Thyroid nodules and thyroid cancers are more common in children than has been previously appreciated, and represent a significant health issue. Older studies used palpation to identify thyroid nodules and reported a childhood prevalence of 2%. However, high-resolution ultrasound identifies thyroid abnormalities in 18% of children.1 Ultrasound also detects thyroid nodules in 13–31.5% of children with autoimmune thyroiditis (AIT).2,3 Once a thyroid nodule is identified, the overall risk of malignancy is higher for children (22%) than for adults (5–10%).4 Ultrasound has been used routinely for more than a decade, and during that time the incidence of differentiated thyroid cancer (DTC) has been increasing among those of all ages, including children. The most recent data from the Surveillance, Epidemiology, and End Results (SEER) Program report an annual incidence for adolescents of approximately 25 cases/million/year.5 These data suggest that approximately 400,000 children and adolescents in the US have a solid thyroid nodule and 100,000 have DTC.2
For decades, treatment of children with DTC was based on adult treatment guidelines.6 The majority of children with DTC already have lymph node involvement at the time of diagnosis (80%) and many (20%) have pulmonary metastases. Based on this metastatic profile, children were routinely treated with total thyroidectomy (TT), lymph node dissection (LND), and radioactive iodine (RAI) ablation. Although this therapy was highly successful in treating disease (disease-specific mortality was only 2%), it was associated with a high rate of complications and an increased risk for secondary malignancies.7 These second malignancies resulted in excess overall mortality (60% overall survival at age 60 years versus 75% overall survival for controls) and have led us to reconsider the universal prescription of RAI for children with low-risk DTC.7
Previous treatment was also designed to completely eradicate disease. Children with persistent disease were administered RAI on a scheduled annual basis until the diagnostic whole-body RAI scan (dxWBS) showed no RAI uptake and the serum thyroglobulin (Tg) was undetectable. However, 30–45% of children with pulmonary metastases do not become free from disease after multiple treatments, but develop stable-persistent disease.8 For them, 10-year survival remains excellent (100%) and the majority do not progress (five-year progression-free survival [PFS] was 65–70%). Important data from Biko, et al.9 have also shown that total body disease burden (as reflected by serum Tg levels) may continue to decline for many years after RAI therapy.9 They followed 20 children who were diagnosed with papillary thyroid cancer (PTC) and extensive pulmonary metastases. Patients received a mean of 5.5 therapeutic courses of RAI, accumulating 24.2 GBq (654 mCi). During that time, serum Tg declined as expected. However, RAI therapy was prematurely discontinued even though the patients had a large persistent tumor burden (mean serum Tg level approximately 100 μg/L). Despite the lack of continued treatment, the serum Tg level continued to decline and became undetectable in many.
For these reasons, a more appropriate risk-stratified treatment and rational end-point for therapy were needed for children. A similar strategy has been successfully employed for adults.10,11 Ideally, children with extensive lymph node involvement and pulmonary metastases would still undergo TT plus LND and RAI, but children with less extensive disease (i.e. disease confined to the gland) would receive less extensive therapy. All children would have an assessment of the individual treatment response and more appropriate end-points. Such
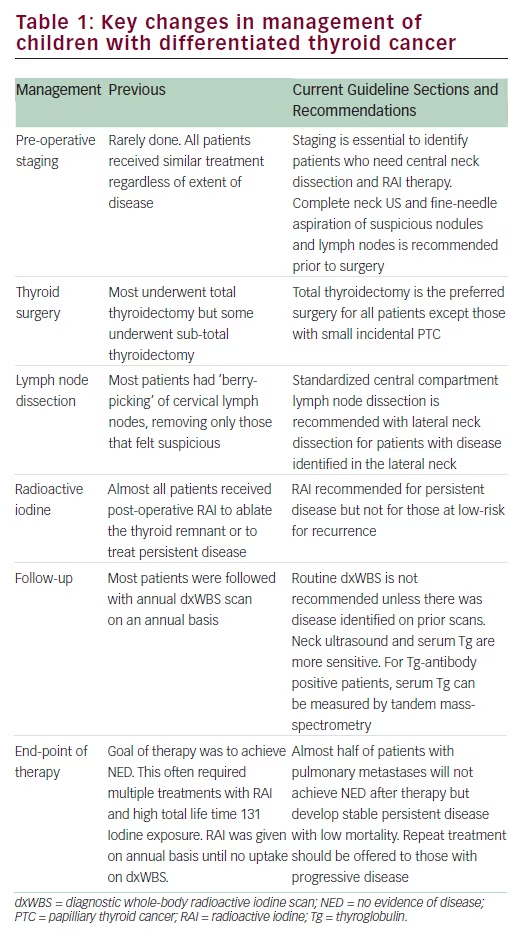
individualized treatment would thereby limit the risk for complications and second malignancies. To address this need, The American Thyroid Association (ATA) empaneled an international, multidisciplinary task force to develop treatment guidelines for children and adolescents with thyroid nodules and cancers. This task force reviewed the available literature and developed evidence-based guidelines that have been recently published.12 Key changes in the management of children with DTC are highlighted in Table 1.
