigation in an effort to identify markers of the syndrome’s severity and predictors of its long-term sequelae.
Metabolic and related features associated with polycystic ovary syndrome
The long-term effects of PCOS are attributed to several metabolic aberrations ensuing the syndrome, including insulin resistance and hyperinsulinaemia, type 2 diabetes mellitus (T2DM), dyslipidaemia and other risk factors for cardiovascular disease (CVD).129–132
Insulin resistance is regarded as the cornerstone of the syndrome’s pathophysiology and seems to have detrimental effects on several target organs, justifying the characterization of PCOS as a multisystem disorder. An overlap between PCOS and insulin resistance or metabolic syndrome has been described, since these two clinical entities share common characteristics such as abdominal obesity, elevated serum triglyceride levels, low serum high-density lipoprotein cholesterol (HDL-C) levels, abnormal blood pressure (BP) and impaired carbohydrate metabolism.133,134 The prevalence of metabolic syndrome in women with PCOS has been estimated to range from 43% to 46%.135,136 More specifically, hyperinsulinaemia and insulin resistance play a critical role in the pathogenesis of PCOS and metabolic syndrome, which are both associated with a high risk for T2DM, hypertension, hyperlipidaemia and atherosclerosis. Insulin resistance is present in approximately 50–75% of women with PCOS130 and seems to be independent from obesity.137–139 In 50% of women with PCOS, insulin resistance appears to be related to abnormal insulin-independent serine phosphorylation of the β-subunit of insulin receptor.130,140 Reduced activity of phosphatidylinositol 3-kinase (PI3K) was also found in muscle tissue of these patients.141
With regard to the criteria used to determine insulin resistance, it is known that the hyperinsulinaemic euglycemic clamp (HEC) remains the gold standard, but due to its complexity and invasiveness it is rarely used in practice compared with other methods, which may be less precise but are non-invasive and widely acceptable in clinical research (Homeostatic Model Assessment of Insulin Resistance [HOMA-IR], Quantitative Insulin Sensitivity Check Index [QUICKI]).142 New markers have also been suggested as indirect criteria for insulin resistance, such as adipocytokines, leptin and ghrelin, but their accuracy is still under consideration.143 In general, insulin resistance, regardless of the diagnostic criteria used, is the hallmark of PCOS as demonstrated by HEC;144 whereas most studies are using control women to define insulin resistance.
Overweight and obesity, defined as a body mass index (BMI) within the range of 25.00–39.99 kg/m2 (mild 25.00–29.99, and severe >30.00–39.99 kg/m2), have been shown to be more prevalent in women with PCOS and are further associated with hyperinsulinaemia and insulin resistance.145 The prevalence of obesity in patients with PCOS differs depending on ethnicity, being less prevalent in Europe146 and more common in the USA.147 However, both lean and obese women with PCOS display central fat distribution.148–150 Additionally, obese premenopausal women with PCOS have a 31–35% incidence of impaired glucose tolerance, whereas 7.5–10.0% develop overt diabetes.151,152 Moreover, other clinical entities, such as non-alcoholic steatohepatitis and non-alcoholic fatty liver disease,153 as well as obstructive sleep apnoea and excessive daytime sleepiness, have been associated with insulin resistance and PCOS (Figure 2).154
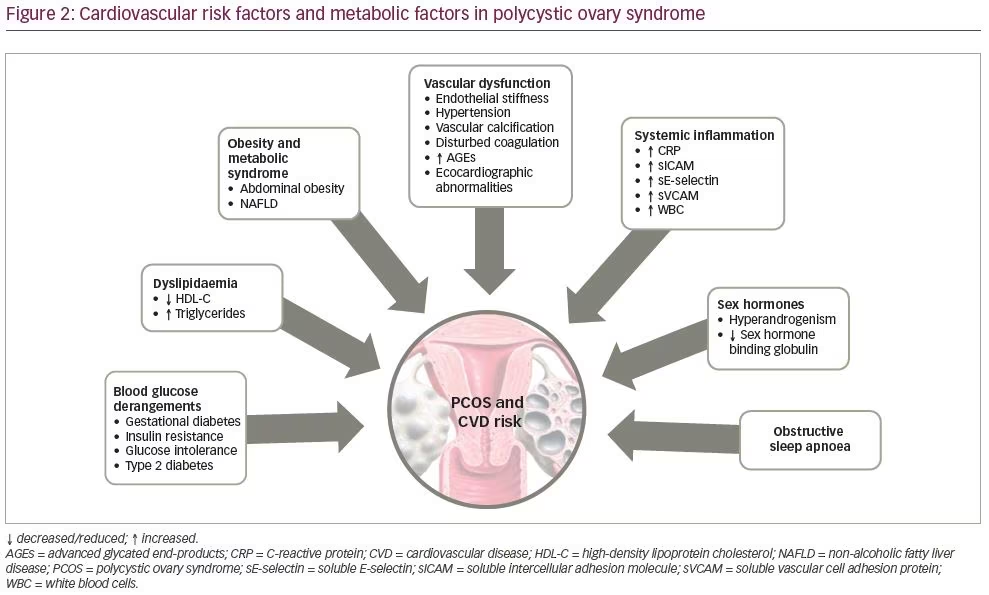
Parameters of cardiovascular disease
PCOS has been reported to bear an increased risk for atherosclerosis with a calculated increased risk (relative risk) of 7.4 for myocardial infarction.155 An early sign of the atherogenic process, which results in overt CVD, is endothelial dysfunction,156 which has been comprehensively studied in PCOS. Since PCOS has been suggested to represent a female subtype of metabolic syndrome,134 carrying a potential pre-atherogenic load, patients with PCOS have increased cardiovascular risk compared with age-matched controls.157 Myocardial infarction was recorded to be seven times more likely in patients with histopathological evidence of PCO,155 and cardiac catheterization studies have shown more extensive coronary artery disease in these patients than in women with normal ovaries.158 Furthermore, significant subclinical carotid atherosclerosis has been demonstrated on carotid artery ultrasound in women with PCOS.3,5
This increased cardiovascular risk is likely to partly be a result of the metabolic disturbances associated with PCOS. Dyslipidaemia, diabetes (from impaired glucose tolerance to overt T2DM), and obesity are potentially cardiovascular risk factors that tend to cluster in women with PCOS.148,159 Many previous studies have documented abnormalities of insulin metabolism in both lean and overweight women with PCOS.130,138,160,161 Dyslipidaemia, insulin resistance and abdominal obesity are variable clinical features of PCOS which, irrespective of the presence
of PCOS, contribute to the presence of metabolic syndrome with characteristic linkage to atheroma development and to overt risk for CVD.162 In addition, hypertension represents an uncommon and inconsistent finding in young women with PCOS, but its prevalence increases to 40% in the perimenopausal period.163–166 However, the epidemiological evidence of CVD events in relation to PCOS has not been confirmed.167
An alternative explanation for the increased cardiovascular risk in PCOS is hyperandrogenism. Possible underlying pathophysiological mechanisms include a correlation between free testosterone and systolic BP,168 and a link between increased androgens and abnormal lipid metabolism.169,170 Nonetheless, the association between hyperandrogenism and cardiovascular risk is not universally accepted.170 Previous studies of obese women with PCOS have shown that hyperinsulinaemia directly reduces sex hormone binding globulin (SHBG).171 This has led to the hypothesis that SHBG might be a surrogate marker of insulin resistance in this population.172 Other studies, however, in both PCOS and non-PCOS populations suggest that BMI and percentage body fat, rather than insulin, are independent predictors of SHBG.173,174
