Highlights
-
Enhanced external counterpulsation (EECP) is evaluated for glycaemic control in type 2 diabetes.
-
Glycated haemoglobin reduction immediately after EECP therapy conclusion, after 2–4 weeks and after 7–12 weeks was -0.70%, 1.04% and 0.98%, respectively.
-
EECP is associated with a significant lowering of blood pressure.
-
EECP significantly lowers the levels of high-sensitivity C-reactive protein, a measure of systemic inflammation.
Enhanced external counterpulsation (EECP) therapy is a noninvasive, nonpharmacological outpatient/daycare treatment approved by the US Food and Drug Administration therapy that has been used for treating refractory angina in people living with coronary artery disease (CAD) for more than three decades now.1,2 During EECP therapy, pneumatic compression cuffs are applied to the calf and the lower and upper thigh of each leg.2 These cuffs are inflated sequentially through computer-generated signals while synchronized to the patient’s R wave on the electrocardiogram. EECP therapy leads to retrograde blood flow in the aorta, resulting in a diastolic augmentation of blood flow and improved coronary perfusion pressure during diastole.2 Thereafter, the cuffs simultaneously deflate before the onset of systole, decreasing vascular resistance, assisting with systolic unloading and decreasing cardiac workload, thereby reducing angina.2 Mechanistically, shear stimulus in the femoral and brachial arteries results in increased endothelial nitric oxide (NO) production, decreased systematic inflammation (high-sensitivity C-reactive protein) and improved endothelin-1 levels.3,4 The increased production of vasodialators such as NO results in reduced myocardial oxygen demand, increased venous return and cardiac output, improved endothelial function, promoted coronary collateral development and recruitment, and prolonged time to exercise-induced ST depression.4 Seven weeks of EECP therapy have been found to reduce angina symptoms and frequency in patients with CAD; however, the benefits of EECP therapy have been found to be more blunted in people with diabetes.5
Skeletal muscles are a major organ for glucose disposition and storage following meal-induced hyperglycaemia. Physiologically, glucose is uptaken and stored in skeletal muscles through three major mechanisms: insulin-mediated glucose uptake, which is impaired in people with type 2 diabetes mellitus (T2DM) due to skeletal muscle insulin resistance;6 muscle contraction-mediated glucose uptake, which explains the beneficial impact of glycaemic control in T2DM;7 and NO-mediated glucose uptake.8 NO-mediated glucose uptake may explain the beneficial impact of EECP therapy on glycaemia in T2DM, as EECP therapy works by enhancing NO formation, thus leading to increased NO-mediated glucose uptake in skeletal muscles. Several randomized controlled trials (RCTs) evaluating the effect of EECP therapy on glycemic parameters in patients with T2DM have been published.9–11 However, no systematic review and meta-analysis providing a holistic view of the role played by EECP therapy in treating T2DM has been published to date. Hence, we conducted a systematic review and meta-analysis to evaluate the safety and efficacy of EECP therapy for glycaemic control in patients with T2DM compared with control patients.
Methods
The systematic review and meta-analysis were conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The study was registered in the International Prospective Register of Systematic Reviews (PROSPERO); the registration number is CRD42023434533. For this systematic review and meta-analysis, we considered RCTs involving people with T2DM or prediabetes receiving EECP therapy in the study group compared with participants receiving placebo or any other medication in the control group. Patients with other forms of diabetes like type-1 diabetes, gestational diabetes, and other rarer monogenic causes of diabetes were excluded. The primary outcome of the meta-analysis was changes in glycated haemoglobin (HbA1c) from baseline. The secondary outcomes were changes in fasting plasma glucose (FPG), post-prandial glucose (PPG), lipid parameters, inflammatory markers and any adverse events. Separate analyses were performed for controls receiving placebo, labelled as placebo or passive control group, and controls receiving other antidiabetes medications, labelled as active control group.
