Creation of a working closed loop/artificial pancreas (CL/AP) system is one of the holy grails of type 1 diabetes mellitus (T1DM). In essence, this would be a mechanical cure for a potentially devastating chronic disease. While the CL/AP still has its limitations, the results of recent research studies are encouraging. The CL/AP system also has the attention and support of organizations such as the US Food and Drug Administration (FDA) and the Juvenile Diabetes Research Foundation (JDRF), which is a large charitable organization dedicated to funding T1DM research.1 The JDRF has created a six-step pathway which ends in a fully automated CL/AP system, and the FDA has created a helpful guide with recommendations and instructions for those interested in developing a CL/AP. The purpose of this paper is to discuss the components of the CL/AP system, describe its limitations, and review recent CL/AP research studies and future directions.
Components of the closed loop/artificial pancreas system
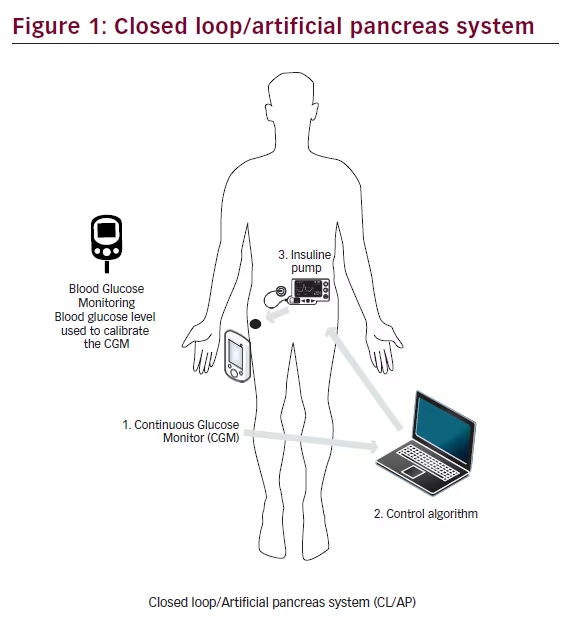
The CL/AP system is composed of an insulin pump, a continuous glucose monitor (CGM) plus calibration fingerstick blood glucose (BG) measurements to assist with accuracy,2 and a control algorithm (Figure 1). Initially, the control algorithm was housed on a laptop computer. Now, it can be either on a laptop computer or on a CL/AP-dedicated smartphone. The CGM, once calibrated with a fingerstick BG, checks the glucose in the interstitial fluid and sends the value to the control algorithm, which decides if insulin is needed or not. If insulin is needed, it instructs the insulin pump to deliver a bolus. All components of the system are in constant communication with each other to try to maintain glucose levels in a target range of 70–180 mg/dl (3.9–10 mmol/l).
Several different control algorithms are being used in CL/AP studies. The most common ones are the Proportional-Integral-Derivative (PID, ePID) algorithm, the Model Predictive Control (MPC)/Multiple Model Predictive Control (MMPC)/Multiple Model Probabilistic Predictive Control (MMPPC) algorithms, and the Fuzzy Logic (FL) algorithm.2,3 Each of these commonly used algorithms are described in more detail below. A comparison of the algorithms performed by Bequette and colleagues (2013) found the ePID to have the highest postprandial glucose values, followed by the MPC and MMPPC algorithms, and then open-loop basal/bolus insulin administration. The significance of the differences between algorithms was not discussed.4
Closed loop/artificial pancreas algorithms
Proportional-integral-derivative algorithm
The PID/ePID algorithm uses calculus and differential equations to interpret the sensor glucose (SG) readings and make decisions about insulin delivery. It has four components: proportional (P), integral (I), derivative (D), and insulin on board (IFB). It is a reactive, one-sizefits- all model.2,3
Model predictive control algorithm
The MPC algorithm and its offshoots (MMPC, MMPPC) is also a differential equation-based model. It has both predictive and adaptive modes, and decides when it is best to use each. This individualistic algorithm is easier to adapt to the user than the PID algorithm because it is able to learn the needs of each individual and accommodate accordingly. The MPC algorithm is flexible and can include things that are a function of time of day, such as meals and exercise, to ensure that the individual with T1DM receives optimal automated glycemic control. CL/AP systems using the MPC algorithm are sometimes semi-closed loop instead of fully automated, requiring the user to initiate delivery of an insulin dose or bolus when they are eating in order to minimize the post-prandial glycemic rise that may otherwise occur with a reactive system.4
Fuzzy logic algorithm
Unlike the other models, the FL algorithm does not use differential equations to determine the individual’s insulin dose. It takes input (CGM values, direction and rate of change, amount of insulin on board, etc.) into something akin to a black box called the fuzzy controller, processes the input, and gives an output (decision whether to give insulin or not). This algorithm takes other physiologic parameters into account, such as illness and stress, and allows for personalized dosing for each individual patient.5
