Disorders related to bone health are commonly seen in clinical practice in India. The most common conditions are vitamin D deficiency-related disorders (rickets, osteomalacia), followed by osteoporosis, hyperparathyroidism, endocrine osteopathies, and impaired bone health associated with systemic diseases.1–3 Vitamin D deficiency prevalence in India ranges from 70–90% of the population.3,4 Bone disorders are managed by doctors from different specialties including general practitioners, physicians, orthopedic surgeons, and endocrinologists among others. A recent paper on the evaluation of a clinical practice scenario of osteoporosis management among orthopedicians revealed significant heterogeneity in diagnostic and treatment approaches.5 However, data on knowledge, awareness, and practices on managing bone health disorders among physicians in India is not available. Hence the aim of this study is to determine the preference and practices among doctors in India with regards to managing bone health disorders among doctors in India, focusing on vitamin D deficiency disorders and osteoporosis management.
Methods
The Society for Promotion of Education in Endocrinology and Diabetes conducted a 1-day continuing medical education (CME) program on April 16 2016, which was attended by 394 doctors from different specialties including family and general physicians, obstetricians, pediatricians, surgeons, and endocrinologists. At the beginning of the CME programme, all the doctors were given a questionnaire consisting of 38 multiple-choice questions with the scope of marking more than one choice as the preferred answer. A time of 20 minutes was allocated for filling the questionnaire based on the pilot study carried out on 20 doctors prior to the event. Questions in the questionnaire were primarily based on evaluating the doctor’s preferred choices with regards to the diagnosis and management of vitamin D deficiency and osteoporosis in their clinical practice.
Questionnaire
Section one of the questionnaire consisted of 16 questions evaluating the doctors’ perception of the burden of vitamin D deficiency in India, the frequency of testing for serum vitamin D in clinical practice, preferred route, formulation, dosage, and regimen of vitamin D supplementation. Questions also assessed the doctor’s preference in the preparation, dosage, and fixed drug combinations with regards to calcium and vitamin D supplementation. Three questions checked the doctors’ perception on the occurrence of hypervitaminosis D/vitamin D toxicity. Section two consisted of 19 questions, which probed the doctors’ clinical practices on osteoporosis management. Queries enquired into the burden of osteoporosis seen in clinical practice, the preferred investigations (biochemical and radiologic) for osteoporosis diagnosis, interpretation of dual energy X-ray absorptiometry (DXA) in osteoporosis assessment, secondary osteoporosis, preferred pharmacologic agents for osteoporosis management, treatment plans, regimen and duration, and assessment of treatment response on follow-up. The last three questions aimed to understand the demography of the participants. The detailed questionnaire is available with the ePub article as an appendix online.
Study participants
Study participants were a diverse group of practicing doctors ranging from family physicians/general practitioners, obstetricians, internal medicine specialists, and endocrinologists practicing in New Delhi, who voluntarily chose to attend the CME. The venue, timing, and the program content of the CME were advertised in the local newspapers, radio, and information bulletin of all major hospitals of Delhi for 1 month prior to the actual event. There were no registration fees for the event, and any doctor was free to attend.
Statistical analysis
Summary statistics were prepared for responses to each question. Because not every participant answered all questions, the percentage of respondents providing a given answer was calculated individually for each question, using the number of respondents to that question as the denominator. Analysis was done using the IBM SPSS Statistics version 20.0 software to compare the treatment preferences among doctors of different specialties.