We systematically searched the Embase, Web of Science, Cochrane Library, MEDLINE (PubMed), ClinicalTrials.gov, CNKI, Clinical Trials Registry-India (CTRI) and Google Scholar databases for the following keywords or MeSH terms: (enhanced external counter-pulsation therapy) OR (external counter-pulsation therapy) OR (counter-pulsation therapy) for the articles published untill May 2023. Methodologic details regarding literature review have been elaborated in a previous meta-analysis published by our group.12 The risk of bias assessment was done by three authors independently using the risk of bias assessment tool in the Review Manager (RevMan) version 5.4 software. The different types of bias that were assessed had been elaborated in previous meta-analyses conducted by our group: selection bias, performance bias, detection bias, attrition bias, reporting bias and other bias.12,13 Other bias included sources of funding, especially when there is a pharmaceutical/organization involvement in the manufacture and sale of EECP devices, and conflict of interests. A random effects model was used for the meta-analysis. Forest plots were generated to assess the heterogeneity for all outcomes. Specifically, heterogeneity was analysed using the χ2 test on N-1 degrees of freedom, an alpha of 0.05 used for statistical significance and the I2 test.13,14 The certainty of the evidence of the major outcomes in this meta-analysis was evaluated using the Grades of Recommendation, Assessment, Development and Evaluation (GRADE) approach.15 A table highlighting the grading of key outcomes was generated using the GRADE software. The details have been elaborated elsewhere.13 Publication bias was assessed for key outcomes using funnel plots (Supplementary Figure S1).16
Results
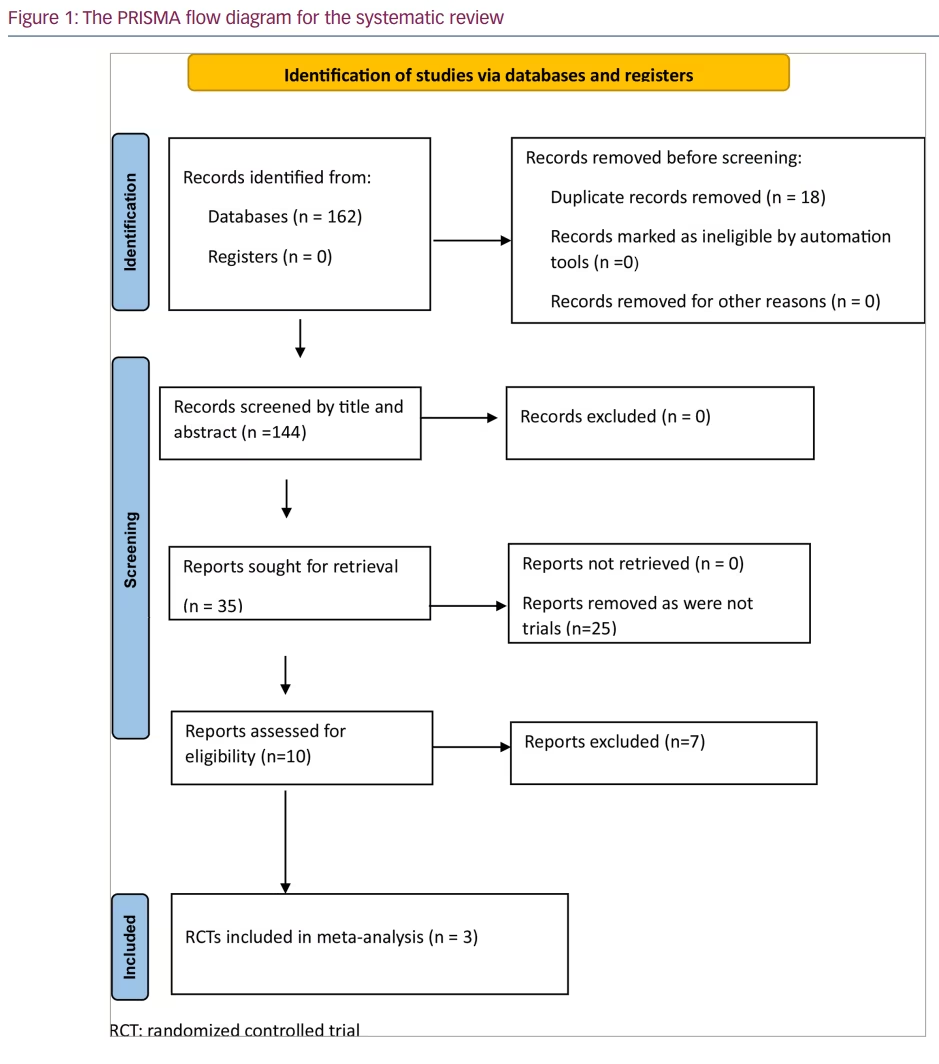
A total of 162 articles were found from databases after the initial search (Figure 1). Eighteen duplicates were removed. After screening the titles and abstracts of the remaining 144 articles, the search was reduced to 35 studies, which were evaluated in detail for inclusion in this meta-analysis (Figure 1). Finally, three RCTs were found to fulfil all the inclusion and exclusion criteria and were analysed in our systematic review and meta-analysis.9,10,17 The three articles included in our systematic review and meta-analysis are described in detail in Table 1.