Closed loop/artificial pancreas research
Early closed loop/artificial pancreas system studies
Like early studies using insulin pumps,6,7 the first CL/AP studies showed the system had the ability to maintain tighter glycemic control than could individuals on open-loop insulin pump therapy. However, individuals using the CL/AP systems still had some postprandial hyperglycemia (BG >200 mg/dl [11.1 mmol/l]) soon after eating, and later postprandial hypoglycemia (BG <60 mg/dl [3.3 mmol/l]) secondary to the insulin that had been delivered to control the postprandial hyperglycemia.8,9 These insulin-only studies instigated researchers to consider using adjuvant medications in the CL/AP system, which could help minimize both the postprandial hyperglycemia and the ensuing hypoglycemia.
There were three major limitations in the early CL/AP studies: insulin action, glucose sensor accuracy, and the control algorithm software. Rapid-acting insulin is still slower than what is needed to maintain BG in the 70–180 mg/dl (3.9–10 mmol/l) range. Because of the delayed onset of action and delayed peak compared with endogenous insulin, blood sugars often rise above 180 mg/dl (10 mmol/l) before the insulin that has been delivered starts working. All of the extra insulin administered to try to control the hyperglycemia then results in blood sugars <70 mg/dl (3.9 mmol/l) four to five hours after the meal.9 Early glucose sensors were less accurate than those that are currently available. There was up to a 20-minute lag time between BG and SG readings, so changes in BG were not necessarily reflected in a timely enough manner by the CGM. Today’s glucose sensors are better, with a lag time of less than 10 minutes between BG and SG readings.10,11 The CGM is also generally less accurate on day one, even today, and improves with time.12
The early control algorithm software was all reactive systems. This allowed the BG and SG to rise first, and only then was more insulin delivered by the system.3,9 Newer algorithms are working to overcome this limitation.
Multihormonal studies
Following the initial insulin-only CL/AP studies, it became clear that a CL/AP system with insulin alone was not sufficient to maintain glucose values in the desired range of 70–180 mg/dl (3.9–10 mmol/l). Investigators became interested both in attenuating hypoglycemia using glucagon and in minimizing postprandial hyperglycemia using medications such as pramlintide, glucagon-like peptide-1 (GLP-1) agonists, such as exenatide and liraglutide, and others.
Closed loop/artificial pancreas with glucagon
In order to attenuate hypoglycemia, a second insulin pump was filled with glucagon and added to the CL/AP system. The algorithm was modified so that if low glucose was predicted, the system would not only stop the insulin delivery, but would deliver small doses of glucagon as well. The addition of glucagon to the system offered a better glycemic profile by minimizing the hypoglycemia that occurred in an insulin-only CL/AP system. However, it did not minimize the postprandial hyperglycemia that occurred after meals.8 Newer CL/AP with glucagon studies, using different algorithms, have observed improved glycemic profiles with less hypoglycemia and hyperglycemia in their subjects.9,13 A study comparing CL/AP with and without glucagon in children and adolescents in the outpatient setting (camp) showed no hypoglycemic events with CL/AP with glucagon, while the single-hormone CL/AP did have hypoglycemic episodes.14
Current challenges to using a glucagon-modified CL/AP system include the stability of the glucagon itself and the need to wear multiple infusion devices. The glucagon that is used today is unstable even during the first 24 hours and only retains its bioactive properties for approximately one day following reconstitution,15 which poses a problem in a pump that could otherwise be worn in the same location for two to three days. It is also difficult to find places to keep two infusion devices on the body at all times. A belt has been devised to carry both pumps and the controller; however, it is not very discrete.