Endothelial function and polycystic ovary syndrome
The endothelium is the target organ for a variety of metabolic risk factors. It has been demonstrated that insulin exerts a direct hypertrophic effect on the vascular endothelium and on the smooth muscle cells.175 Abnormal vascular function has been shown in PCOS, and the syndrome has been associated with surrogate cardiovascular markers, such as increased serum levels of plasminogen-activator inhibitor 1 (PAI-1),176 elevated serum levels of C-reactive protein (CRP),177 elevated advanced glycation end-products (AGEs) serum levels178 and echocardiographic abnormalities.179–181 The pathophysiology remains unclear but a number of mechanisms could be implicated to link endothelial dysfunction with insulin resistance, including disturbances of subcellular signalling pathways to insulin action or other potential unifying links.182–184
Haemodynamic methods for endothelial function assessment in polycystic ovary syndrome
The endothelium plays a crucial role in regulating arterial function. Endothelial dysfunction is one of the best-understood preliminary steps in the development of atheromatous disease.185 It has been considered not only as a marker of early, possibly reversible, arterial abnormality, but also as a prognostic marker for future cardiovascular morbidity.186 Endothelial dysfunction can be studied by haemodynamic methods, like brachial flow-mediated dilatation (FMD) and venous obstructive plethysmography (VOP) on peripheral arteries, which both examine different sections of arterial bed (macro- and microcirculation, respectively) and should be considered complementary, providing different information.21
The association between insulin resistance and endothelial dysfunction has been demonstrated consistently in subjects with T2DM, obesity and metabolic syndrome.187 Thus, at a mechanistic level, endothelial dysfunction appears to occur early in the insulin-resistant state; the progression of insulin resistance to diabetes in parallel to the progression of endothelial dysfunction to atherosclerosis.187 There are several mechanisms through which insulin resistance can adversely affect the endothelium. Insulin-resistant states, such as T2DM, are characterized by increased production of free fatty acids and proinflammatory cytokines, such as tumour necrosis factor α (TNF-α) and leptin, which contribute to endothelial dysfunction.188 There is also evidence to suggest that insulin resistance induces increased oxidant stress, which may have an important pathogenic role.175
Endothelial dysfunction and flow-mediated dilatation in polycystic ovary syndrome
Endothelial function can be estimated in conduit arteries (macrovascular function), non-invasively by high-resolution ultrasonography on the brachial artery, assessing FMD,189 an endothelial-dependent dilatation, which has been found to be correlated with coronary endothelial function,190 consequently representing an independent predictor of cardiovascular events.191 In the first published study to investigate endothelial function by FMD in patients with PCOS, no difference was shown compared to control women.4 However, few studies have confirmed these negative findings (Table 1).3–104
On the contrary, most studies undertaken in young women with PCOS, of normal weight or overweight and obese, confirm overt endothelial dysfunction. First, in young, lean women with PCOS, a 20% drop of FMD response has been shown, compared with normal women of a similar age and BMI.10 Furthermore, the combination of FMD and the biochemical assessment by endothelin 1 (ET-1) plasma levels to assess endothelial dysfunction in young, overweight insulin resistant women with PCOS and controls, with similar age and BMI, documented impairment in women with PCOS, after exclusion of smooth muscle cells injury detected by nitrate-induced endothelial-independent dilatation (NID).15 Other parallel studies confirmed FMD impairment along with NID impairment, implying a global vascular deficit.12,16 In all these studies, insulin resistance had an impact on FMD,12,15,16 whilst a tendency of FMD to deteriorate from lean to overweight and to obese women with PCOS was observed, even without reaching statistically significant differences.16 Notably, in lean women with PCOS, FMD was found to be negatively influenced only by hyperandrogenaemia, and in overweight women by insulin resistance and hyperandrogenaemia.16 In an effort to correct for other cardiovascular risk factors, FMD and NID were assessed in a normoglycaemic, eulipidaemic, normal BP and normal glucose tolerance PCOS group, and a control group with comparable age, BMI and waist:hip ratio. FMD, but not NID, was impaired in women with PCOS who remained insulin-resistant.21 Finally, in young overweight women with PCOS, FMD was impaired compared with ovulatory controls matched for age and BMI, and this alteration was correlated to insulin resistance but not to the adipokines measured.23 Endothelial function has also been assessed by the use of novel technologies using cardiovascular magnetic resonance imaging, confirming pronounced endothelial dysfunction with normal endothelial independent dilatation in PCOS, compared with controls with similar age and BMI.27
We reviewed 39 studies that assessed FMD, and 27 (69%) of them, which included 1,359 patients with PCOS and 1,073 control subjects, showed impaired FMD in PCOS as opposed to 12 studies (including 448 patients with PCOS and 307 control subjects) that did not show any difference between these groups (Table 1).3–104 On the contrary, endothelial independent dilatation has been found to be mostly normal in women with PCOS (seven studies with 232 patients with PCOS and 211 control subjects versus three studies with 129 patients with PCOS and 72 controls).