Results
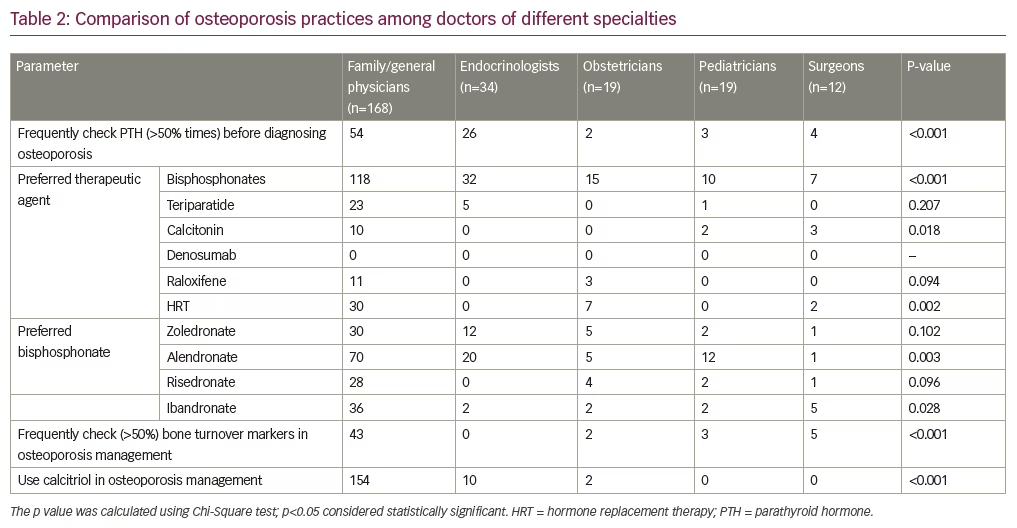
A total of 394 doctors attended the meeting; a total of 316 doctors gave consent and participated in this questionnaire-based study, of which 279 doctors returned the questionnaires. Twenty-three questionnaires with less than 70% of the questions answered were excluded from the analysis, therefore responses from 256 questionnaires were analyzed. A majority of the attending doctors were family/general physicians (n=168), followed by obstetricians (n=19), pediatricians (n=19), surgeons (n=12), endocrinologists (n=34), and others (n=4). Family/general physicians were doctors with a Bachelor of Medicine and Bachelor of Surgery (MBBS) degree, with or without a Doctor of Medicine (MD) in internal medicine. Obstetricians were doctors with a MBBS and a Masters of Surgery (MS) degree in obstetrics and gynecology. Pediatricians were doctors with a MBBS and a MD in pediatrics. Surgeons were doctors with a MBBS and a MS degree in general surgery. Endocrinologists were doctors with a MBBS, a MD in internal medicine, and a Doctorate of Medicine (DM) in endocrinology. The median age (25th to 75th percentile) of the study participants was 44 (range 34–58) years, with 195 being males (76.18%). Comparisons of vitamin and calcium supplementation practices and osteoporosis management practices among the doctors of different specialties have been elaborated in Table 1 and Table 2, respectively.
Section one
When asked about the perception of the prevalence of vitamin D deficiency in India, a majority of the doctors (90.63%; 232/256) believed there was >70% prevalence of vitamin D deficiency in Indians. Only four doctors (1.56%) had the perception that the prevalence of vitamin D deficiency in Indians is <30%. Only 28 doctors (10.94%) routinely (>90% times) checked serum 25-hydoxy-vitamin-D (25-OH-vitamin-D) levels before supplementation. A majority of the doctors (57.43%) were infrequent (0–25% times) in checking serum 25O HD levels before vitamin D supplementation, and believed in empirical vitamin D supplementation based on the symptoms. Of these, there were 48 doctors (18.75%) who never suggested serum 25O HD testing before vitamin D supplementation. Serum vitamin D testing was significantly more common among physicians and pediatricians (Table 1).
In clinical practice, six doctors (2.34%) routinely used intramuscular vitamin D for the treatment of vitamin D deficiency (>50% of all patients treated). Eleven doctors infrequently used intramuscular vitamin D injections for managing vitamin D deficiency (25–50% of all patients treated). One hundred and fifty-three doctors (59.77%) had less than a quarter of patients on parental vitamin D supplementation, and 86 doctors (33.59%) exclusively prescribed oral vitamin D for treating vitamin D deficiency. Endocrinologists had a significantly higher use of oral vitamin D supplementation as compared to doctors of other specialties (Table 1). The most preferred dose for vitamin D supplementation was 60,000 international units (IU) (79.69%; n=204), followed by 600,000 units (16.80%; n=43). Only nine doctors (3.52%) preferred other dose formulations of vitamin D (e.g. 2000 and 1000).
Among doctors using 600,000 IU intramuscular injection for vitamin D deficiency, the most preferred regimen was a single intramuscular injection (39.00%). Other regimens used with 6 lakh IU intramuscular injection included weekly injections for 1 month (26.17%), monthly injections for 6 months (19.14%), and once-6-monthly injections (14.45%). Three doctors preferred using daily injections of 6 lakh IU of vitamin D for 1 week for treating vitamin D deficiency. Among doctors using 60,000 IU of oral vitamin D supplementation, the most preferred regimen was once-weekly for 2 months (72.66%). Other regimens included weekly 60,000 IU for 1 month (15.62%), daily 60,000 U for 1 week (5.08%), monthly 60,000 IU for 1 year (3.91%), and dose once every two weeks of 60,000 IU for 6 months (2.73%). Among the different oral vitamin D formulations available in India, the most preferred by doctors were sachets and capsules (41.4% each), followed by tablets (14.84%), and liquid preparations (2.34%).
