Type 1 diabetes (T1D) is a lifelong condition that results from autoimmune destruction of insulin-secreting beta cells, resulting in an absence of insulin production. In the US, it is estimated that 29.1 million people have diabetes,1 with T1D accounting for 5–10% of all cases. Furthermore, its incidence is increasing, particularly among children under the age of 15.2 A doubling of new cases of T1D in European children younger than five years has been predicted between 2005 and 2020, with a 70% rise in children younger than 15 years.3 Optimal glycaemic control is essential in people living with T1D; intensive treatment of T1D has been associated with delayed onset and slowed progression of numerous complications, including diabetic retinopathy, nephropathy and neuropathy.4
Management of T1D involves multiple daily injections of insulin or use of an insulin pump, both of which require the user to actively track glucose and calculate the needed insulin dose. There is also a significant time lag between when a dose is administered and when it takes effect. Other therapies, including immunotherapy and islet cell transplantation, have been investigated, but with limited success.5,6 There is therefore a need for an automated system that removes the patient from the loop: a closed-loop system.7 This article discusses the potential clinical benefits of closed-loop systems, with a focus on the Hypoglycaemia-Hyperglycaemia Minimizer System (HHM System; Animas Corporation, West Chester, PA, US).
The Treatment Burden of Living with Type 1 Diabetes
Exogenous insulin therapy is not subject to the usual physiological feedback mechanisms so may induce hypoglycaemia.8 The risk of hypoglycaemia limits the efficacy of insulin therapy; the average patient suffers two episodes of symptomatic hypoglycaemia per week and one severe episode per year.4,8–10 Severe hypoglycaemic episode s often occur during sleep.11 These episodes, termed nocturnal hypoglycaemia, can cause convulsions and coma,12 and can be a rare cause of death in individuals with T1D.13 The fear of hypoglycaemia has been associated with decreased quality of life in children with T1D14 and their parents.15 Fear of hypoglycaemia can also result in avoidance of activities beneficial to health, such as exercise.16
In addition, insulin therapy is associated with poor compliance. In the US, children with T1D often do not meet their treatment goals in terms of glycated haemoglobin (HbA1c) values.17 Several factors contribute to this failure, including difficulty in correctly estimating the amount of carbohydrates in a meal, missed meal boluses and anxiety about hypoglycaemia resulting in under-treatment. Poor glycaemic control may affect cognitive development in children with T1D.18 In addition, short-term variation in glucose levels can have an effect on complications of T1D.19 It is well-known that compliance is difficult to achieve with complicated treatment regimens.
Insulin pump therapy has been associated with improvements in glycaemic control in adults and children with T1D.20 Continuous subcutaneous insulin infusion (CSII) therapy is conducted using a durable or patch pump that delivers insulin continuously from a cartridge reservoir via a subcutaneously inserted cannula. The pump can be programmed to deliver varied basal rates of insulin throughout the day, with additional boluses of insulin delivered via self-administration at meal times. Although all pump models can deliver insulin continuously, a technical evaluation conducted on durable pumps such as the OneTouch® Ping® (Animas Corporation, West Chester, PA, US), Accu-Chek® Combo (Roche Diagnostics, Indianapolis, IN, US) and MiniMed Paradigm® Revel™/ Veo™ (Medtronic, Northridge, CA, US), and one patch pump, such as the Insulet OmniPod, showed significant differences in single-dose accuracy performance. At a 0.5 U/hr basal rate over 20 hours, durable pumps delivering in 3-minute intervals showed better single-dose accuracy than the patch pump delivering in 6-minute intervals. Among the durable pumps, the OneTouch Ping demonstrated significantly better accuracy.