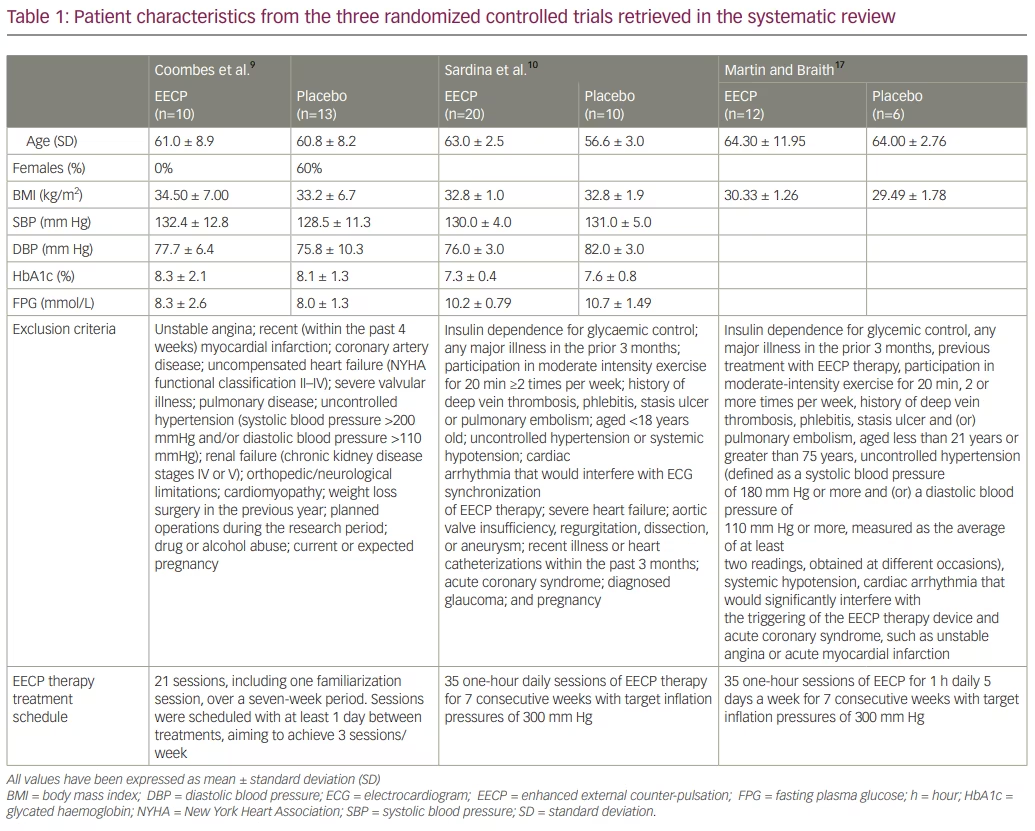
In 2 of the RCTs, the study population was patients with T2DM.9,10 In the study by Martin and Braith, 1 of 6 participants (17%) in the control group and 4 of 12 (33%) participants in the EECP therapy group had prediabetes; the remaining participants had T2DM.17 All three trials compared EECP therapy with placebo over and above the standard of care in T2DM.9,10,17 The study by Hoong et al. did not have a comparator group and hence was excluded from the analysis.11
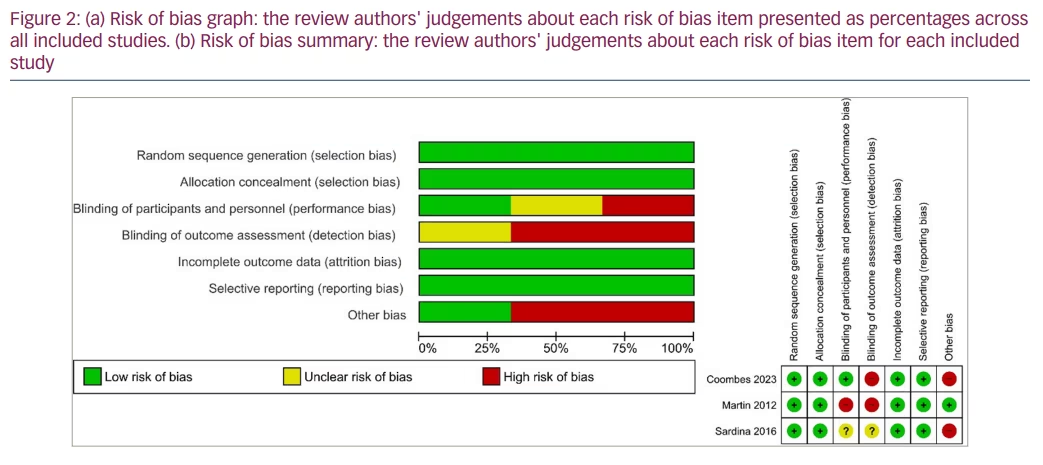
The risk of bias in these studies is summarized in Figures 2a and b. The risk of selection, attrition and reporting bias was judged to be low in all three studies.9,10,17 There was a low risk of performance bias in one out of the three studies.9 The risk of detection bias was high in two of the three studies,9,17 while the risk of bias was unclear in Sardina et al.10 Other bias was judged to be at high risk in two out of the three studies.9,10
Outcomes after therapy completion
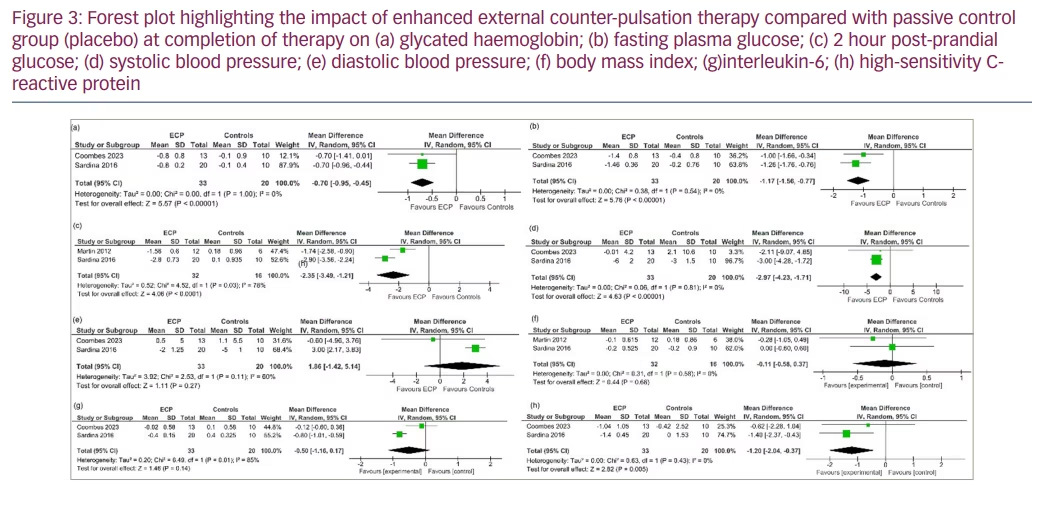
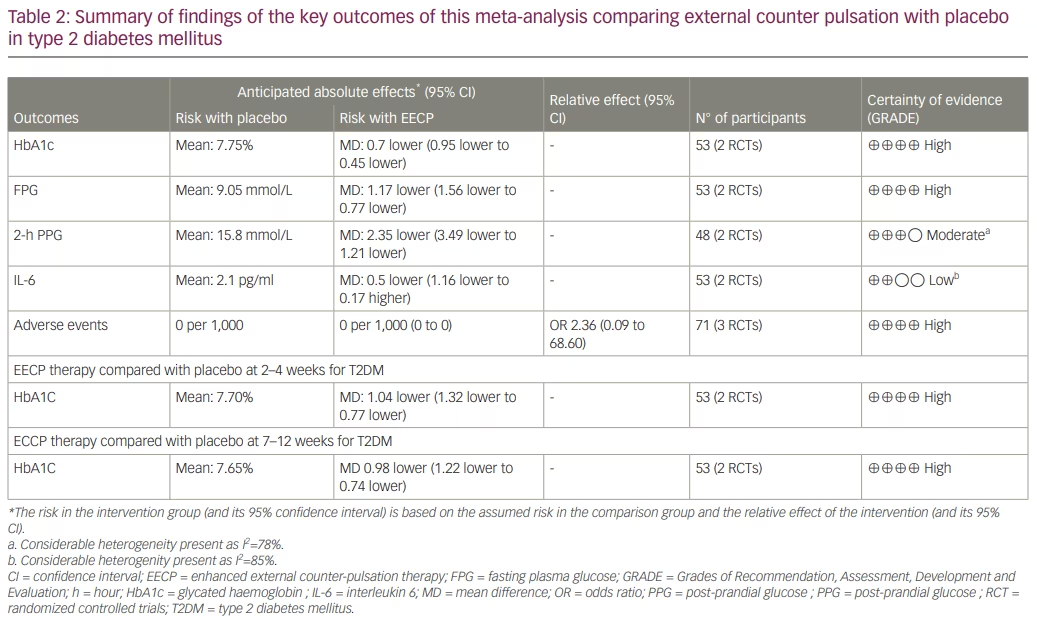
Therapy was considered completed at around 35 sessions of EECP therapy over 7–8 weeks. Data from three studies involving a total of 71 people with T2DM or prediabetes undergoing EECP therapy were analysed to determine the impact of EECP therapy compared with the control