This risk-stratified approach generates several questions that must be addressed for each individual patient.
• Who needs TT, LND and RAI, and who can do just as well with less?
• What is a rational end-point for therapy, i.e. do we need all patients to have no evidence for disease in order to have excellent survival?
• How can we tell if therapy is working and how long will it work, i.e. do we need annual RAI therapy or should we wait until all effects from prior treatments have occurred?
Materials and methods
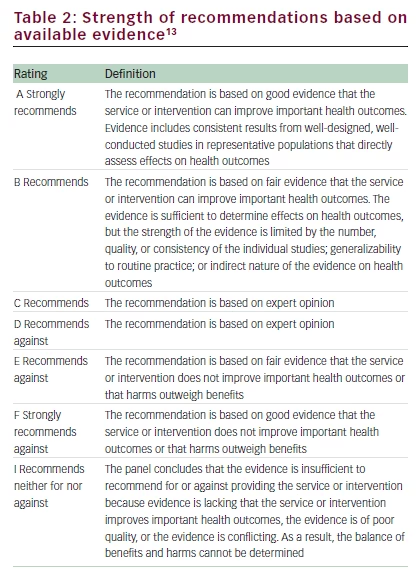
Recognizing the need for providers to gain experience in applying these guidelines to the care of children with DTC, the Program Committee of the Pediatric Endocrine Society invited a panel of experts to review and discuss representative cases of pediatric thyroid nodules and cancers at their annual meeting in April, 2015. Eight cases were submitted. The cases are presented here, and each discussion demonstrates application of the ATA pediatric guidelines to patient care with citation of the pertinent recommendations from the ATA guideline and the strength of the evidence using a modified schema from the US Preventive ServicesTask Force (Table 2).13
The ATA pediatric guidelines represent a paradigm shift in the care of children with DTC.12 Key evaluation and management points from the pediatric guidelines include the following.
• All pediatric patients are at low risk for mortality, but are classified as low-, intermediate-, or high-risk for persistent or recurrent disease based on pre- and post-operative staging.
• Pre-operative staging is recommended to facilitate selection of appropriate surgery. The majority of children will undergo TT and
central compartment lymph node dissection (CLND) but more extensive LND may be required if disease is identified beyond the central neck (level VI). Removal of only suspicious lymph nodes (berry-picking) is not recommended. Cross-sectional imaging is recommended for patients with extensive bulky neck disease that precludes adequate visualization, especially if disease is encroaching on or into the aerodigestive tract. Pre-operative chest radiograph or computerized tomography (CT) of the chest is recommended to identify pulmonary metastases.
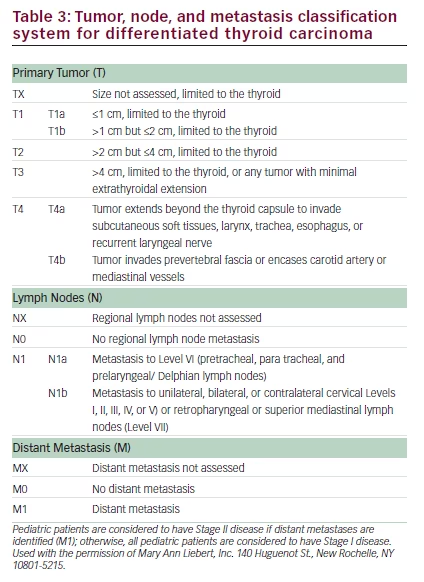
• Post-operative staging is recommended and utilizes standard tumor, node, and metastasis (TNM) nomenclature (Table 3) to describe the extent of disease.