Closed loop/artificial pancreas with pramlintide
Two studies have been published using pramlintide with the CL/AP system, with the goal of minimizing postprandial hyperglycemia.14,16 One study evaluated 10 subjects with T1DM (average age 23.2 ± 1.0 years old, baseline HbA1c = 7.3 ± 0.3%, 50% M) and found that giving a 30 μg dose of pramlintide with each meal delayed, but did not significantly decrease peak postprandial glucose. Less prandial insulin was administered, but the finding was not significant. Additionally, the pramlintide did not suppress endogenous glucagon production, as had been hoped. This group concluded that pramlintide was not the best option for a bihormonal CL/AP system.14 The other study had much more encouraging results. Weinzimer and colleagues (2012) used the same dose of pramlintide (30 μg) in eight subjects with T1DM (age 15–28 years old, baseline HbA1c = 7.5 ± 0.7%, 50% M), and found that pramlintide significantly delayed (p<0.0001) and also decreased peak postprandial glucose by an average of 25 mg/dl (1.4 mmol/l) (p=0.006). They also saw significant differences in pre-meal insulin levels (p≤0.05). They concluded that pramlintide was a beneficial addition to the CL/AP system.16
Even though one study had encouraging results, challenges remain to adding pramlintide to the CL/AP system. The results from the two studies were not consistent in the effects of pramlintide on peak postprandial glucose or in amount of mealtime insulin administered. Furthermore, no endogenous glucagon suppression was seen in the subjects. Pramlintide must be injected subcutaneously with every meal, adding hassle to the otherwise fully automated CL/AP system. Additionally, tolerance to pramlintide is believed to develop (unpublished data), which would render the medication less useful in suppressing postprandial hyperglycemia over time.
Closed loop/artificial pancreas with exenatide
In the same study as the one mentioned above, Renukuntla and colleagues (2014) also looked at the ability of exenatide to minimize postprandial hyperglycemia when added to the CL/AP system. While their pramlintide results were not encouraging, this group found that administration of 2.5 μg of exenatide before meals significantly decreased peak postprandial glucose and suppressed endogenous glucagon response (both p<0.03). Less prandial insulin was administered in the exenatide study visit arm, however, it was not statistically significant. This research group concluded that adjuvant therapy with exenatide in the CL/AP setting effectively decreases postprandial hyperglycemia, blunts glucagon response, and may decrease prandial insulin requirements without causing undue hypoglycemia.14
As with pramlintide, one of the challenges of using exenatide with the CL/AP system is the need for a separate subcutaneous injection multiple times per day. Additionally, the dose of exenatide used in this study is not currently available by pen injector. It is half of the smallest dose that can be delivered by the exenatide pen, requiring the user to draw up their exenatide dose out of the pen using an insulin syringe.14
Other multihormonal Studies
Other multi-hormonal CL/AP studies are being conducted, however, the results of these studies have not yet been published. Various researchers are using several classes of medications used for type 2 diabetes in subjects with T1DM in conjunction with the CL/AP system to try to minimize both postprandial hyperglycemia and the ensuing hypoglycemia (information from the American Diabetes Association’s 75th Scientific Sessions, June 5–9, 2015).
