Type 2 diabetes mellitus (T2DM) is a severe public health issue notably impacting human life and health expenditure. Around 9.3% (463 million people) of the global population were living with diabetes in 2019, and this is projected to increase to 10.2% (578 million people) by 2030 and 10.9% (700 million people) by 2045.1,2 Diabetes impacts functional capacity and quality of life, and ultimately causes significant morbidity and premature mortality. In 2019, diabetes was the tenth biggest cause of death worldwide, directly causing an estimated 1.5 million deaths.3 Despite lifestyle treatments such as a healthy diet, frequent physical activity and maintaining a normal body weight being critical pillars of diabetes management, achieving persistent glycaemic control with non-pharmacological techniques is difficult.4,5
Recent studies have investigated the benefits of intermittent fasting (IF), which involves repeatedly and purposefully interrupting or drastically reducing energy intake for a period, for people with obesity and T2DM. IF has also been suggested as a glycaemic control and weight loss strategy with additional cardio-metabolic benefits.6–9 Although it has not yet been standardized, intermittent or short-term energy restriction through very low-calorie diets is a common IF regimen.4,5 Time-restricted feeding, which allows for only 4–8 hours of feeding per day (16–20 hours of fasting per day), is one of the most popular IF regimens.10,11 Other popular IF techniques include alternate-day fasting and periodic fasting, which call for a circular diet that includes fasting for 1 or 2 days per week (burning ≤25% of the required calories) and eating normally for the rest of the week.12–14
The impact of IF has been observed on a range of health outcomes including risk factors for metabolic diseases, such as weight, blood pressure, waist circumference, body fat, lipid distribution and blood glucose.15–18 Previous studies on people with T2DM have shown that IF can result in comparable weight loss and glycated haemoglobin (HbA1c) reduction as traditional dietary recommendations.19–22 However, in some randomized crossover experiments, IF had no impact on lipid and glucose metabolism.23–24
These findings demonstrate that IF inconsistently impacts numerous metabolic parameters. Furthermore, the small sample sizes of these studies prevent drawing of firm conclusions. Therefore, a thorough and methodical meta-analysis that includes all eligible randomized controlled trials, a large sample size, and a range of IF types is needed to ascertain the effectiveness of IF interventions on glycaemic control in people with T2DM. This comprehensive review and meta-analysis evaluates the effect of IF treatments on glycaemic control in people
with T2DM.
Materials and methods
This systematic review was submitted to the International Prospective Register of Systematic Reviews and followed the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines.25 The protocol of this study was registered (PROSPERO ID: CRD42022328528).
Databases and search strategy
This meta-analysis was done and presented in accordance with the PRISMA standards. We conducted a comprehensive search of electronic databases, including PubMed, Embase and Google Scholar, for articles published before 24 April 2022, regardless of area. In addition, the reference lists of particular articles were examined. As search terms, we used intermittent fasting, intermittent energy restriction, type 2 diabetes, HbA1c and fasting blood glucose.
Inclusion and exclusion criteria
We selected articles that met the following standards: (1) the participants had T2DM and were at least 18 years old; (2) interventional research, which may consist of randomized parallel-arm or crossover trials; (3) intervention: i) 24 hours of complete fasting; ii) intermittent restricted energy intake; iii) time-restricted feeding (feeding permitted for only 4–8 hours daily with 16–20 hours of fasting); (4) the IF intervention could be applied on alternate days, twice weekly or continuously and was compared with standard dietary recommendations consisting of regular eating hours (control group); (5) the changes in HbA1c were recorded; and (6) the duration of the trial exceeded 6 weeks.
The criteria for exclusion were: (1) trials without a control group, or other study designs; (2) studies that lack a HbA1c factor as an outcome or did not provide enough information; (3) non-human samples, reviews and case studies; (4) studies that were reported in a language other than English; (5) absence of time restrictions for intermittent energy restrictions (IER) and fasting. Cienfuegas et al. and Harvie et al. have performed studies with outcomes measured at different levels or time. Both of their outcomes were included in our meta-analysis and stated as Cienfuegas et al. [a] & [b]; Harvie et al. [a] & [b]. Therefore, the total included studies in our meta-analysis are 11 but HbA1c level outcomes shows analysis of 13 studies.