–– For patients at low risk for recurrence (disease confined to the gland with no lymph node metastasis (American Joint Committee on Cancer [AJCC] TNM designation of N0) or incidental central neck lymph node metastasis (N1a disease), post-operative staging may only require serum Tg.
–– Patients at intermediate risk for recurrence based on the presence of extensive central lymph node metastasis (N1a disease) or minimal metastasis to lateral neck lymph nodes (N1b disease) usually require Thyroid stimulating hormone (TSH)-stimulated Tg and dxWBS (123I).
–– Patients at high risk for recurrence based on the presence of extensive lateral neck lymph node metastasis (N1b disease) or
invasive tumors with extra thyroidal extension in the peri-thyroidal soft tissue space (T4), with or without distant metastases require TSH-stimulated Tg and dxWBS (123I) in all cases.
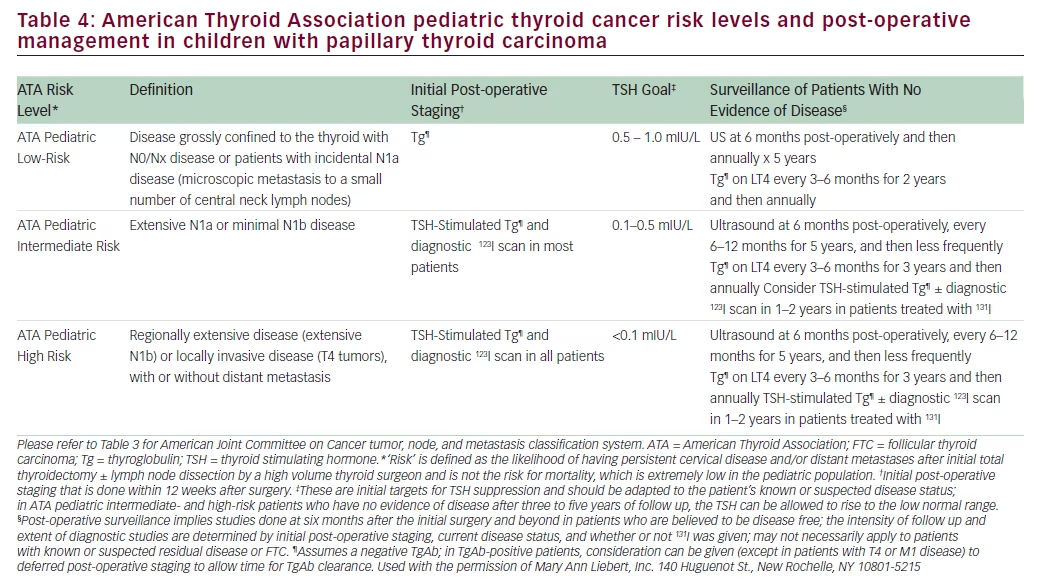
• Therapy and follow up are individualized based on the extent of disease and response to prior treatments (Table 4).
Case review
Case one
A 13-year-old male had an enlarged, firm thyroid gland (8.4 cm), normal serum thyrotropin (TSH, 1.9 μIU/ml), and neck ultrasound that showed a heterogeneously enlarged thyroid with hyperechoic foci throughout the gland associated with multiple enlarged lymph nodes with increased peripheral vascularity. This case demonstrates several important points in the evaluation of children with goiter or thyroid nodules.