Closed loop/artificial pancreas and exercise
Early exercise studies using the CL/AP system still resulted in hypoglycemia.17–19 However, the addition of glucagon to the system,18 and modification of the algorithms may minimize exercise-induced hypoglycemia. Newer studies propose adding heart rate monitoring to further minimize exercise-induced hypoglycemia. One pilot study with 12 subjects, who exercised at a nine out of 10 intensity, found that there was a lower risk of hypoglycemia and a higher percentage of time in the target range of 70–180 mg/dl (3.9–10 mmol/l) when the CL/AP algorithm was enhanced with a heart rate monitor. All findings were non-significant because the study was not powered to find significance, but the results look promising.19
Closed loop/artificial pancreas and dining out
It is one thing for the CL/AP system to work in the artificial hospital environment, and quite another for it to work in more real-life situations. To simulate real-life situations, one group of researchers tested the CL/ AP system (using the MPC algorithm) using restaurant meals, with and without wine. The overnight glycemic profiles of 24 adults with T1DM were analyzed following two different situations: a medium meal and a large meal with wine. This was done in both the open loop (user-controlled) and closed loop (algorithm-controlled) settings. Overnight CL/AP control resulted in tighter glycemic control and less hypoglycemia compared with when the individuals with T1DM tried to manage their own BG overnight. When using open loop control, subjects had 51% of glucose readings in the target range of 70–144 mg/dL (3.9–8.0 mmol/l), 36% above target, and 13% below target. When closed loop insulin delivery was used, 77% of the glucose values were in the target range, 20% were above target, and only 3% were below target.20
Overnight control with closed loop/artificial pancreas
As mentioned previously, research studies using new diabetes technologies often start by looking at situations that are easiest to measure and in which one may possibly see an effect. Similar to when the popularity of insulin pumps started to increase and clinicians were sometimes using them in their patients only overnight,6,7 the CL/AP system is also being tested for its effectiveness overnight in the outpatient setting compared with standard insulin pump therapy. One study looked at the effectiveness of the CL/AP system overnight at a diabetes camp, in 56 subjects ages ten to 18 years old. They found that BG readings were much better with less hypoglycemia when the children were on CL/AP compared with when they were on usual insulin pump therapy.21 Another study used the CL/AP system in 10 adults (average age 46.4 ± 8.5 years) for multiple overnights. They found that their subjects spent more time in the target range of 80–140 mg/dl (4.4–7.8 mmol/l) overnight, awoke with a lower fasting glucose, and had better glycemic control the next day.21
Closed loop/artificial pancreas at home
As the results of CL/AP studies become increasingly positive, studies are being moved into simulated and actual home environments to see if the system remains robust in these settings. A multicenter European study of 13 adults with T1DM (average age = 45 ± 14 years, HbA1c = 7.4 ± 0.9%, 42 hours on CL/AP) kept their subjects in a hotel to simulate the ‘at home’ experience. This way the subjects could be closely monitored and attended to outside of the hospital setting. The authors found that when on the CL/AP system, subjects spent more time in the target glucose range (70–180 mg/dl (3.9–10 mmol/l), 84% versus 62%, p=0.04) and less time in hypoglycemia (1% versus 10%, p=0.01) compared with when they were managing their insulin doses themselves. Based on these findings, they concluded that the CL/AP system is safe and efficacious in an athome setting, and results in more time in the target range and less time with hypoglycemia.22
The first truly outpatient CL/AP studies were published in 2014.13,23 The first one used an insulin-only CL/AP system in 18 adults with T1DM (average age = 46 ± 10 years, baseline HbA1c = 7.4 ± 0.7%, 75% M) and a smartphone controller that contained the algorithm on it. They found that their subjects spent less time in hypoglycemia and had less risk of hypoglycemia on CL/AP. The subjects’ glucose values ran slightly higher overall on CL/AP (161 versus 152 mg/dl [8.9 versus 8.4 mmol/l]), which was attributed to the emphasis placed on avoiding hypoglycemia in the study. The researchers concluded that closed loop control via smartphone in the outpatient setting is safe and effective. However, further optimization of the control algorithm is necessary to both minimize hypoglycemia and not compromise overall glycemic control.23