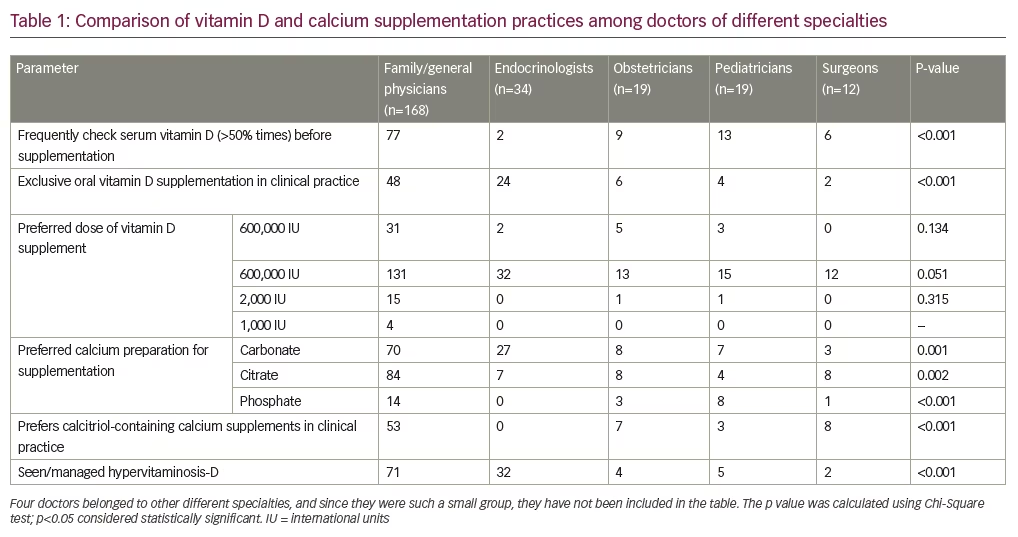
For calcium supplementation, the most preferred formulation was calcium carbonate (45.70%; n=117), followed by calcium citrate (41.80%; n=107) ,and calcium phosphate (9.77%; n=25) among others. Endocrinologists had a significantly higher use of the calcium carbonate preparations compared to doctors of other specialties (Table 1). One hundred and fifty-seven doctors (61.33%) felt that a fixed dose combination of calcium with vitamin D was useful in clinical practice, whereas 99 doctors (38.67%) felt it was not useful. The most preferred calcium and vitamin D fixed dose combination was calcium with calcitriol (0.25 mcg/tablet [28.12%; n=72]) followed by calcium with 25-OH-vitamin-D (500 IU/tablet [23.83%; n=61]), and calcium with 25-OH-vitamin-D (250 IU/tablet [17.19%; n=44]). Physicians had a significantly higher preference for calcitriol-containing calcium supplements in clinical practice, as compared to doctors of other specialties (Table 1). One hundred and thirty (50.78%) and 169 (66.02%) doctors correctly defined the serum 25-OH-vitamin-D cut offs used in clinical practice for defining hypervitaminosis-D (>100 ng/ml), and vitamin D intoxication (>150 ng/ml) respectively. One hundred and thirteen doctors (44.14%) admitted that they had seen, and managed, patients with hypervitaminosis-D in their clinical practice. Endocrinologists most frequently managed hypervitaminosis-D, followed by physicians (Table 1).
Section two
Of the doctors questioned, 29.69% (n=76) reported that they manage, on average, 5–10 patients with osteoporosis in a month of clinical practice, and 22.27% (n=57), 21.88% (n=56), and 20.70% (n=53) of doctors reported that they managed less than five patients, 10–20 patients, and more than 20 patients with osteoporosis per month, respectively, in their clinical practice. Back aches and bone pains (72.66%; n=186) were the most common osteoporosis symptoms noted. Seventy-one doctors (27.73%) reported to have diagnosed asymptomatic osteoporosis based on bone mineral density (BMD) screening. Fracture and height loss were observed by 34 (13.28%) and 17 (6.64%) doctors, respectively, in their patients with osteoporosis.