Data extraction
Two researchers separately reviewed databases and deleted redundant studies. Pairs of independent reviewers first looked over the titles and abstracts of all articles that met the inclusion criteria, before reading the entire text of applicant studies. Disputes concerning a study’s inclusion were discussed and resolved by a third reviewer. The reference lists of the chosen papers were also examined. Independent data extraction was done by two researchers. For each included study, the following parameters were extracted: basic information (first author, year, title and country), clinical features (participants’ characteristics, dietary habits, intervention follow-up duration and results), and method and design (randomization procedure and data analysis technique). The variation in HbA1c and fasting glucose levels were the most crucial finding. We emailed the corresponding author when we needed information specific to the study.
Risk of bias assessment
Using the updated Cochrane risk of bias assessment tool for randomized trials,26 two independent reviewers assessed the likelihood of bias in trials based on the outcomes (HbA1c or fasting glucose). The Cochrane Handbook for Systematic Reviews categorizes the risk of bias for each domain as low, high or unclear based on the signal questions for each item.
Data analysis
The mean difference between before and after IF implementation and their respective 95% confidence intervals (CIs) were used to evaluate the effects of IF on HbA1c. To measure trial heterogeneity, Cochrane’s Q statistic and the I2 statistical approach were applied. A random-effect meta-analysis model was used if the pertinent p value was less than 0.05 and I2 was higher than 50%. Otherwise, a fixed-effect model was chosen.For each outcome, funnel plots depicting effect sizes versus standard errors were constructed and visually evaluated to assess the probability of bias. For statistical analysis, we used RevMan 5.4 software (Cochrane, London, UK).
Results
Study characteristics
Figure 1 depicts a flowchart of the literature search procedure. Using this search method, we evaluated 3,153 studies after deleting 582 duplicates. Screening titles and abstracts eliminated an additional 3,087 articles. The remaining 66 citations’ entire texts were evaluated in greater detail to determine their eligibility. A further 31 articles were excluded because they were review articles, 15 studies were excluded because they were guidelines or recommendations, and nine studies were excluded because they were systematic reviews and meta-analyses. Eleven studies including 879 patients were included in the final data synthesis.22,27–36
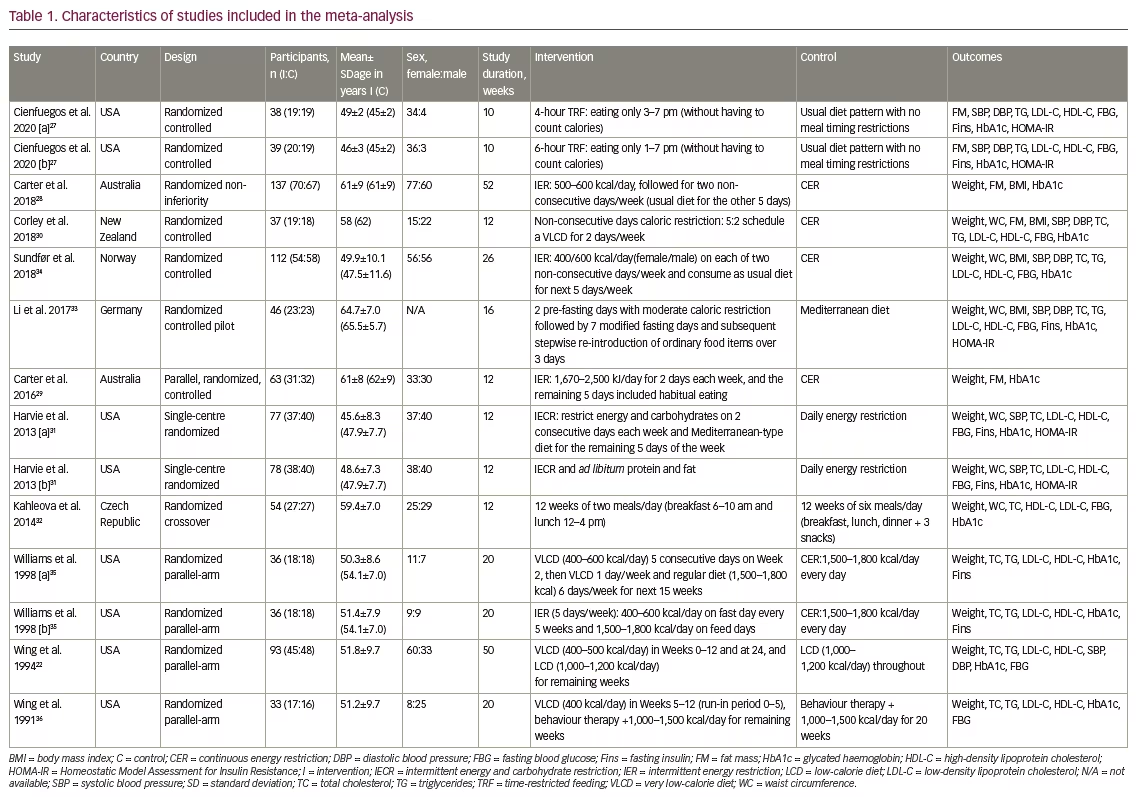
Table 1 presents the features of and interventions used in the 11 selected studies. All were randomized parallel-arm trials, with the exception of one crossover trial and one non-inferiority trial. The studies were published between 1991 and 2020. The sample sizes in these trials were 33–137, and the duration of the interventions was 10–12 months. The mean age of the patients was 45.2–65.5 years, and 38.77% were male. The IF interventions used varied, with one study evaluating time-restricted feeding, two studies evaluating caloric restriction, four studies evaluating intermittent energy restriction and four studies evaluating extremely low-caloric diet.