participants.9,10,17 At therapy completion, patients receiving EECP therapy had significantly lower HbA1c levels (mean difference [MD] -0.70%, 95% confidence interval [CI] -0.95, -0.45; p<0.00001; I2=0% [low heterogeneity (LH)]; high certainty of evidence [HCE]; Figure 3a), FPG (MD -1.17 mmol/l, 95% CI -1.56, -0.77); p<0.00001; I2=0% [LH]; HCE; Figure 3b) and 2-hour PPG (MD -2.35 mmol/l, 95% CI -3.49, -1.21; p<0.0001; I2=78% [moderate heterogeneity (MH)]; moderate certainty of evidence; Figure 3c) levels compared with the placebo or passive control group. SBP was significantly lower in the EECP therapy group compared with the placebo or passive control group (MD -2.97 mm Hg, 95% CI -4.23, -1.71); p<0.00001; I2=0% [LH]; Figure 3d), while DBP was similar between the two groups (MD 1.86 mm Hg, 95% CI -1.42, 5.14; p=0.27; I2=60% [MH]; Figure 3e). Body mass index (MD -0.11, 95% CI -0.58, 0.37; p=0.66; I2=0% [LH]; Figure 3f) and interleukin-6 (MD -0.50 pg/ml, 95% CI -1.16, 0.17; p=0.14; I2=85% [high heterogeneity (HH)]; low certainty of evidence; Figure 3g) were similar between groups. C-reactive protein (MD -1.20; 95% CI -2.04, -0.37; p=0.005; I2=0% [LH]; Figure 3h) was significantly lower in the EECP therapy group compared with the placebo or passive control group after therapy completion. Adverse effects were similar between the groups (risk ratio 2.36, 95% CI 0.11–52.41; p=0.59; LH; HCE). The