During calcium metabolism work-up and as a part of diagnostic evaluation for osteoporosis, 43 doctors (16.80%) routinely carried out parathyroid hormone testing (100% of their patients). Forty-five doctors carried out parathyroid hormone testing in a majority of their patients (50–90% of all patients evaluated). Forty-six doctors went for infrequent parathyroid hormone testing as a part of osteoporosis work-up (25–50% of all patients evaluated). One hundred and twenty-three doctors (48.05%) infrequently tested for parathyroid hormone in their patients (<25% of patients) as a part of osteoporosis evaluation. Parathyroid hormone testing was significantly more common among endocrinologists as compared to doctors from other specialties (Table 2). Hormone replacement therapy (HRT) and raloxefene were almost exclusively used by obstetricians and physicians for managing osteoporosis (Table 2).
Two hundred and forty-one doctors (94.14%) routinely used DXA for diagnosis and follow-up of patients with osteoporosis. Lumbar spine and hip was the most preferred site for BMD assessment during screening for osteoporosis. Wrist BMD screening was carried out by 20 doctors (7.81%) in their clinical practice. One hundred and nine (42.58%), 74 (28.91%), and 73 (28.52%) doctors reported that postmenopausal osteoporosis is best diagnosed using T-score, Z-score, and absolute BMD (g/cm2) respectively. Sixty-two (24.22%), 123 (48.05%), and 71 (27.73%) doctors reported that secondary osteoporosis is best diagnosed using T-score, Z-score, and absolute BMD (g/cm2), respectively. When asked about the causes of secondary osteoporosis seen in clinical practice, the most commonly observed etiology was glucocorticoid-induced osteoporosis (52.73%; n=135), followed by hyperthyroidism (17.58%; n=45), primary hyperparathyroidism (8.98%; n=23), and rheumatoid arthritis (8.59%; n=22).
The preferred agents for managing post-menopausal osteoporosis were bisphosphonates (71.09%; n=182), followed by teriparatide (11.33%; n=29), HRT (15.23%; n=39), calcitonin (5.86%; n=15), and raloxifene (5.47%; n=14). Endocrinologists almost exclusively used bisphosphonates for managing osteoporosis, as compared to doctors from other specialties (Table 2).
A majority of the doctors preferred using oral over intravenous bisphosphonates (69.14%; n=177). Among oral bisphosphonates, doctors felt that a once-a-month regimen was most preferred by patients (42.97%; n=110), followed by once-a-week bisphosphonates (39.84%; n=102). Only 10 doctors (3.91%) used a daily bisphosphonate treatment regimen. Alendronate was the most commonly used bisphosphonate in clinical practice (41.02%; n=105), followed by zoledronate (24.61%; n=63), ibandronate (17.97%; n=46), risedronate (13.67%; n=35), and pamidronate (2.73%; n=7). The median duration of bisphosphonate use by the study doctors for managing osteoporosis was 11 months (6–30). Seventy (27.34%) and 126 doctors (49.22%) used calcitriol for the treatment of osteoporosis in all and some of their patients, respectively. Only 60 doctors (23.44%) never used calcitriol as part of a treatment regimen for managing osteoporosis. The use of calcitriol for managing osteoporosis was significantly more common among family/general physicians compared to doctors of other specialties (Table 2).
For assessing the response to therapy, most doctors preferred doing a repeat BMD study at 12 months (49.61%; n=127), followed up at 6 months (32.42%; n=83) and at 18 months (7.03%; n=26). One hundred and twenty-two doctors (47.66%) felt T-score/Z-score can be used for assessing a response to osteoporosis treatment during follow-up. One hundred and one doctors (39.45%) felt that osteoporosis treatment can be called a success only if there is an improvement in BMD on follow-up as compared to baseline. Only 33 doctors (12.89%) correctly defined the measure for success of treatment of osteoporosis, i.e., absolute BMD not lower on follow-up as compared to baseline.
Discussion
This study highlights several important facets with regards to the diagnosis and management of vitamin D deficiency and osteoporosis by doctors in India. There is a good awareness among doctors about the high prevalence of vitamin D deficiency in Indians. This may explain why a majority of the doctors preferred empirical vitamin D supplementation in their patients, without confirming the diagnosis by testing 25-OH-vitamin-D. Only 10.93% of doctors routinely tested for serum 25-OH-vitamin-D levels before vitamin D supplementation. Another factor may be the cost associated with serum 25-OH-vitamin-D testing in India, which ranges from 800–2,500 rupees in different centers across the country. In contrast, the cost of a 60,000–600,000 IU of vitamin D preparation in India ranges from 20–50 rupees.