The diffusely enlarged, heterogeneous, and hypervascular thyroid with multiple enlarged lymph nodes is a common presentation for PTC in children who do not always have a discrete nodule.14,15
• This is a large lesion and although size alone is not a major criterion for fine-needle aspiration (FNA) in children; considering history, family history, and US features are important.16–18
• Enlarged lymph nodes with increased peripheral vascularity are a risk factor for PTC.14,15
Due to a concern for possible DTC, FNA was performed but was nondiagnostic. The ATA pediatric guidelines suggest caution in evaluating large lesions (>4 cm) by FNA due to a high probability of sampling error. Furthermore, non-diagnostic FNA may be associated with a higher probability of malignancy in children. The FNA should either be repeated after three months to avoid confusion caused by cytologic changes which occur during post-FNA reparation, or the lesion should be removed.19,20
RECOMMENDATION 5
The evaluation and treatment of thyroid nodules in children should be the same as in adults with the exceptions that: 1. Ultrasound characteristics and clinical context should be used rather than size alone to identify nodules that warrant FNA, 2. All FNA in children should be performed under ultra sound-guidance, 3. Pre-operative FNA of a hyper functioning nodule in a child is not warranted as long as the lesion is removed, 4. A diffusely infiltrative form of PTC may occur in children and should be considered in a clinically suspicious gland, and 5. Surgery (lobectomy + isthmusectomy) is favored over repeat FNA for most nodules with indeterminate cytology.1 Recommendation rating: B
Based on the potential for error in the FNA, the patient was closely followed, as suggested by the guidelines.12
Repeat ultrasound confirmed micro-calcifications which are highly suspicious for PTC.14,15 Neck CT showed thyromegaly and enlarged cervical lymph nodes, prompting excisional lymph node biopsy that confirmed PTC. In this case, FNA of the involved lymph node and Tg assay of the FNA needle-wash were other options21 that are less invasive, highly accurate, and can detect DTC in lymph nodes even in the presence of high levels of serum Tg and Tg-antibody (Tg-Ab).21–25
SECTION B4
Insufficient or non-diagnostic cytopathology refers to a specimen with limited cellularity, absence of follicular cells or poor fixation and preservation. Repeat FNA is an option in children but should be delayed for a minimum of 3-months in order to decrease the potential for atypical cellular features that may arise during the reparative process. In adults, the risk of malignancy in indeterminate nodules ranges from ~5-15% in the AUS/FLUS category to 15-30% in the follicular neoplasm or suspicious for neoplasm group. The limited data available suggest these indeterminate FNA categories account for ~35% of pediatric FNA and that, in children, 28% of AUS/FLUS lesions and 58% of suspicious for follicular or Hürthle cell neoplasm are malignant. Due to the apparent increased probability of malignancy among these indeterminate categories in children, the task force recommends definitive surgery (lobectomy + isthmusectomy) for indeterminate FNA findings in children.1 Recommendation rating: B
RECOMMENDATION 8
Benign lesions should be followed by serial ultrasound and undergo repeat FNA if suspicious features develop or the lesion continues to grow. Lobectomy should be considered in all apparently benign solid thyroid nodules > 4cm, those lesions demonstrating significant growth, or in the presence of other clinical concerns for malignancy.1 Recommendation rating: B
Once a diagnosis of PTC is made, pre-operative staging is recommended for all children, including either chest radiograph or non-contrast chest CT. In this case, both neck and chest CT were performed and showed no indication of pulmonary metastases.
RECOMMENDATION 10
A comprehensive neck ultrasound to interrogate all regions of the neck is required in order to optimize the pre-operative surgical plan. FNA of suspicious lateral neck lymph nodes is recommended. Anatomic imaging by MRI or CT with contrast should be considered in patients with large or fixed thyroid masses, vocal cord paralysis, or bulky metastatic lymphadenopathy in order to optimize surgical planning.1 Recommendation rating: A
As recommended in the guidelines, the patient underwent TT and central neck LND. With such a large lesion, there is an increased risk for metastasis to the lateral neck and beyond. Surgical exploration of the lateral neck is not recommended, rather FNA should beperformed with planned, therapeutic compartment-based, lateral neck dissection, performed if FNA confirms metastasis to the lateral neck lymph nodes.12
Pathology confirmed PTC measuring 8.4 cm, adherent to the sternothyroid muscle, and metastatic to lymph nodes in the right (levels 2a, 2b, 3, 4, and 5) and left lateral (levels 3 and 4) neck. There was minimal extra-thyroidal invasion, but extensive lymphovascular invasion, resulting in TNM classification: pT3N1b.