The other groundbreaking CL/AP at home study was the Beacon Hill Study, which used the CL/AP system with glucagon in the home environment. Twenty adults and 32 adolescents wore the CL/AP system for five days in the outpatient setting. They lived in a restricted area in a major metropolitan area in the US during the intervention part of the study and were always accompanied by study personnel wherever they went. They had no restrictions on food or exercise. Alcohol was limited to low or moderate intake. Results were very encouraging. The average BG over the five days of CL/AP wear was 138 mg/dl (7.7 mmol/L), with 6.1% or less of the time spent with a BG <70 mg/dl (3.9 mmol/l). Adolescents experienced slightly more hypoglycemia (6.1%) than did the adults (4.8%). The researchers concluded that the CL/AP system with glucagon allows for better glycemic control with less hypoglycemia in the outpatient setting in both adolescents and adults.13
The Beacon Hill Study was repeated in a summer camp setting to study the CL/AP system in children and adolescents in the most controlled outpatient setting possible. A diabetes summer camp is essentially a controlled real-life clinical laboratory. Phase one of the study put the CL/ AP with glucagon on 32 adolescents and young adults, ages 12–20 years old, for five days. One hundred sixty full days of real-life CL/AP data were collected. The following summer, an additional 4,560 hours of data were collected on 19 children ages six to 11 years old using the CL/AP with glucagon. Study results have not yet been published. However, anecdotal results from the camp newsletters include: “Yes. I trusted it. It works. If you check it, it’s perfect. My blood sugars are perfect,” and “It’s like a vacation from diabetes.”24–26
In the most recent unsupervised overnight CL/AP studies, 16 adolescents with T1DM in one study and 24 adults with T1DM in the other were able to successfully use the CL/AP system at home without the watchful eye of study personnel. An analysis of these studies reported a significant decrease in mean overnight glucose (decrease of 16.2 mg/dl [0.9 mmol/l]) and reduced hypoglycemia, as well as an increased proportion of time spent in the target range (increased by a mean of 18.4% between 00:00 hours and 08:00 hours). In these studies, CL/AP was initiated on 89% of the 978 nights, a decision made by the participants themselves.27–29
Closed loop/artificial pancreas and the patient experience
Although most research has focused on the efficacy and safety of CL/ AP there are a few research studies that have assessed the patient experience and psychological aspects of CL/AP. Understanding the lived experience of CL/AP is key for the successful use of the system. Due to the current need for user input, training, and other factors that influence CL/AP, researchers must understand the psychosocial issues that arise.30
Respondents from a survey study specified that the reasons for not wanting the CL/AP were: size, visibility, and lack of effectiveness, however, about half of the survey participants indicated they would use an overnight CL/AP and indicated a strong desire for a device that would minimize the burden of diabetes and improve quality of life.31 When parents of children with T1DM were surveyed, all parents welcomed the development of CL/ AP and 90% were not worried about their child using the system at night.32 A study investigating the psychosocial impact of the CL/AP at night in adolescents indicated positive themes such as reassurance, confidence, safety, and improved diabetes control, while negative themes included difficulties with calibration, alarms, and size of the device.33 Another study investigated the psychological effect of the CL/AP home system in children and adults with T1DM. This study demonstrated high acceptance both before and increasing acceptance after use, as well as reduced worries of hypoglycemia and high satisfaction after use.34 Further qualitative research must be done to assess the psychosocial implications of the CL/AP.
Future directions
Current results of CL/AP studies are encouraging. However, there are still limitations to the CL/AP systems. Limitations include: the amount of usable subcutaneous tissue area for insulin pump infusion set and glucose sensor insertions, the number of devices that need to be carried, and the start-up/calibration time. Children do not have a lot of usable subcutaneous tissue in which to insert one or two insulin pump sites, one or more glucose sensors, and still have places to be able to rotate the sites to, and insulin pump infusion set failure is one of the biggest issues that affect integrity of the CL/AP.2,28 Current CL/AP systems require the user to carry one or two insulin pumps plus a unit that contains the control algorithm, making the individual look like a bionic person because of all of the hardware on their belt. Additionally, the minimum amount of time required between glucose sensor insertion and first calibration in FDA-approved CGM systems is two hours. Due to the sensor being less accurate on the first day, the integrity of the fully closed-loop system is compromised on the first day after a glucose sensor site is changed, unless multiple sensors are used. Lastly, as discussed in each section, the algorithms being used in the system are still not perfect or able to account for all real-life situations, resulting in hyperglycemia and hypoglycemia.