The use of intramuscular vitamin D injections is not uncommon in India. Only 33.4% of doctors were exclusively using oral vitamin D supplementation for correcting vitamin D deficiency. Among the doctors of different specialties, endocrinologists used intramuscular vitamin D injections the least, and favored the use of oral cholecalciferol. In 17% of doctors, 600,000 IU of vitamin D preparation was the preferred dose for treatment, while 49.31% of doctors using 6 lakh IU intramuscular vitamin D injections for treatment preferred a regimen of weekly injection for 1 month, or monthly injection for 6 months, making a cumulative vitamin D dose of 24–36 lakh IU. A few previous reports have also suggested that the use of high-dose intramuscular vitamin D (600,000 IU), often given as multiple doses in a short span of days to weeks, is common in India.6,7 The advantage of high-dose intramuscular vitamin D is the associated low costs, and it avoids treatment non-compliance that is likely with daily or weekly oral cholecalciferol supplementation.
It must, however, be highlighted that the use of even a single annual high oral dose of vitamin D (>500,000 IU) has been consistently linked with hypercalcemia, hypercalciuria, renal failure, falls, and fractures in different studies.8,9 Even lower doses of vitamin D (300,000 IU) given intramuscularly intermittently for 3 years in the elderly population was associated with increased fractures.10 In this context, the previous reports of 16 patients with vitamin D intoxication from Gurgaon, Haryana, India, and 10 patients from Kashmir, India, where the median cumulative dose of vitamin D received was 3,600,000 IU (2,220,000–210,000,000) over periods of 1–4 months, either in the form of multiple parenteral injections or weekly oral sachets, highlights the severity of vitamin D over-dosage and intoxication.11,12 There has also been a report of vitamin D intoxication related to vitamin D addiction.13 We have previously reported the rapid increase in the occurrence of hypervitaminosis-D at a tertiary care institute in northern India (from 1.48% in 2011 to 7.82% in 2016; of all blood samples tested for serum 25-OH-vitamin-D).6 This concerning trend has been reconfirmed in this study, where 44.14% of all doctors evaluated admitted that they had seen and managed patients with hypervitaminosis D.
It is also a matter of concern that, although there is a high awareness among doctors on the burden of vitamin D deficiency in India, only half of the doctors could correctly define the serum 25-OH-vitamin-D cut-offs for diagnosing hypervitaminosis D and vitamin D intoxication. This study highlights that there is an urgent need to spread awareness among the doctors about vitamin D intoxication, and the dangers of unmonitored vitamin D supplementation using high supraphysiologic doses of vitamin D, especially parental, over short periods of time. The pleotrophic benefits of vitamin D in different disease states have been highlighted in the past decade (beneficial effects of glycemia, coronary artery disease, anticancer properties, immune-activation in infective states, beneficial effects in autoimmune disorders, neurologic disorders).14–18 However, it must be highlighted that vitamin D should not be considered as a panacea against all illness, and rampant unmonitored use should be discouraged.
Another concerning trend observed in this study was the high preference of use for calcium with calcitriol fixed dose combination in routine clinical practice. Calcium with calcitriol (0.25 mcg/tablet) was the preferred calcium supplement of choice for 28.12% of doctors in this study. This was especially common among the physicians. Both forms of activated vitamin D, i.e., alfacalcidol and calcitriol (but not cholecalciferol), have been consistently linked with increased hypercalcemia, nephrocalcinosis, and nephrolithiasis.19 It needs to be highlighted among doctors that calcitriol is an activated form of vitamin D which cannot be regulated by the body. In contrast, cholecalciferol is a precursor of activated vitamin D, which can be converted to the active form as per the requirements of the body, provided the person has normally functioning liver and kidneys. It is only cholecalciferol, and not alfacalcidol or calcitriol, which has been linked to reduced mortality in the elderly,20 hence it is the calcium with cholecalciferol combination that is safer to use in routine clinical practice. Calcitriol-based combination use should be restricted to patients with any condition where the 1-alpha-hydroxylase enzyme activity is reduced or the endogenous production of active vitamin D is reduced. These include chronic renal failure, phosphatonin-mediated disorders such as oncogenic osteomalacia, hypophosphatemic osteomalacia, and vitamin D-resistant rickets, among others.