Venous occlusion plethysmography and microvascular function in polycystic ovary syndrome
Microvascular function can be estimated in resistance arteries, and has been assessed in women with PCOS. In a HEC study in obese women with PCOS, a 50% reduced endothelial-dependent and
insulin-mediated response of leg blood flow was found in the PCOS group, along with resistance to the vasodilating action of insulin, compared with the control group.7 This reduction was in response to intrafemoral injection of methacholine chloride. Insulin resistance was suggested to contribute to endothelial dysfunction, whereas BMI and androgens predicted endothelial dysfunction.7
Microvascular function has also been assessed, non-invasively, on resistance arteries by VOP on the forearm in women with PCOS who were normoglycaemic, eulipidaemic, and normotensive, compared with controls of comparable age, BMI and waist:hip ratio.21 The only parameter that differed between groups was time to peak hyperaemia, a parameter considered to reflect endothelial dysfunction, which was found protracted in the more insulin-resistant women with PCOS.21 Interestingly, time-to-peak hyperaemia was negatively related to SHBG, indirect index of hyperinsulinaemia and hyperandrogenaemia. Another study, using the same methodology, confirmed a shorter duration of reactive hyperaemia in the PCOS group, and a significant positive correlation between the duration of reactive hyperaemia and dehydroepiandrosterone-sulfate (DHEAS) levels in the PCOS group was reported, a finding that may support the cardioprotective effect of this androgen.25
Young women with PCOS have shown impaired endothelial-dependent dilatation as assessed by blood flow in the microvasculature of the forearm by the water displacement method during insulin infusion into the forearm in insulin-resistant PCOS, whereas insulin-sensitive women with PCOS responded similarly to controls.14 On the other hand, vascular function estimated by VOP assessing forearm vasodilatation in response to both endothelium-dependent (acetylcholine/bradykinin) and endothelium-independent (nitroprusside/verapamil) dilatators was not impaired in women with PCOS, compared with age- and weight-matched controls.29 Overall, forearm vasodilatation to all four drugs was reduced (<50%) in obese women with PCOS, compared with lean women with PCOS, but no significant difference in vascular function was detected between women with PCOS, compared with corresponding controls. On the other hand, obese women with PCOS exhibited markedly reduced vascular smooth muscle function compared with lean subjects with PCOS.29 Similarly, using a VOP technique to assess reactive hyperaemia of forearm microcirculation showed no evidence of endothelial dysfunction in obese women with PCOS compared with age- and weight-matched controls with a similar metabolic and inflammatory profile.13
Microvascular function has also been studied by wire myography, measuring the concentration response curve to norepinephrine before and after incubation with insulin in young women with PCOS and controls with similar cardiovascular risk factors.8 A functional defect in the vascular action of insulin ex vivo in patients with PCOS was demonstrated, suggesting a deleterious effect of insulin resistance at a vascular level in women without overt CVD. The defect in resistance arterioles was suggested to be specific to the action of insulin, since constriction to norepinephrine and relaxation to acetylcholine did not differ between women with PCOS and controls. In healthy subjects, insulin caused a dose-related attenuation of norepinephrine action; maximal response and sensitivity to norepinephrine decreased after exposure to physiological and supraphysiological concentrations of insulin. However, no response was observed at physiological doses in the vessels of women with PCOS, whereas at supraphysiological doses, sensitivity, but not maximum response, was altered, suggesting an abnormal resistance artery response to insulin, even in the absence of any other risk factor.8
In another study, microvascular function was assessed by forearm skin microvascular erythrocyte flux responses to cumulative iontophoretic doses of 1% (w/v) acetylcholine and 1% (w/v) sodium nitroprusside, using laser Doppler imaging in women with PCOS and age-matched controls.17 Basal microvascular perfusion was comparable in PCOS and controls, but the increase in skin microvascular perfusion in response to acetylcholine was blunted and peak acetylcholine-induced erythrocyte flux was reduced in women with PCOS compared with controls. Twenty per cent of acetylcholine response was attributed on insulin and BMI, while altered acetylcholine response was influenced by BMI and androgens levels. No difference in the response to nitrates was documented, implying an exclusively endothelial microvascular dysfunction.