only adverse event related to EECP therapy was chafing on the legs where the cuffs had been applied. No patient experienced withdrawal from undergoing EECP therapy at trial completion. In one trial, the participants reported that they had sensitive skin, and wearing cotton pants and applying moisturiser during therapy helped to reduce their symptoms.9
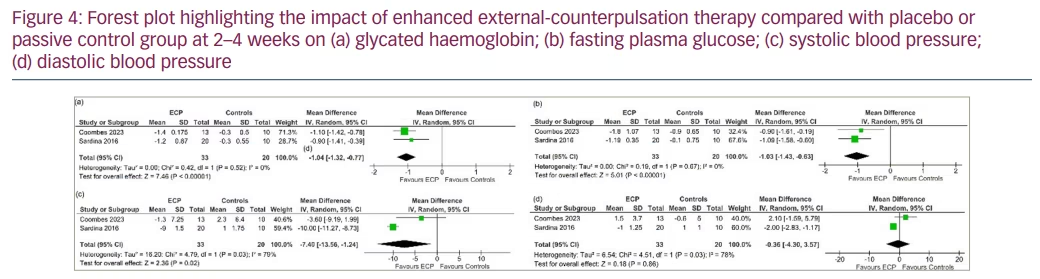
Outcomes at 2–4 weeks after therapy completion
Data from 2 studies involving 53 people with T2DM were analysed to determine the impact of EECP therapy compared with placebo at 2–4 weeks after therapy completion.9,10 Compared with patients in the placebo or passive control group, patients receiving EECP therapy had significantly lower HbA1C (MD -1.04%, 95% CI -1.32, -0.77; p<0.00001; I2=0% [LH]; HCE; Figure 4a), FPG (MD -1.03 mmol/l, 95% CI -1.43, -0.63; p<0.00001; I2=0% [LH]; Figure 4b) and SBP (MD -7.40 mm Hg, 95% CI -13.56, -1.24; p=0.02; I2=79% [MH]; Figure 4c) levels at 2–4 weeks after therapy completion. DBP (MD -0.36 mm Hg, 95% CI -4.30, 3.57; p=0.86; I2=78% (MH); Figure 4d) was similar between the groups.
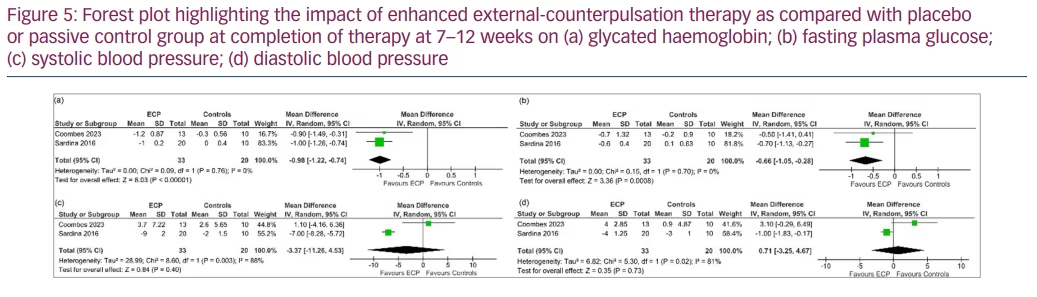
Outcomes at 7–12 weeks after therapy completion