RECOMMENDATION 11
For the majority of children, TT is recommended. This is based on multiple studies showing an increased incidence of bilateral and multi-focal disease. Bilateral lobar resection compared with lobectomy has been shown to decrease the risk for persistent/ recurrent disease. Recommendation rating: A
RECOMMENDATION 12(A)
Central neck dissection (CND) is recommended for clinical evidence of gross extra-thyroidal invasion and/or loco-regional metastasis and may be associated with a reduced need for second surgeries and an increase in disease-free survival (DFS). Recommendation rating: B
RECOMMENDATION 12(B)
For PTC and no clinical evidence of gross extra-thyroidal invasion and/or loco-regional metastasis, prophylactic CND may be selectively considered based upon tumor focality and size and the experience of the surgeon. For patients with unifocal disease, ipsilateral CND, with pursuit of contralateral CND based on intra-operative findings, may help balance the risks. Recommendation rating: C
RECOMMENDATION 12(C)
Compartment-oriented resection is recommended for LND. “Berry picking” and palpation to detect metastatic disease are not recommended. Recommendation rating: A
RECOMMENDATION 13
Confirmation of disease in the lateral neck is recommended prior to surgery. Routine prophylactic lateral neck dissection is not recommended. However, lateral neck dissection should be performed on patients with evidence of metastases to the lateral neck. Recommendation rating: B
As recommended by the guidelines, post-operative staging was performed. A post-operative dxWBS showed uptake in the thyroid bed and cervical lymph nodes but not lungs. The stimulated serum Tg was 228 ng/ml with a TSH level of 39.8 uIU/ml. However, the serum Tg-Ab was positive (1.8 IU/ml, reference range 0.0-0.9 IU/ml).
In adults with PTC, the magnitude of serum Tg correlates with location and histology of DTC.26 Although this has not yet been replicated in children, the magnitude of the serum Tg in this case suggested an increased risk of pulmonary metastases. However, the presence of even low titers of Tg-Ab can interfere with the Tg assay leading to falsely low (more frequent) or falsely high serum Tg.27,28
RECOMMENDATION 20
Based on the lack of data comparing empiric treatment and treatment informed by dosimetry, we are unable to recommend for or against either approach in most patients. Many experts provide the first activity of 131I based on an empiric estimate and reserve dosimetry for patients with diffuse pulmonary metastases or subsequent activities of 131I. However, dosimetry can be considered prior to the first 131I treatment in small children and patients with limited bone marrow reserve. Due to the differences in body size and iodine clearance in children compared with adults, it is recommended that all activities of 131I should be calculated by experts with experience in dosing children.1 Recommendation rating: I
The dxWBS showed uptake in the thyroid bed and several cervical lymph nodes, and the patient was treated with 154 mCi 131iodine (131I). Posttherapy scan showed diffuse pulmonary metastases. The dxWBS is not as sensitive as the post-therapy scan.29 Thus, a negative dxWBS is fairly common in a child with pulmonary metastases and should not imply absence of lung disease. Pulmonary metastases would be typical based on the TNM classification, the presence of extensive lympho-vascular invasion, lymph node involvement, and the magnitude of serum Tg.
The ATA pediatric thyroid cancer guidelines offer no standardized doses of RAI for children, but indicate that doses are adjusted by the ratio of their body size compared to that of an adult.1
The patient was placed on suppressive levothyroxine and followed. Fortunately, the TSH-stimulated Tg has declined from 228 to 2.3 ng/ml, indicating an excellent response. Serum Tg may continue to decline over 18 months in most cases.30 The guideline suggests that continued observation is warranted.1 Should the Tg increase, then repeat staging would be required to determine if there is recurrent disease in the neck or lungs.
Case two
A 17-year-old male had dysphagia, enlargement of the neck, and normal thyroid function, but ultrasound showing a markedly enlarged and heterogeneous right thyroid lobe without a distinct nodule but with hyperechoic foci throughout, which were concerning for microcalcifications. FNA was consistent with PTC. He underwent TT with central compartment LND. Pathology revealed diffuse-sclerosing variant of PTC (dsvPTC) with metastasis in 21/25 lymph nodes. The post-operative serum Tg was undetectable but the Tg-Ab was positive (181.3 IU/mL; normal 0-4 IU/mL). Post-operatively the patient was placed on thyroid hormone suppressive therapy.
Two months later, a spot urine iodine was 591.8 μg/mL (normal range 26.0–705.0 μg/L). The patient was started on a low-iodine diet, the levothyroxine was discontinued and the serum TSH increased (92 mIU/L). Due to the dsvPTC with extensive N1b disease, the patient was given 106.7 mCi 131iodine. dxWBS was not performed. Unfortunately, three hours later, he had a single episode of emesis. The post-therapy scan (RxWBS) five days later showed no tracer uptake.