Continuous glucose monitoring (CGM) in conjunction with home blood glucose monitoring can improve glycaemic control21 and reduce hypoglycaemia22 in adult patients with T1D. CGM has also been associated with benefits in quality of life (QoL), which are correlated with satisfaction with device accuracy and usability and trust in one’s ability to use CGM data.23 Widespread adoption of CGM has been constrained by its cost and limited reimbursement in healthcare schemes,24 but a growing body of evidence supports its use,21,25 resulting in expert opinions that reimbursement is justified in certain patient groups.26
In order to be used within a closed-loop system, good continuous glucose sensor performance is crucial. However, continuous glucose sensors have been associated with a failure to detect more than half of hypoglycaemic events as well as giving false alarms of impending hypoglycaemia.27 While standards for accuracy exist (International Organization for Standardization 15197:2013), there is no consensus on a standard method for assessing accuracy. Studies do not always assess CGM across all glycaemic ranges.27,28 A comparison of the two most widely used sensor systems, the G4® platinum (Dexcom, San Diego, CA, US) and the Paradigm Veo Enlite™ (Medtronic, Northridge, CA, US) found that the G4 sensor was significantly more accurate than the Enlite system, and that both were less accurate in the hypoglycaemic range.29 In another study the Dexcom G4 sensor showed greater overall accuracy than the Enlite system both overall and for glucose levels in the hypoglycaemic range. In addition, patient satisfaction was higher using the G4 system than the Enlite.30 A further head-to-head comparison of three CGMs (FreeStyle Navigator, [Abbott Diabetes Care, Alameda, CA, US]; G4 Platinum and Enlite) in adult and paediatric patients with T1D under closed-loop blood glucose control demonstrated that the G4 was the most accurate and precise of the devices studied, followed closely by the Navigator, and both were markedly more accurate and precise than the Enlite sensor.31
Perhaps the most important benefit of CGM is avoidance of hypoglycaemic events. Hypoglycaemia can also be avoided by aiming for a slightly higher glucose target: a set point of 140 mg/dL could significantly reduce the risk of severe hypoglycaemia with an acceptable increase in time spent at higher glucose range.32
However, CGM systems have limitations as they still require considerable patient participation in terms of glucose testing, counting carbohydrates and estimating insulin dosages to be administered. This is feasible, if demanding, during the day, but not at night, when many hypoglycaemic episodes occur. There remains a need for control algorithms that adjusts insulin delivery according to daily food intake and activity.
The Concept of a Closed-loop Delivery System
There remains a need to ‘close the loop’ between the glucose sensor and insulin pump. The aim of such a closed-loop delivery system is to at least partially automate insulin delivery based on CGM so as to obtain improved glucose control. The first closed loop system was developed over 40 years ago. However, this device was used only for academic purposes.33 Around 20 years ago, large bedside systems using intravenous blood sampling and intravenous insulin infusion became available in intensive care settings in Japan, but their use was infrequent.34 Transition of such systems to routine clinical use requires accurate, minimally invasive CGM technology integrated with subcutaneous insulin delivery devices – and physiologically informed algorithms to connect the two.
Recent systems use a control algorithm, i.e. software stored on a pump, smartphone or other device, to regulate the insulin delivery based on real-time glucose levels obtained by the sensor. The development of a closed-loop system has faced many difficulties including accuracy of CGM, the physiological time lags involved in the diffusion of the glucose from the plasma to the interstitial fluid when glucose is changing rapidly and delays in both the absorption of insulin and its onset of action after a subcutaneous injection.7
A number of systems are currently undergoing clinical investigation. A growing body of data indicates that these are safe and feasible in daily living situations and result in improved glycaemic control (see Table 1).35–43
Industry’s first foray into commercially available closed-loop technology was the MiniMed® 530G (Medtronic, Northridge, CA, US). In brief, the algorithm in such a device uses simple, straightforward logic to suspend insulin delivery when a breach of a low threshold is registered by the integrated CGM, with an associated patient notification. The suspension continues until that confirmation is acknowledged, or when two hours have elapsed, whichever comes first.44 Such systems have been shown to be effective in improving glycaemic control and reducing hypoglycaemia45 in patients with T1D. Other clinical development studies sponsored by Medtronic are in progress, investigating more sophisticated algorithms.46 The predictive low glucose suspend system represents an advance from the glucose suspend device, and halts insulin delivery overnight with the aim of preventing nocturnal hypoglycaemia.47–49 Such systems have also been evaluated for use during exercise, with positive results.50
Studies have found that attitudes towards closed-loop systems are positive among patients51 and caregivers of children with T1D.52 Nighttime blood glucose control is the biggest concern for parents and a small study (n=19) indicated that 90% of parents trusted an algorithm to control overnight insulin delivery.52
The Hypoglycaemia–Hyperglycaemia Minimizer System
In 2006, the Juvenile Diabetes Research Foundation (JDRF) launched the artificial pancreas programme, which involved direct funding and collaborations with academic institutions, research centres and industry in the US and Europe. Many leading diabetes device manufacturers have participated in this project, developing CGM systems and pumps with enhanced capability for closed-loop use.