Data from 2 studies involving 53 people with T2DM was analysed to determine the impact of EECP therapy compared with placebo at 7–12 weeks after completion of therapy.9,10 Compared with patients in the placebo or passive control group, patients receiving EECP therapy had significantly lower levels of HbA1C (MD -0.98%, 95% CI -1.22, -0.74; p<0.00001; I2=0% [LH]; HCE; Figure 5a) and FPG (MD -0.66 mmol/l, 95% CI -1.05, -0.28; p=0.0008; I2=0% [LH]; Figure 5b). SBP (MD -3.37 mm Hg, 95% CI -11.26, 4.53; p=0.40; I2=88% [HH]; Figure 5c) and DBP (MD 0.71 mm Hg, 95% CI -3.25, 4.67; p=0.73; I2=81% [HH]; Figure 5d) levels were similar between the two groups.
The key findings of the study and the side effect profile of EECP therapy are summarized in Table 2. Funnel plots were plotted to evaluate publication bias and are shown in Supplementary Figure S1.
Discussion
Standard EECP therapy consists of around 35 1-hour sessions, which typically occur once per day from Monday to Friday.2 A maximum of two sessions per day can take place, subject to the patient’s wishes and tolerance. Nearly a fifth of the patients who are not able to complete the 35-sessions program may need extended therapy.2,18 Commonly accepted contraindications for EECP therapy include arrhythmias that interfere with machine triggering, bleeding diathesis, active thrombophlebitis, severe peripheral artery disease, severe aortic valve disease, prior history of aortic surgery and severe tachycardia (>120 beats/min).18 Decompensated homeostasis needs to be stabilized before considering EECP therapy.