Summarizing, based on different approaches of microcirculation assessment, it could be concluded that women with PCOS have impaired function in micro-vessels, but there are limited data regarding the association of this alteration with the hormonal and metabolic disturbances of the syndrome. On the other hand, among the four studies that have used plethysmography to assess microvasculature,7,13,21,25 only one (11 patients with PCOS versus 12 controls) did not show impairment in women with PCOS,13 whilst the other three studies (66 patients with PCOS versus 53 controls) showed a clear difference7,13,21,25 (Table 1).3–104 In another two studies performed by the same group where reactive hyperaemia was assessed by an alternative technique, no difference was documented.75,91
Another important manifestation to consider as a result of microvascular endothelial dysfunction would be pre-eclampsia, particularly the late-onset, where endothelial involvement has a key role in the pathogenesis.192 In fact, a recent prospective cohort study showed that pre-eclampsia, gestational diabetes and lower birth weight among newborns were significantly higher in the women with PCOS compared with healthy controls, identifying higher BMI and hyperandrogenaemia as the strongest predicting factors.193
Intima-media thickness and polycystic ovary syndrome
Intima-media thickness (IMT) is a morphological marker of vascular injury.194 An increase in carotid artery IMT has been observed in women with PCOS, and especially in middle-aged women.3,5 In 16 premenopausal women, aged >40 years old with a history of PCOS, carotid IMT was increased, compared with a control group of similar age.3 Nevertheless, this difference was observed between patients and controls, up to the age of 45 and not in younger groups. PCOS was found to represent the unique prognostic factor of variable IMT in multiple regression analyses, while, after correction for age and BMI, the correlations of PCOS changed slightly, implying that part of the observed relation between PCOS and IMT is likely due to central obesity and hyperinsulinaemia.3 In a study of 125 women with PCOS and 142 controls, there was a significantly higher prevalence of carotid index of atheromatic plaques in women with PCOS (7.2% versus 0.7%) compared with controls.5 The difference in carotid IMT between women with PCOS and controls was detected in women aged ≥45 years, compared with women aged 30–44 years. Since CVD is characterized by long-term subclinical latency, metabolic disturbances are becoming overt at middle-age only. PCOS and aging appear to interact, resulting in deleterious changes to the carotid arterial wall, since differences in lipids levels between women with PCOS and controls are blunted approaching the menopausal period.194,195 As expected, contradictory data have been published in younger women.9,18,23
Notably, a clinical study investigated the haemodynamic changes in the medial cerebral artery and the internal carotid artery in 28 young women with PCOS and polycystic ovaries (PCO) compared to 24 healthy control subjects.196 Blood flow rate and pulsatility index were measured by transcranial Doppler ultrasonography, but no significant differences in haemodynamic parameters were detected between groups.196 In another study, impaired carotid IMT in young women of normal weight who were eulipidaemic, normotensive and had PCOS, suggested a premature structural vascular injury.10 In a large-population study, 75 women with PCOS were compared with a group of 55 healthy women of similar age, BMI, smoking habits, familial history for CVD, blood pressure and glucose, and higher IMT values were evidenced in the PCOS group.19 In PCOS, IMT was positively correlated with age and BMI and negatively with low HDL-C, DHEAS and androstenedione plasma levels, indicating that adrenal androgens and particularly DHEAS can partially compensate the unfavourable consequence of dyslipidaemia and insulin resistance. This outcome has been supported in other published work, which has focused on the effect on adrenal sex steroids on the metabolic profile of women with PCOS, but did not satisfy the criteria to be included in this review.197,198 In fact, in a recent study a positive correlation of 17-hydroxyprogesterone (17-OH-Pg) with HDL-C and HOMA-S was found, which suggests that patients with hyperandrogenaemic PCOS with high levels of 17-OH-Pg could have a certain protection against CVD.199 Finally, in 43 young women with PCOS and 43 age-matched controls, common carotid IMT was higher in women with PCOS compared with controls.20
Overall, we reviewed 71 studies that assessed IMT; 44 (62%) of them, including 2,761 patients with PCOS and 2,218 control subjects, showed impairment in PCOS, as opposed to 27 studies (including 1,571 patients with PCOS and 1,286 subjects) that did not show any difference between the groups (Table 1);3–104 however, the mixture of population characteristics may have had a major impact on statistics.
Other cardiovascular risk factors
In a study evaluating serum aldosterone concentration and cardiovascular risk in women with PCOS, a direct correlation between plasma aldosterone, IMT and mean BP was found.
Insulin resistance in 50 women with PCOS was positively related to serum aldosterone levels, compared with 50 age- and BMI-matched healthy controls.24 The activation of the renin-angiotensin-aldosterone system has an impact on endothelial dysfunction and is involved in the process of atherogenesis in women with PCOS, so spironolactone treatment might reverse endothelial dysfunction in women with PCOS.200
Cardiac function and polycystic ovary syndrome
Other morphological and functional cardiovascular parameters have also been studied in PCOS. An increased prevalence of coronary artery calcium (a marker of coronary atherosclerosis) was found when assessed by non-invasive electron beam computed tomography (EBCT) in 36 women with PCOS, compared with 71 controls of a similar age and BMI.201 Similarly, a higher prevalence of coronary artery calcium (45.9% versus 30.6%) and aortic arterial calcification (68.9% versus 55.3%), measured by EBCT, was documented in women with PCOS compared with controls in a prospective study, whereby women were screened in 1993–1994 and re-evaluated in 2001–2002.11 After adjustment for age and BMI, PCOS was found to be a significant predictor of coronary artery calcium, but not aortic arterial calcification. In addition, an increased risk of metabolic syndrome was reported in women with PCOS.11