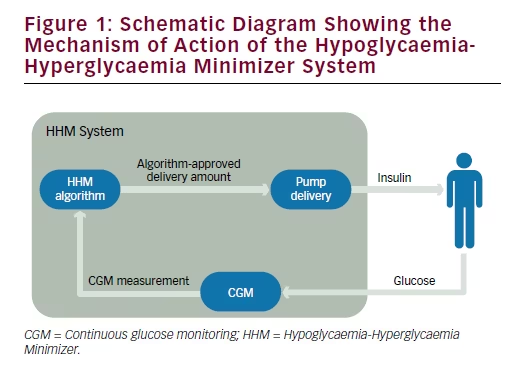
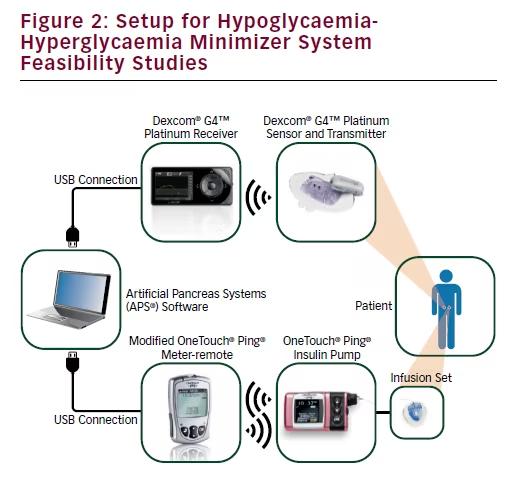
The HHM System has been developed by Animas Corporation in collaboration with the JDRF, and is based on a closed-loop technology that it is designed to predict potential hypo- or hyperglycaemic excursions, and proactively decrease or increase (respectively) insulin infusion in order to mitigate, if not avoid, the excursions. The System is illustrated in Figures 1 and 2, and comprises a CGM (Dexcom, Inc., San Diego, CA, US), insulin pump (Animas Corporation, West Chester, PA, US), and control algorithm. The main component of the HHM System is the control algorithm, which calculates the optimal amount of insulin to deliver to the patient based on CGM trends, previously infused insulin and model predictions. The algorithm determines how much insulin is needed to result in the optimal glucose trajectory for the near future, and transmits this information to the insulin pump for delivery. This process is repeated every f minutes, each time a new CGM datum is received.