Risk of bias and quality assessment of studies
Figure 2 and the supplementary table provide information on all Cochrane risk-assessment domains and methodology findings. The majority of studies had a minimal risk of bias. In case of any missing information from the study findings and after receiving responses from the corresponding authors of included studies, all authors of this analysis reached a consensus on the next steps.
Outcome measures
Glycaemic control (glycated haemoglobin and fasting blood glucose levels)
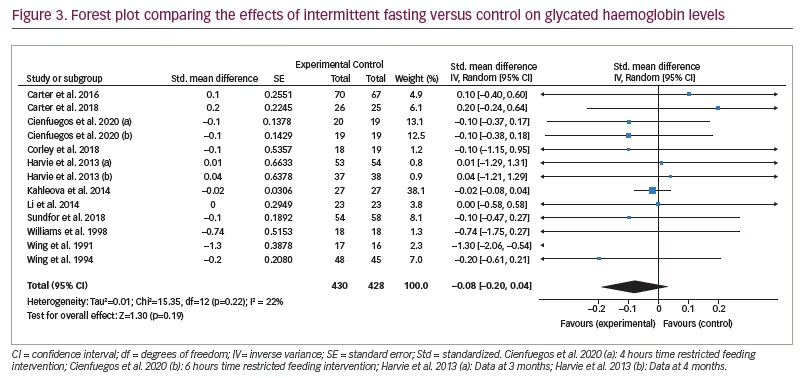
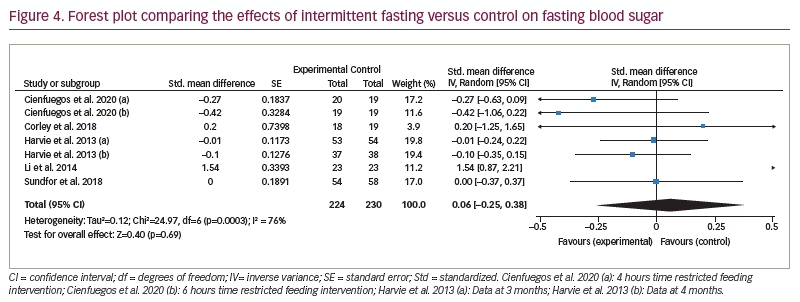
Figure 3 represents the overall analysis of 11 studies (13 arms) to measure the effect of IF on patients’ HbA1c level. There was no statistically significant difference between IF and control groups (statistically meaningful difference [SMD] -0.08, 95% CI -0.20 to 0.04;p=0.19, I2=22%). Overall, seven studies on patients’ fasting blood glucose value (FBG) were analysed, and the meta-analysis revealed no significant difference between the IF and control groups (SMD 0.06, 95% CI -0.25 to 0.38;p=0.69, I2=76%; Figure 4).