In 60 women (20 with PCOS, 20 with PCO and 20 healthy controls), compliance and stiffness indices were assessed in the common and internal carotid arteries using ultrasonographic methods. An impairment was reported in women with PCOS, compared with controls.202 Furthermore, macrovascular function at the brachial artery (but not at the aorta), as well as microvascular function, have been found to be impaired, suggesting increased vascular stiffness and a functional defect in the vascular action of insulin ex vivo in patients with PCOS.17 This has been confirmed by other studies evaluating arterial stiffness, where insulin resistance was an independent prognostic factor of pulse wave velocity and arterial stiffness in women with PCOS.18 Endothelial function has also been assessed using peripheral arterial tonometry methodology, namely a post-ischaemia reactive hyperaemia technique that determines endothelial function as the ratio between the arterial pulse wave amplitude following a 5-minute arterial occlusion in the forearm to the pre-occlusion value, in normal-weight young women with PCOS compared with a control group with similar BMI.30 However, taken together, these parameters (arterial stiffness, augmentation index, resistance indices, aortic wave reflections) showed contradictory results (Table 1).3–104
Cardiac function parameters have been also investigated in PCOS. In 26 lean young women with PCOS (mean age 22.8 ± 0.9 years; mean BMI 23.0 ± 0.8) and 11 healthy slightly older women with similar BMI (mean age 26.3 ± 1.7 years; mean BMI 22.9 ± 0.9), parameters of cardiac output were estimated by ultrasonographic method; impaired parameters of systolic function, as well as their negative association with fasting insulin, were observed in the PCOS group without impairment of diastolic function.179 In another study, several diastolic and systolic parameters were impaired in women with PCOS; however, women with PCOS had higher total cholesterol and LDL-C levels compared with controls.180 In another study, 30 women with PCOS differed in diastolic cardiac parameters compared with 30 older healthy women, and hyperinsulinaemia and lipidaemic load were higher in the PCOS group.181 Another study found that ultrasonographic cardiac parameters were evaluated and women with PCOS had higher left atrium size and left ventricular mass index, lower left ventricular ejection fraction and early-to-late mitral flow velocity ratio than controls.203 When patients and controls were grouped according to weight into normal weight, overweight and obese groups, differences in the metabolic profile and cardiac findings persisted, but a progressive impairment of metabolic profile and echocardiographic pattern was seen in patients with normal weight compared with those with obesity. Young women with PCOS had a significantly increased cardiac size compared with controls, and other structural, systolic and diastolic function cardiac parameters were altered. Notably, most cardiac abnormalities persisted even in young patients with normal weight, suggesting that the pathogenesis of cardiac abnormalities in PCOS is not only dependent on BMI, but also to insulin resistance indices.203
Finally, autonomic innervation of the heart, evaluated non-invasively with power spectrum analysis of heart rate variability from electrocardiographic recordings, can be affected in women with PCOS with increased sympathetic and decreased parasympathetic components of heart rate variability.204
Biochemical studies for endothelial function assessment in polycystic ovary syndrome
Endothelial dysfunction can also be assessed biochemically by ET-1 plasma levels measurements. In 2001, Diamanti-Kandarakis et al. showed that ET-1 plasma levels are increased in women with PCOS.6 ET-1 plasma levels were positively related to androgen levels and negatively to insulin sensitivity, assessed by HEC (the gold standard assessment of insulin sensitivity), suggesting insulin resistance and hyperinsulinaemia implication in altered endothelial function. These results were later confirmed.10,15 Overall, we reviewed seven studies that assessed ET-1 levels, five (71%) of which included 109 patients with PCOS and 83 controls, and showed increased ET-1 levels in PCOS, whereas two studies (including 59 women with PCOS and 49 controls) did not show any difference between the groups (Table 1).3–104
Markers of low-grade inflammation and certain components of the haemostatic system have been also shown to predict atherosclerotic risk in insulin-resistance states such as metabolic syndrome and T2DM.205,206 Recent advances in basic science have demonstrated a fundamental role for inflammation in mediating all stages of this disease, from initiation through progression of atherosclerosis. The current notion that inflammation and immune response contribute to atherogenesis has garnered increased interest.207 Evidence of low-grade chronic inflammation in PCOS is indicated by the presence of several elevated markers such as CRP levels,12,177,208,209 inflammatory cytokines,210–212 increased leukocyte count213 and adhesion molecules177 (Table 1).3–104
A number of studies in humans have assessed the relationship between endothelial function and markers of inflammation, but the results have been variable and sometimes conflicting regarding the relationship between CRP and brachial artery FMD, or between CRP and coronary endothelial dysfunction. In sum, we reviewed 34 studies that assessed CRP levels; 21 (62%) of them (including 1,589 patients with PCOS and 1,209 control subjects) showed increased CRP levels in PCOS, as opposed to 13 studies (including 591 PCOS and 473 control subjects) that did not show any difference between the groups (Table 1).3–104
Management of endothelial function impairment in polycystic ovary syndrome: Treatment with insulin sensitizers
A number of mechanisms have been implicated to link endothelial dysfunction with insulin resistance, including disturbances of subcellular signalling pathways to insulin action or other potential unifying links.182,183,214 Therefore, utility of insulin sensitizers can be justified in patients with PCOS, and specifically for endothelial function improvement, and we will discuss their function along with other therapeutic agents that have been used for PCOS management (Figure 3).
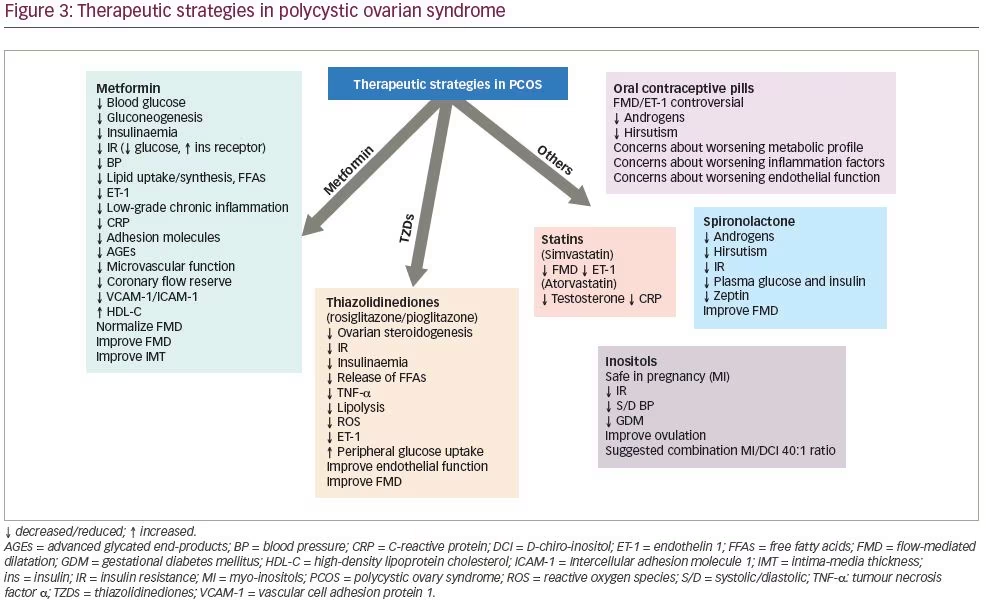