Feasibility Studies Investigating the Hypoglycaemia-Hyperglycaemia Minimizer System
Three clinical research centre (CRC)-based studies have investigated the feasibility of the HHM System. One of these studies was conducted in 13 adults with T1D, who underwent closed-loop control lasting approximately 20 hours, including an overnight period and two meals. The predictive HHM System decreased insulin infusion rates below the participants’ preset basal rates ahead of excursions below the prespecified target zone (CGM <90 mg/dL), and delivered 80.4% less basal insulin during such excursions. Similarly, the HHM System increased infusion rates when a breach of the upper threshold (CGM >140 mg/dL) was predicted, and delivered 39.9% more insulin than basal during these excursions. Subjects spent a mean ± standard deviation (SD) of 0.2±0.5% of the study at glucose levels <70 mg/dL, including 0.3±0.9% during the overnight period. The mean glucose level across the entire study period was 164.5±23.5 mg/dL. The HHM was able to administer insulin safely and, on nine occasions, recommended the administration of carbohydrate in the form of which was 15 g of carbohydrate as juice or glucose tablets. During the study, there were no instances of severe hypoglycaemia or diabetic ketoacidosis.53
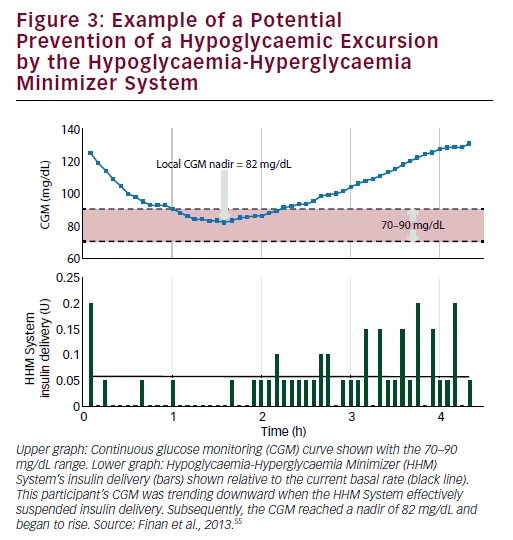
A second feasibility study was conducted in 20 adults with T1D to assess the predictive capability of the HHM System in terms of mitigating hypoglycaemic excursions, defined as a breach of the 70 mg/dL threshold, by CGM. First, the potential of the system to prevent hypoglycaemic excursions was investigated by quantifying the frequency of times that the glucose level assessed by CGM reached a local nadir of between 70–90 mg/dL (see Figure 3), indicating that it was potentially due to the algorithm’s hypoglycaemia-mitigating, insulin-reducing action that the CGM began to rise following its nadir. In order to isolate the effects of the algorithm, the glucose-increasing effects of carbohydrate meals were excluded in the calculation of this metric, by ignoring data received during the hour following each meal. Second, the capability to proactively mitigate hypoglycaemic excursions was quantified as the average lead time, before a breach of the CGM 70 mg/dL threshold, that the algorithm reduced or suspended insulin delivery. In addition, the amount of insulin withheld during this period was assessed. Thirdly, the ability of the algorithm to warn of imminent hypoglycaemic events was quantified by the average lead time, before the breach, that the alarm was triggered. Results showed that a CGM nadir between 70 and 90 occurred an average of 1.75 times per subject per day. On average, over 24 h, subjects spent 80–90% the time within the normal glycaemic range. Additionally, fewer than half of the subjects had blood glucose values less than 70 mg/dL during the overnight period. It must be noted, however, in this small feasibility study, that it cannot be known with what frequency the actions of the algorithm were directly responsible for avoiding hypoglycaemic events. The algorithm reduced insulin delivery for an average of 39 minutes prior to breaches of the CGM 70 mg/dL threshold, accounting for an average of 0.5 U of insulin not delivered to the participant, relative to their corresponding basal rates. The algorithm delivered warnings of imminent hypoglycaemia on average 7.1 minutes before such
breaches. The investigators concluded that the HHM System reduced insulin delivery and triggered warnings before the CGM breached the low glucose threshold.