This case raises several questions for management.
• First, dsvPTC is not a rare lesion in children. It tends to invade the entire lobe or gland and presents with diffuse thyroid enlargement and not a thyroid nodule.31–33 Suspicion should be raised in patients with a particularly firm gland, abnormal palpable cervical lymph nodes, or as in this case, the presence of micro-calcifications without a distinct nodule. The neck ultrasound did not reveal any suspicious lateral neck lymph nodes so the patient had a central LND.
• A thorough pre-operative staging with a neck US that examines all levels of the lateral neck (levels 2, 3, 4, and 5) as well as axial imaging with neck CT or MRI might have revealed extensive neck disease that would have prompted surgical re-exploration prior to the RAI therapy. RAI is effective for treatment of microscopic pulmonary metastases and small residual neck disease but is not useful for treatment of large neck masses.8,34
• Prior to therapy the urine iodine was elevated to 591.8 μg/L, which is higher than the pre-RAI treatment target of <75 μg/L.35 A repeat urine iodine was not performed to confirm that the urine iodine level decreased prior to RAI administration and if it were still elevated, this may have prevented tumor RAI uptake. The effectiveness of RAI might have been further reduced by the patient’s emesis. In retrospect, waiting until the urine iodine was confirmed to be <75 μg/L may have facilitated treatment. Pre-treating with ondansetron may have prevented nausea and vomiting, which are common following RAI.
RECOMMENDATION 25(A)
During follow up of children with PTC who are suspected to have residual disease, a dxWBS can be used to inform the decision of whether or not to use 131I and the activity of 131I to be administered. Recommendation rating: C
RECOMMENDATION 25(B)
A dxWBS should be performed in children with ATA Pediatric Highrisk disease who were previously treated with 131I or known to have iodine-avid metastatic disease based upon a previous post-treatment scan. The dxWBS should be obtained after at least 12 months of clinical follow-up, and deferred even longer in those children who continue to demonstrate a clinical response to previous treatment. Recommendation rating: B
RECOMMENDATION 25(C)
Once a negative dxWBS is obtained, there is no benefit from serial dxWBS to survey for disease recurrence as long as the patient otherwise remains without clinical evidence of disease. Recommendation rating: B
The patient had a repeat neck ultrasound six months later and FNA of suspicious lymph nodes revealed persistent metastatic PTC in the neck. A right lateral neck dissection was performed with PTC confirmed in 12/47 lymph nodes (2/14 Level IIA, 0/11 Level IIB, 7/12 Level III, 3/7 Level IV, 0/3 Level V). The Tg-Ab remains elevated (166.5 IU/ml, normal range 0-4 IU/mL) but the most recent serum Tg is undetectable by liquid chromatography-mass spectrometry (LC-MS/MS, ARUP Laboratories, 500 Chipeta Way, Salt Lake City, UT 84108-1221) and dxWBS one year later remains negative. Recent data suggest that the Tg measured by LC-MS/MS is not affected by the presence of Tg-Ab and is a good tumor marker for Tg-Ab positive patients as is the Tg-Ab titer which should decline in patients without residual disease.36 The undetectable Tg by LC-MS/MS suggests the patient may be free from disease37,38 in which case the Tg-Ab will continue to decline due to lack of thyroid antigens that maintain the immune stimulus. However, it takes an average of three years for the Tg-Ab to become negative in adults.39 If the Tg by LC-MS/MS was obtained under TSH-stimulation then the patient is highly likely to be free from disease.
Case three
A six-year-old Asian female had been diagnosed with stage 4 neuroblastoma at the age of two years. Sixteen months following treatment (tumor resection, chemotherapy, autologous bone marrow transplant, external beam radiation therapy to the abdominal tumor site [total 35Gy -estimated 3cGy to thyroid] and metaiodobenzylguanidine [MIBG] radionuclide whole body scanning [estimated 60 cGy to thyroid]), a thyroid nodule was incidentally detected on routine chest CT. There was no family history of neuroblastoma, thyroid nodules, or thyroid cancer.
She underwent lobectomy, followed by completion thyroidectomy revealing a unifocal, 3.2 cm, follicular variant papillary thyroid carcinoma (FvPTC) with focal capsular invasion but without lympho-vascular invasion. The tumor margins were free from disease.