Future directions include smarter insulin pumps, such as the inclusion of the GlucoSitterTM software (DreaMed Diabetes, Israel) designed by one of the insulin pump companies. GlucoSitter, also called MD-Logic Artificial Pancreas (MDLAP), is an algorithm that is based on limited user input into the CL/AP software.29,30 This is at least three generations away from this company’s currently available insulin pump. However, an overnight version of the GlucoSitter has been approved for use in Europe.21 Faster insulin is being developed, which will more closely match endogenous insulin action. Some researchers and companies are looking at warming the insulin infusion site in order to make the insulin work faster, even before the faster insulin formulations are available.31 CL/AP studies are also in progress, assessing the addition of other hormones and medications to the system. Companies are working on the creation of a more stable glucagon to remove one of the current hassles and complications associated with the glucagon-enhanced CL/AP.13,32 Better algorithms are being developed in an attempt to improve the CL/AP’s performance, as well as CGMs that last longer, are easier to insert, and do not need calibration.35 Others are working on patch-pump systems, which are small and free of external tubing and implanted (internal) CL/AP systems are being researched, instead of the external ones that are currently being used.36 Researchers are also investigating the cyber-security of the CL/AP. Threats to the accuracy of commands and confidentiality of data, as well as unauthorized access to the software and devices, could possibly occur. Creating a cyber-security standard will help increase the safety of, and confidence in, the CL/AP.37
While individuals with T1DM eagerly await the first CL/AP system to be approved for general use, they have their own list of things they would like in the meantime. Insulin pump and CGM users want system improvements such as smaller devices, greater sensor accuracy, and faster insulin. There is a group called #wearenotwaiting that is creating open-source software and devices that communicate with multiple platforms, so individuals with T1DM can basically mix and match diabetes self-management components and create their own system instead of being bound to one company’s products for their diabetes care. Two groups have already created CL/AP systems on their own, as lay users: Do-It-Yourself-Pancreas-System (DIYPS) (www.diyps.org) and the individuals who have now formed the company Bigfoot Biomedical (www.bigfootbiomedical.com).
As mentioned earlier, the JDRF has outlined a six-step pathway for CL/ AP progress and the availability to individuals with T1DM. The steps are as follows:
1. Very Low Glucose Insulin Off Pump (currently exists);
2. Hypoglycemia Minimizer (currently exists and available in Europe and Australia);
3. Hypoglycemia/Hyperglycemia Minimizer;
4. Automated Basal/Hybrid Closed Loop;
5. Fully Automated Insulin Closed Loop; and
6. Fully Automated Multi-Hormone Closed Loop.
They are in the process of changing this pathway, however, as current research and development is progressing much faster than outlined by this initial linear model.33
Additionally, the FDA has provided recommendations for those interested in developing a CL/AP system. The FDA recognized the need to encourage developers and provide them with the guidance required to develop a safe and effective CL/AP system.1
Conclusions
At a diabetes technology meeting in 2002, the then-President and CEO of one of the major insulin pump companies stated that in the next five to 10 years, everyone would be able to have a CL/AP system of their own. While this goal has not yet been reached, CL/AP research has made great advancements towards the attainment of a mechanical cure for T1DM. The CL/AP system improves glycemic control and time that glucose is in target range while minimizing hypoglycemia compared with open loop systems. This is true for any time of day, and for all age groups. The system has been tested in children as young as three years old, with favorable results. Newer systems and algorithms are more efficacious than earlier systems and at-home systems are now available for testing. There are many different algorithms being used with the CL/AP, all of which have their benefits and current findings suggest that a multihormonal CL/AP system is likely the best model for achievement of optimal glycemic control. Faster insulin, more improved sensors, and stable, easy-to-administer adjuvant hormones (e.g. glucagon) are needed in order to make a multi-hormonal CL/AP system work even better. Scientists predict that the first at-home CL/AP systems may be available to the general public as early as 2017.33 However, the actual timeline is usually longer than anticipated. People with diabetes want the solution now, and are working to create their own platforms while they wait. In summary, CL/AP findings are promising and with the continuing progression of research and technological advances, the system may be available for widespread use in the not-so-distant future.