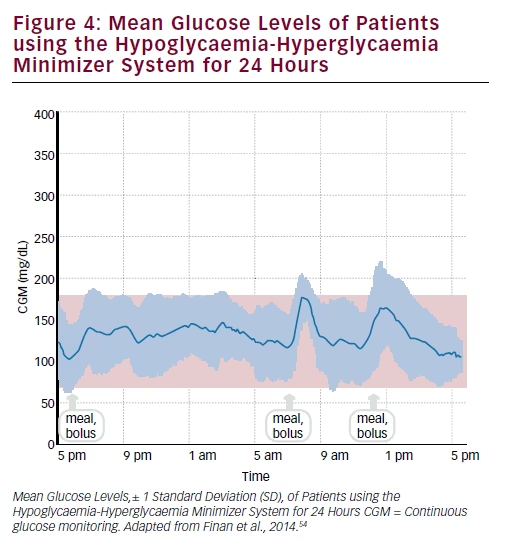
This study also evaluated the ‘aggressiveness factor’, which refers to the speed and magnitude at which the control algorithm adjusts insulin infusion in response to changing CGM measurements (see Figure 4). Enrolment criteria were current use of an insulin infusion pump with rapid-acting insulin, and a HbA1c level <10%. Participants were assigned to conservative, medium and aggressive values. Time spent within the normal glycaemic range was highest using the aggressive setting.54,55
In a third study, 12 adults with T1D were studied for approximately 24 hours. The purpose of this study was to isolate the hypoglycaemia minimisation aspect of the HHM System. As the aggressiveness setting was increased from ‘conservative’ to ‘medium’ to ‘aggressive’, the controller recommended less insulin (-3.3% versus -14.4% versus -19.5% relative to basal) with a higher frequency (5.3% versus 14.4% versus 20.3%) during the critical times when the CGM reading was decreasing and in the range 90–120 mg/dL. The most aggressive setting resulted in the least time spent at low blood glucose levels (<70 mg/dL) and the most time spent within the normal glycaemic range (70–180 mg/dL), particularly in the overnight period. Hyperglycaemia,
diabetic ketoacidosis or severe hypoglycaemia were not observed at any of the aggressiveness values.56
Discussion
T1D is a chronic condition that can currently only be well managed with constant vigilance, placing a huge burden on patients with T1D and their families. It is hoped that the development of closed-loop systems may result in significant improvements to the quality of life of patients with T1D.
While current closed-loop technology for artificial pancreas applications has matured to the point of commercial viability, there remain a number of scientific and technological limitations in such systems. Such limitations include the pharmacodynamic and pharmacokinetic delays associated with subcutaneous insulin absorption and action; the infeasibility of automatically measuring carbohydrate intake; the disparate patterns of carbohydrate absorption, which varies based on a number of factors such as meal composition – and therefore the inability to accurately model the effects of meals on blood glucose; and the unavailability or nascence of biometric sensors that may inform the control algorithm.
At least some of these barriers to optimal diabetes control may soon be broken down by developments in tangential scientific fields. For
example, the formulations of faster-acting insulins may open the door to fully automated meal-time control; continued improvements in the accuracy and reliability of CGM systems may alleviate the burden, at least in part, of blood glucose self-monitoring; the development and commercialisation of med-tech devices able to measure biometrics or surrogates thereof – such as physiological stress and energy expenditure – may help realise a more holistic, robust control algorithm. Future algorithms and systems have a great deal of opportunity in the years ahead to further improve the lives of people with T1D.
The HHM System is a predictive system designed to mitigate, if not avoid completely, hypo- and hyperglycaemic excursions. The superior accuracy and reliability of its CGM sensor and insulin pump are well established. The HHM System has shown promising results in feasibility studies, enabling patients to stay within the designated normal glucose range for up to 90% of the time during a 24-h period. The authors acknowledge the (unavoidable) caveats of these CRC-based studies, namely, the artificial and sedentary environment in which the studies were performed. Nonetheless, the system demonstrated, through its controller actions, the potential to minimise, if not avert entirely, some hypo- and hyperglycaemic excursions. A pivotal study is planned in a larger patient population in ambulatory (‘free-living’) conditions that will investigate the safety and efficacy of the system, including the final controller configuration and human factors considerations.