Metformin is a biguanide agent that has been used in the treatment of T2DM. It lowers blood glucose levels by significantly reducing hepatic glucose production and by increasing peripheral glucose utilization. It lowers serum insulin levels and improves insulin sensitivity, not only by its glucose-lowering effect, but also by increasing insulin-binding to its receptor.215 Moreover, the drug appears to enter into the cells and enhance the tyrosine phosphorylation of the intracellular portion of β-subunit of the insulin receptor and of insulin receptor substrate (IRS) proteins.216 Recently, it has been suggested that IRS genotype may modulate the response to metformin treatment in women with PCOS.217
A number of studies have been published investigating the effect of metformin on insulin resistance in women with PCOS. One study showed that metformin treatment in 26 obese women with PCOS for 2 months reduced hyperinsulinaemia and systolic BP, and improved hormonal and reproductive abnormalities.218 The first European study, published in 1998, confirmed the increase in insulin sensitivity (glucose utilization rate) using the HEC, irrespective of the changes in body weight, after administration of 1,700 mg metformin for 6 months in 13 obese women with PCOS.219 Metformin has been shown to improve lipid profile, mainly by increasing serum HDL-C concentrations.220,221 However, some studies have shown only a negligible or no effect on lipids in women with PCOS.218,222 The mechanisms by which metformin improves lipid profile are not clear; it has been suggested to reduce lipid uptake or synthesis in the intestine and in the hepatocytes.220,223 The improvement of obesity, especially abdominal obesity, with a subsequent decreased release of free fatty acids from adipose tissue observed during metformin therapy could also partly explain the improvement of lipid profile during metformin treatment, at least in obese women.224
A study investigating the effect of metformin on endothelial function in obese and non-obese women with PCOS and increased plasma ET-1 levels, revealed a statistically significant reduction of those levels in both groups after 6 months of treatment.6 Interestingly, hyperandrogenaemia, as well as insulin resistance (assessed by HEC), were improved in parallel with ET-1 levels post-metformin without a concomitant reduction in BMI. Later, the administration of 1,700 mg metformin daily for 6 months in young overweight women with PCOS normalized FMD on the brachial artery, and at the same time significantly decreased ET-1 plasma levels.15 On the other hand, when metformin was used for 3 months, no improvement was observed in endothelial function assessed using peripheral arterial tonometry methodology in normal-weight young women with PCOS.30 The exact mechanism, direct or indirect, by which metformin acts on endothelium, has not been clarified. Nevertheless, metformin treatment has been associated with a significant decrease of low-grade chronic inflammatory markers.177
In a randomized study, a significant reduction in serum CRP levels by 31% was shown in non-obese patients and 56% in obese patients, post-metformin administration.225 Adhesion molecules have also been reduced after 6 months of treatment with metformin in 62 patients with PCOS, independently of body weight changes.177 In addition, AGEs, a complex and heterogeneous potent atherogenic group of molecules,226 and their receptors, were found to be elevated in young women with PCOS.178 Although the mechanism implicated in this phenomenon is not clear, it could be due to insulin action defects; dietary-exogenous AGEs may also contribute to it.227 Metformin administration for 6 months reduced, but did not normalize, AGEs serum levels compared with
pre-treatment levels; this effect of metformin, if confirmed, could be of clinical significance in improving adverse long-term sequelae of the syndrome.228 Metformin was also administered in women with PCOS with and without insulin resistance, and improved microvascular function and coronary flow reserve after 6 months, as assessed by transthoracic second-harmonic Doppler echocardiographic methodology.229
We reviewed metformin administration in 19 study groups, in a total number of 465 women with PCOS for a median period of 6 months’ time (range 3–6 months). Oral contraceptive pills were co-administered in two studies. FMD improved (increased) in 8 out of 10 (80%) studies, ET-1 improved (decreased) in all the four studies in which this was assessed, IMT improved (decreased) in 8 out of 10 (80%) studies, and amelioration in reactive hyperaemia in two out of the three studies in which this was assessed. These findings indicate the beneficial effect that metformin exerts on micro-, macro-circulation as well as on the structural arterial properties of young women with PCOS (Table 2).15,35,44,57,58,63,68,77,86,87,91,96,101,104–125 In contrast, CRP levels were not reduced in five out of six (83%) studies,35,63,77,86,116 not confirming the documented anti-inflammatory action of metformin on this low-grade inflammation present in this young population.35,63, 77,86,101,117
Thiazolidinediones
Thiazolidinediones include three drugs: troglitazone, rosiglitazone and pioglitazone. They act by enhancing glucose uptake in adipose and muscle tissues. In comparison to the effect observed by metformin, thiazolidinediones increase more peripheral glucose uptake and decrease lesser hepatic glucose output. They improve insulin sensitivity and they decrease not only circulating insulin but also the release of free fatty acids and TNF-α from adipose tissue. At the cellular level they bind and activate the peroxisome proliferator activated receptor-γ (PPAR-γ) and they induce the transcription of genes that are involved in glucose control and lipid metabolism.230
In women with PCOS, the therapeutic effect of insulin sensitizers is mediated by the reduction of hyperinsulinaemia and insulin resistance, and consequently by alteration of ovarian steroidogenesis. Moreover, an inhibitory effect in steroidogenesis has been demonstrated on humanized yeast cells, and thiazolidinediones (rosiglitazone and pioglitazone) have been found to inhibit estradiol and testosterone production in cultured human ovarian cells.231 Dunaif was the first to study the effects of troglitazone on metabolic profile in women with PCOS in a double-blind, randomized, 3-month trial of two doses (200 mg, 400 mg) of troglitazone.232 Hyperinsulinaemia, as well as insulin sensitivity, was assessed by a frequently sampled intravenous glucose tolerance test and improved significantly.