Our analysis noted an impressive reduction of -0.70% in HbA1c level in participants with T2DM or prediabetes following EECP therapy completion. These patients had EECP therapy for glycaemic control and had no major underlying cardiac disease. Notably, this reduction in HbA1c is not transient but last even up to 7–12 weeks after completing therapy. HbA1c level reduced by 1.04% and 0.98% at 2–4 weeks and 7–12 weeks after completing therapy, respectively. However, the glycaemic durability of EECP therapy for glycaemic control beyond 12 weeks is not known. Hence, longer follow-up studies investigating glycaemic durability are warranted for determining whether repeat EECP therapy follow-up sessions are needed in patients with T2DM.
A similar reduction was also noted in FPG and 2-hour PPG. It is also important to note that a significant reduction in blood pressure was also noted with EECP therapy at the end of the therapy, which persisted till about 2–4 weeks after therapy and thereafter became not significant after 7–12 weeks of therapy. EECP therapy was well tolerated without any major side effects warranting treatment discontinuation.
Our analysis shows that EECP therapy is effective in reducing blood glucose and blood pressure in people with diabetes without CAD. This reduction in blood glucose was accompanied by a reduction in hs-CRP levels, which is a measure of systematic inflammation. However, interleukin-6 levels were similar in the study and the control groups. Our analysis is limited by the small number of patients evaluated. Hence, we need bigger trials with a larger number of people with T2DM with a longer duration of follow-up. The rationale for analysing both people with T2DM and prediabetes together, as was done in one of the RCTs, is that dysglycaemia is a continuum, and there is no reason for EECP therapy not to work in prediabetes if it works in T2DM.17 Furthermore, the number of patients was too small to analyse data from people with prediabetes to be analysed separately and is a limitation of this meta-analysis.
Our analysis supports the use of EECP therapy as an adjunctive therapy in people on polypharmacy for T2DM. EECP therapy may help to lower the pill burden or reduce the total daily dose of insulin requirement in these patients. However, these data need to be confirmed by bigger, multicentre clinical studies before they can be replicated in clinical practice. Furthermore, the process of EECP therapy needs to be simplified to be implemented on a large scale; as of today, this is not possible. The cost also remains a major barrier to routinely using EECP therapy, which is a required treatment for a chronic condition such as T2DM. Furthermore, patients with T2DM must go to a medical centre to undergo EECP therapy; this represents a major limitation to using EECP for glycaemic control. Finally, EECP therapy is a time-consuming procedure; consequently, it interferes with the personal and professional lives of the patients.
An important corollary that can be derived from our analysis is that people with T2DM on EECP therapy for angina due to CAD need to reduce the dose of their antidiabetes medication (oral antidiabetes medications and/or insulin) to prevent the risks of hypoglycaemia during and up until at least 12 weeks after EECP therapy completion. Similarly, these patients would also need to modulate the doses of their hypertension medications.
The current systematic review and meta-analysis have a few limitations. First, data are available only up to 12 weeks after EECP therapy completion. What happens beyond that point is not known. The total duration of glycaemic durability of EECP therapy remains to be determined, and it represents an important area of future research in EECP therapy. Second, our analysis included data from only 3 RCTs because not enough RCTs have been published on this subject. Hence, RCTs are urgently needed to evaluate the different metabolic aspects of EECP therapy. EECP therapy has been shown to improve post-exercise recovery in elite rugby league players.19 It has also been shown to be beneficial in CAD, one of the macrovascular complications of diabetes, thus improving the quality of life in these patients.20 EECP therapy improves exercise tolerance in people with CAD.21 EECP therapy has also been shown to reduce the risk of contrast-induced nephropathy in patients with T2DM with CAD undergoing coronary angiography or percutaneous coronary intervention.22 A few small studies have suggested the beneficial impact of EECP therapy on transcranial Doppler middle cerebral artery flow velocities and National Institutes of Health Stroke Scale scores in patients with acute ischaemic stroke.23,24 The impact of EECP therapy on other macrovascular and microvascular complications of diabetes is not known and remains an important area of future research.
To conclude, EECP therapy is effective in reducing blood glucose and blood pressure in people with T2DM, whose effects extend till at least 7–12 weeks of therapy completion. Hence, blood glucose and blood pressure should be routinely monitored in patients with angina undergoing EECP therapy with suitable modulation of drug doses to prevent hypoglycaemia and hypotension.