Our study highlights that osteoporosis is commonly seen in clinical practice in India. Nearly one-third (27.73%) of the patients with osteoporosis diagnosed in clinical practice were asymptomatic and were detected only on routine BMD screening. Fracture and height loss were observed by 13.28% and 6.64% of doctors respectively, highlighting that the diagnosis may be delayed in nearly one out of five patients diagnosed. Only 16.79% of doctors were routinely testing for parathyroid hormone levels as a part of metabolic work-up for osteoporosis. Endocrinologists had a significantly higher clinical practice of checking serum parathyroid hormone levels as a part of metabolic work-up for osteoporosis. Hence it is likely that a significant number of patients with primary hyperparathyroidism are being missed, especially those with milder disease and normocalcemic primary hyperparathyroidism. However, it must be highlighted that the high costs associated with parathyroid hormone testing, lack of availability in all diagnostic centers, and the challenges associated with sample collection and processing (cold chain maintenance) limits the use of parathyroid hormone testing in routine clinical practice.
Less than half of the evaluated doctors could correctly define the DXA criteria for the diagnosis of post-menopausal and secondary osteoporosis. Also, there was a lack of consensus regarding when to repeat BMD testing on follow-up in patients on osteoporosis treatment, with nearly one-third of doctors (32.42%) preferring to repeat DXA at 6 months of follow-up. Current guidelines have suggested that repeat BMD testing should be done not before 1–2 years of follow-up to detect meaningful changes in BMD.21 BMD changes occur early in cancellous bone-like lumbar spine and are much slower in cortical bone-like greater trochanter.21 Only 12.89% correctly defined the measure for success of treatment of osteoporosis, i.e., absolute BMD not lower on follow-up as compared to baseline. This highlights that there is a considerable gap in knowledge, awareness, and practice in the diagnosis and management of osteoporosis in clinical practice. There is an urgent need to spread awareness regarding the correct and optimal use of DXA in the diagnosis and management of osteoporosis. In addition, awareness needs to be spread with regards to DXA machine maintenance and ensuring quality control over repeated BMD measurements. Bisphosphonates were the preferred agents for managing osteoporosis, with a large majority of doctors preferring oral bisphosphonates with a weekly or monthly intake regimen. Aledronate followed by zoledronate were the two most commonly used bisphosphonates in clinical practice. While endocrinologists almost exclusively used bisphosphonates to manage osteoporosis, the use of HRT and raloxifene was primarily restricted to obstetricians and gynecologists.
Calcitonin was very rarely used by doctors for managing osteoporosis, which is in accordance with international norms. Since 2012, the European Medicines Agency has strictly restricted the use of calcitonin in clinical practice, due to the associated small increased risk of cancer. Calcitonin is no longer recommended as a treatment for osteoporosis. Calcitonin use should be restricted to the shortest possible period of time in patients with refractory Paget’s disease, acute bone loss due to sudden immobilization, and hypercalcemia caused by cancer.22
To conclude, it may be said that there is an urgent need to spread awareness about vitamin D intoxication, the dangers of unmonitored vitamin D supplementation, and the dangers associated with calcitriol-based calcium supplements in clinical practice. Testing for 25-OH-vitamin-D is considered to be expensive in many clinical situations, and the prevalence of both rickets and osteomalacia is significant in India—a consequence of vitamin D deficiency compounded by low dietary calcium intake. Therefore, good clinical practice would be to advise combined calcium/cholecalciferol supplementation, with doses of the latter not exceeding the daily intake requirement in the case of unmonitored vitamin D supplementation (recommended daily allowance that meets the needs of 97.5% of the population of otherwise healthy adults being 800 IU daily). There is a need to conduct more independent CME programs among doctors promoting the judicious use of calcium and vitamin D preparations. The low use of parathyroid hormone testing in osteoporosis evaluation can lead to missing patients with primary hyperparathyroidism, especially normocalcemic primary hyperparathyroidism. There is a considerable gap in knowledge, awareness, and practice for osteoporosis. There is an urgent need to spread awareness regarding the correct and optimal use of DXA in osteoporosis management. Patient-centered bone care is the need of the hour.