In a later study, troglitazone was administrated for 3 months in 13 obese women with PCOS and found to have impaired endothelial function, as assessed by leg blood flow responses to graded intrafemoral artery infusion of the endothelium-dependent vasodilator methacholine chloride. Basal leg blood flow was found unchanged after troglitazone, but the increase in leg blood flow in response to methacholine chloride in PCOS was markedly more pronounced after treatment and was similar to that observed in obese women without PCOS.233 It was suggested that troglitazone improves endothelial function by reducing lipolysis, reactive oxygen species production,234 ET-1 secretion235 or vascular inflammation.236 Confirming the above results, as troglitazone was retracted from clinical use, rosiglitazone was administered in 31 young normal to overweight women with PCOS for 1 year and it was observed to improve endothelial function, and at the same time, insulin sensitivity indices, hyperandrogenaemia, hyperinsulinaemia and systemic inflammation, when assessed by CRP.237
Overall, in two of the included studies, where rosiglitazone was given for a median time of 4.5 months (range 3–6 months), FMD was improved, but not NID, in one study,107 and in the second study using a different method to assess microcirculation, no improvement was noted in endothelial-dependent function as opposed to a significant improvement in endothelial-independent function.119 On the contrary, in the study of 14 women with PCOS with pioglitazone administration, FMD was improved.57
Oral contraceptive pills represent a traditional treatment for the hyperandrogenic sequelae of women with PCOS. We reviewed 13 study groups of women with PCOS receiving oral contraceptives, who were compared, or not, with other treatments. Metformin was co-administered in two of the studies included. FMD improved in three (27%) out of 11 study-groups; ET-1 levels did not improve in one study where this was assessed; whilst IMT improved in three (33%), but deteriorated in one out of nine study groups (Table 2).15,35,44,57,58,63,68,77,86,87,91,96,101,104–125 Similarly, CRP levels were not reduced in all four study groups that assessed it. A meta-analysis including three randomized controlled studies that compared oral contraceptives containing oestrogen (ethinylestradiol) with progestogen drospirenone or chlormadinone acetate, showed a beneficial effect of ethinylestradiol/drospirenone over ethinylestradiol/chlormadinone acetate in reducing androstenedione levels and total testosterone after 3 months, and Ferriman–Gallwey score after
6 months.238
Although oral contraceptive pills improve clinical and biochemical hyperandrogenism, there are concerns about worsening the metabolic and inflammatory profile of these patients. Adipokines, such as adiponectin, visfatin and resistin, which are considered indices of both inflammation and insulin resistance, have deteriorated in women with PCOS after 6 months of oral contraceptive treatment compared with controls.239 Additionally, the use of oral contraceptives in obese andnon-obese patients with PCOS with impaired glucose tolerance increases asymmetric dimethylarginin, which is a marker of endothelial dysfunction and CRP levels, so creates an increase in metabolic risk.240 On the other hand, the combination of oral contraceptive pills with metformin in overweight women with PCOS may lower this risk, as it seems to improve both hyperandrogenism and inflammatory parameters.241 Similar results have been shown when metformin has been combined with anti-androgens containing oral contraceptives, which may improve androgen and insulin resistance. However, this addition of metformin has achieved a reduction in adhesion molecules as markers of low-grade chronic endothelial inflammation and CVD.242
Statins and glucagon-like peptide-1
Statins have been also investigated in PCOS. Atorvastatin failed to improve FMD but reduced CRP in nine women with PCOS in only 6 weeks,114 whereas simvastatin given for 6 months improved FMD and hormonal profile, and reduced ET-1 levels.96,243 Liraglutide has also been trialled for a 6-month period, but no overt benefits were seen in reactive hyperaemic index, IMT, CRP and adhesion molecules.91 Although, the reproductive and hyperandrogenaemic profile of these patients was not examined in these studies, a very recent meta-analysis of nine randomized controlled trials (613 patients) showed a superiority of atorvastatin compared with all other treatments (simvastatin, spironolactone, oral contraceptives and metformin) to reduce total testosterone levels in patients with PCOS.244
Spironolactone, due to its anti-androgenic and aldosterone antagonistic effects, has been explored among PCOS treatment strategies. It has been shown to be effective in improving both reproductive and metabolic parameters, equally to pioglitazone and superiorly to metformin, especially when combined with high doses of vitamin D supplementation.245 Thirty non-obese patients with PCOS with normal mean aldosterone plasma levels had significantly lower FMD compared with 20 BMI-matched control subjects, but this difference was reversed after 6 months’ therapy with spironolactone.115
Inositols are polyols with insulin-sensitizing properties. Myo-inositol is the most common naturally occurring stereoisomer, which can be converted to a second stereoisomer, D-chiro-inositol (DCI). Inositols are involved in insulin signalling, with both myo-inositol and DCI functioning as insulin second messengers, although they mediate different actions of insulin in humans. Specifically, myo-inositol is considered safe in pregnancy and beneficial in reducing insulin resistance, systolic/diastolic BP and the risk of gestational diabetes, which have a higher incidence among women with PCOS.246 The clinical experience with DCI is inferior to myo-inositol, but more interesting data are being published regarding the combination of the two. A recent meta-analysis evaluated the efficacy of treatments with myo-inositol, alone or combined with DCI (40:1 ratio between myo-inositol and DCI) for 12–24 weeks, in nine randomized controlled trials comprising 247 cases and 249 controls. Significant improvement in metabolic parameters (insulin levels, HOMA-IR), and to a lesser extent testosterone levels, was seen after inositol supplementation.247 Moreover, the combination of myo-inositol and DCI in a 40:1 ratio seems to improve the reproductive profile of these patients by restoring ovulation in 62.5%.248 The synergistic effect of metformin with myo-inositol, as opposed to metformin alone, was also investigated in women with PCOS undergoing ovulation induction, and showed not only an increase in live birth rate, but also an improvement in insulin sensitivity and reduced requirements for metformin dosage.249
Taken altogether, controversial data exist regarding the presence of endothelial dysfunction and arterial structure impairment in women with PCOS, with certain studies reporting no impairment and others showing significant alterations. Differences in cardiovascular risk factors (principally differences in metabolic profile between patients and controls) among the various populations studied might explain these discrepancies. However, endothelial dysfunction seems to be a well-recognized trait of PCOS in both micro-and macro-circulation, although not all the investigators agree.