Sensitivity analysis
Glycated haemoglobin level
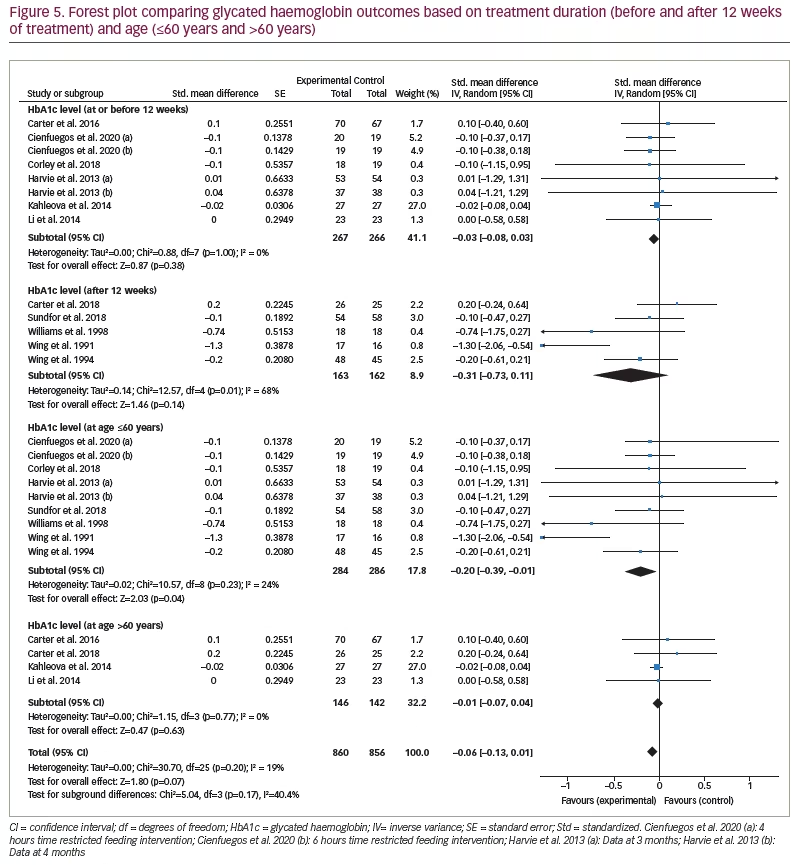
Five studies monitored HbA1c levels after 12 weeks’ treatment duration, with the remaining eight studies monitoring HbA1c level at ≤12 weeks’ treatment duration. When analysed by treatment duration, there was still no significant difference between the IF and control groups (SMD -0.03, 95% CI -0.08 to 0.03;p=0.38, I2=0%). Further, meta-analysis was performed including only the five studies with reported HbA1c level after 12 weeks’ treatment (SMD -0.31, 95% CI -0.73 to 0.11;p=0.14, I2=68%; Figure 5).
Four studies included participants >60 years of age and nine studies included those ≤60 years of age. When analysed by patient age, the reported change in the HbA1c levels of IF and control groups was statistically significant (SMD -0.20, 95% CI -0.39 to -0.01;p=0.04, I2=24%; Figure 5).
Fasting blood sugar levels
One study monitored FBG after 6 months, and six studies monitored FBG at ≤6 months. When the >6 months study was excluded from analysis, there was still no significant difference between the FBG values of the IF and control groups (SMD 0.09, 95% CI -0.30 to 0.47;p=0.65, I2 =80%; Figure 6).
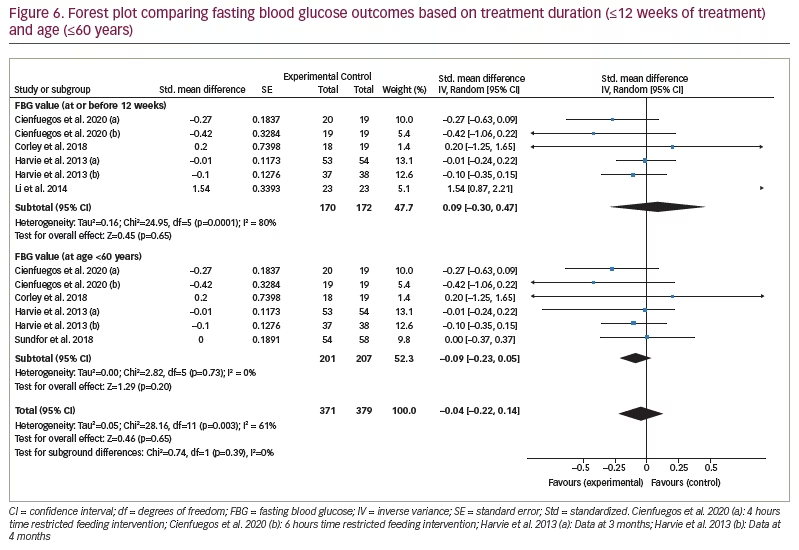
Similarly, one study had participants over the age of 60 years, and the remaining six had participants aged ≤60 years. When the >60 years of age study was excluded, there was no significant difference in the FBG values of the IF and control groups (SMD -0.09, 95% CI -0.23 to 0.05;p=0.20, I2=0%; Figure 6).