Regarding the pathophysiology of the phenomenon, it has been postulated that insulin-resistant individuals exhibit resistance to both the metabolic and vascular actions of insulin. PI3K, a key intracellular signalling step of insulin action, has been shown by in vivo studies to be defective in the insulin-resistant state, as well as in PCOS.141 The signalling pathways by which insulin mediates glucose uptake and nitric oxide production converge at the PI3K/Akt.182,250 Consequently, disruption of this pathway may link different pathogenic mechanisms, leading to impaired glucose utilization and endothelial dysfunction. Furthermore, hyperinsulinaemia drives ET-1 production by endothelial cells via the PI3K pathway, further impairing endothelial function by competing with NO; ET-1 has been shown to inhibit insulin signalling via the same pathway.183,214 ET-1 levels have been found increased in women with PCOS.6 These findings support the hypothesis that, in women with PCOS, the abnormal vasculature status could be due to impaired insulin vasodilator functions, mediated by PI3K.251
On the other hand, there are data that support a role of hyperandrogenaemia in vascular reactivity in PCOS,7 but the mode of action of androgens remains unknown. Androgen receptors are known to exist on the vessel wall, and a direct effect of androgens in the vasculature cannot be excluded.252 Alternatively, androgens may act synergistically with insulin resistance,253 inflammatory cytokines177,254 or angioconstrictive peptides6 on endothelial function. Androgens may promote monocyte adhesion to endothelial cells, a proatherogenic effect mediated, at least in part, by increased endothelial vascular cell adhesion protein-1 (VCAM-1) expression,255 which has been found elevated in women with PCOS.26 There are also some interesting data favouring a compensatory role of adrenal androgens and particularly DHEAS for the adverse effects of dyslipidaemia and insulin resistance.19
Recently, correlations between various cardiovascular risk factors, insulin resistance, hyperandrogenaemia, endothelial dysfunction and chronic inflammation have been detected in PCOS populations, supporting the concept of their interplay on the vascular bed pathophysiology of women with PCOS.26 Whether these interactions could contribute to long-term sequelae, namely in CVD events, has not been clarified. Nevertheless, the principal candidates’ effectors for cardiovascular risk factors and for endothelial dysfunction presence remain insulin resistance and hyperandrogenaemia, the basal characteristics of PCOS.
Hypotheses for endothelial dysfunction pathogenesis also implicate a number of additional atherogenic molecules. Increased expression of soluble intercellular adhesion molecule-1 and soluble VCAM-1 found in patients with PCOS26,177 plays an important role in focal leukocyte accumulation in subendothelial regions of atheroma;254,255 however, the causes of elevated inflammatory molecules remain unclear.26 The associations between chronic inflammation and endothelial dysfunction with features of PCOS, such as obesity,256,257 may have an additional aggravating role. On the other hand, high testosterone levels have been found to induce VCAM-1 expression.
The central role of insulin resistance and hyperandrogenaemia can also be certified by the beneficial effects of the action of insulin sensitizers on the endothelium, since those agents improve those pathogenic effectors. Recent studies make efforts to shed light on mechanisms of action of insulin-sensitizing agents on the endothelium. Notably, in one study, metformin was shown to improve endothelial function, by lowering atherogenic molecules, like adhesion molecules177 or AGEs serum.228 It has been suggested that metformin has an inhibitory effect in the glycation process258–260 by interfering with the synthetic pathway of AGEs through the trapping of methyl-glyoxal and of other dicarbonyl compounds responsible for the glycation process in vivo.261 Therefore, the action of metformin may lead to reduced synthesis and/or increased detoxification of AGE metabolites.262,263 Regarding the clearance of AGEs, intact PI3K activity is required for the activation of the macrophage scavenger receptor, which is one of the major mechanisms for their clearance.264 PI3K, a key intracellular signalling step of insulin action, has been shown to be defective in an insulin-resistant state and in PCOS.141 The decreased PI3K activity present in insulin-resistant PCOS may link the pathogenic mechanisms of PCOS insulin resistance with decreased AGE clearance.141 Nevertheless, a mechanism independent from the antihyperglycaemic effect has been shown for metformin in preventing microvascular alterations.265
Inositols also have insulin-sensitizing properties that may be proved to be of clinical significance, with or without metformin co-administration.249 However, FMD has been improved by spironolactone administration alone as opposed to its co-administration with oral contraceptive pills, where no improvement was seen in 6 months in the context of FMD, carotid artery stiffness index and IMT; whereas at 12 months, a deterioration in IMT was observed.118 Notably, the combination of oral contraceptives, drospirenone and metformin for 6 months resulted in FMD improvement, possibly due to the impact of metformin.116 However, the addition of drospirenone in oral contraceptives also improved FMD but deteriorated IMT without alteration of CRP levels, implying an ill-defined role for drospirenone.68
Finally, lifestyle amendments, such as a structured exercise training programme, heat therapy, carnitine or vitamin administration, as well as laparoscopic ovarian drilling, improved FMD and/or IMT, implying that the role of lifestyle changes or hormonal control are necessary for a better cardiovascular risk-factor profile.87,122,123–125
It can be concluded that endothelial dysfunction is more likely to occur among patients with PCOS than in healthy controls. In order to confirm this widely accepted observation, there is a need for large-scale, well-designed prospective studies of cardiovascular outcomes in PCOS for a better understanding of the role that the cardinal features of PCOS, insulin resistance and hyperandrogenaemia may have on vascular function. Long-term cohort studies are particularly needed in the different phenotypes of PCOS, in order to provide information for the target–to–treat in each phenotype, since different modes of treatments may be used in different sub-phenotypes with differences in the cardiometabolic or reproductive risk profile of the syndrome.
Since, there are no universal specific diagnostic signs or characteristic phenotypes that could preclude the response to different therapeutic modalities, it seems important to assess the predominant symptoms and risk factors in the metabolic and reproductive aspects of each woman, with the aim of producing individualized treatments. Based on the pathophysiological role of insulin resistance, insulin-sensitizing agent administration can be justified, particularly as a treatment of choice in hyperinsulinaemic obese or non-obese women. Other interventions, such as lipid-lowering agents, antioxidants, oral contraceptives and anti-androgen agents have to be further investigated in order to assess their role as therapeutic tools in PCOS management, since their effects on the cardiovascular properties in PCOS are missing.
Finally, large-scale, long-term follow-up studies should be designed based on the established cardiovascular risk factors in order to identify the best clinical and biochemical markers that could be used in clinical practice for the prevention of a potentially ‘silent’ trigger of CVD, which can occur early in life, masked behind hormonal and reproductive signs and symptoms.