Publication bias
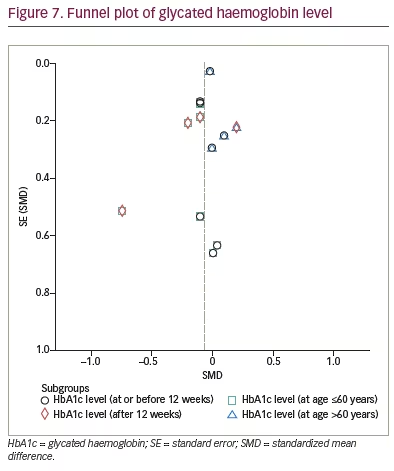
The funnel plot illustrating the effect of IF on glycaemic control demonstrates a pattern that is nearly symmetric (Figure 7 and 8), which suggests that the findings were less likely to be influenced by publication bias.
Discussion
IF has gained recognition as a method to improve metabolic health. In IF, eating habits are based on eating very few or no calories during periods from 12 hours to many days, while following a regular routine. An imbalance in the levels of adiponectin and leptin is a factor in the altered metabolism that increases the risk of developing T2DM.37,38 Interestingly, various studies have shown that IF leads to lower leptin levels, as well as higher adiponectin levels, which can improve insulin resistance.37–39
We have gathered clinical trials on the impact of IF on glycaemic control among patients with T2DM. There were only a handful of studies that focused on the effect of IF on metabolism of lipids which was not included in our meta-analysis. Also, these studies varied in terms of participants’ age, duration of IF, restriction of calorie intake and timing of outcome measurements. Hence, this meta-analysis was designed to test whether IF significantly impacted patients’ HbA1c and FBG levels, with a pooled analysis of outcomes measured at different intervals. Overall, there was no significant change in patients’ HbA1c and FBG levels between IF and control.
In one study, significant reductions in HbA1c and weight were reported for almost all patients.19 Also, there were no side effects noted among the patients on IF. Similarly, another meta-analysis also reported the positive impact of IF on reducing HbA1c level and body weight.40,41 These results are in line with some of the studies included in this review, which have reported that IF is superior to control group in terms of weight reduction and glycaemic control.30–32,35,36 On the contrary, other included studies showed no significant difference in HbA1c levels following IF versus the control group.27–29,33,34 Another meta-analysis has also concluded that there was no significant impact of IF on patients’ HbA1c levels, although IF may be useful in preventing metabolic disorders.40 These results are in line with the present meta-analysis, which reports no significant difference in glycaemic control between patients using IF versus another intervention.42,43 Our meta-analysis on performing sensitivity analysis found that benefits of IF also depend on participants’ age as IF was well tolerated among patients aged ≤60 years and has a significant impact on HbA1c levels in this age group. There was no significant impact of IF on HbA1c among patients over the age of 60 years.
Pooled analysis of the studies included in our meta-analysis showed no significant reduction in patients’ FBG levels following IF. On the contrary, a study where IF was followed for 12 months reported significant reductions in fasting insulin levels and homeostatic model assessment of insulin resistance (HOMA-IR) levels in the alternate-day fasting group.44 Similarly, another systematic review found that IF reduces participants’ FBG levels.41 Studies have also reported that insulin decreases because of increased insulin sensitivity and, hence, decrease fasting and post-prandial blood glucose in patients with diabetes.40, 44
A few studies comment that IF and continuous energy restrictions have equal benefits in achieving long-term weight and glycaemic control.19,40,42,44 The IF diet differs from the ketogenic or low-calorie diet in that it does not restrict carbohydrate intake; therefore, the direct effect on blood glucose levels in the short-term is unknown. Yet, IF is certainly beneficial in regulating blood glucose levels during fasting. IF can enhance insulin sensitivity in the long-term and therefore needs to be practised by patients with diabetes. Moreover, it is important to follow standard guidelines while practicing any dietary restrictions to avoid serious adverse effects.
There were some limitations on this analysis. First, there is heterogeneity among the studies and dietary interventions, with the treatment duration likely being the primary source of heterogeneity. As a result, the random-effects models were used in this analysis for merging, and sensitivity analyses were carried out in accordance with potential sources. Second, there were only a small number of randomized controlled trials that met the inclusion criteria, and sample sizes were small. It was also difficult to reach a firm conclusion about how IF affected glycaemic control, because the intervention duration ranged from 8 weeks to 12 months. Additionally, it was not possible to determine whether IF was safe for patients with T2DM who were taking insulin in our analyses, which is particularly important.
Conclusion
IF and usual diet pattern have no difference in terms of glycaemic control. Although, IF might be used as a preventative diet pattern in the pre-diabetic population, as it works well in the long-term to achieve controlled sugar levels. It is clearly evident from our meta-analysis that IF alone does not reduce blood glucose levels in patients with diabetes. Further clinical trials are required with uniform or standard IF intervention to study its impact in depth.