Following surgery her serum Tg was 3 ng/ml (TSH = 1.98 uIU/ml and negative Tg antibody) but four months later the serum Tg increased to 8.4 ng/ml (TSH = 1.4 uIU/ml). Neck US identified a suspicious lesion in the thyroid bed that was removed and confirmed persistent FvPTC. Subsequently, the serum Tg declined to 0.3 ng/ml.
This case raised several questions.
• Did the radiation exposure contribute or was this an ‘incidental PTC’?
• What mutations do you expect to find in radiation associated PTC?
• Are there unique genetic associations between neuroblastoma and PTC?
• Is there a role for pre-operative staging and CND for an ‘incidental’ lesion of 3 cm?
• What is an ‘acceptable Tg’ after surgery?
• What is a ‘concerning’ rise in serum Tg?
Thyroid cancer is a common second malignancy in the Childhood Cancer Survivor Study, with a standardized incidence ratio of 11.3 (95% confidence interval [CI] = 8.2–15.3).40,41 Radiation exposure is associated with an increased risk for PTC but the peak incidence is 10–30 years after exposure. PTC that arise following radiation have a greater frequency of RET-PTC3 gene rearrangements than are found in spontaneous PTC.42 Treatment with alkylating agents or other chemotherapeutic agents is also associated with an increased risk for PTC, but only if the radiation dose is <20 Gy (alkylating agents [relative risk (RR) 3.25; 95% CI 0.9–14.9], anthracyclines [RR 4.5; 95% CI 1.4–17.8], bleomycin [RR 3.2; 95% CI 0.8–10.4]).40,43 Most relevant to this discussion, PTC is five times more common as a second malignancy following radiation therapy for neuroblastoma when compared with radiation therapy for any other primary malignancy.44 In a long-term follow-up study of 16 survivors of childhood neuroblastoma, 81% had thyroid hormone abnormalities, 56% had thyroid nodules and 12.5% had PTC.45,46 This suggests the possibility of shared genetic predisposition which was recently emphasized by publicaiton of heterozygous missense mutation in STK11 in both neuroblastoma and FvPTC implicating STK11 as a possible shared driver mutation.47
In this particular case, a 3 cm lesion in a six-year-old yr child is concerning and would not ordinarily be considered an ‘incidental’, lesion irrespective of how it was discovered. Pre-operative staging should be performed to ensure surgical planning that would increase the likelihood of complete surgical resection.
The goal following total thyroidectomy is to achieve a TSH-suppressed serum Tg <1.0 ng/ml. In this case, the serum Tg was 3 ng/ml and increased to 8.4 ng/ml over four months, both of which suggested persistent disease that was later confirmed and removed. After the second surgery, the patient’s serum Tg declined to 0.3 ng/ml, which is reflective of successful resection. (See Recommendation 23 A–E in case one).
RECOMMENDATION 10
A comprehensive neck ultrasound to interrogate all regions of the neck is required in order to optimize the pre-operative surgical plan. FNA of suspicious lateral neck lymph nodes is recommended. Anatomic imaging by MRI or CT with contrast should be considered in patients with large or fixed thyroid masses, vocal cord paralysis, or bulky metastatic lymphadenopathy in order to optimize surgical planning.12 Recommendation rating: A
Case four
A 15-year-old obese Hispanic male with subclinical hypothyroidism and AIT (TSH = 6.72 μIU/mL, reference range = 0.35–5.5 μIU/mL; and positive thyroid peroxidase antibody 75 IU/ml, reference range 0–35 IU/ml) had a significant family history of thyroid disease. The father and a maternal cousin had autoimmune hypothyroidism and the mother and maternal cousin had thyroid cancer. On exam, the patient’s thyroid was not enlarged and there were no palpable nodules or cervical lymphadenopathy, however, examination was limited by body habitus.
Based on the strong family history of thyroid cancer, ultrasound was performed, revealing a 1.8 x 1.6 x 0.8 cm hypoechoic nodule contained punctate hyperechoic foci as well as multiple suspicious cervical lymph nodes.14,15 Total thyroidectomy and central compartment LND revealed a 1.5 cm PTC in a background of lymphocytic thyroiditis with lymph node metastasis; TNM stage pT1bN1aMx.
This case raised the following questions.
• Should a family history of PTC prompt additional evaluation in children?
• Should a thyroid ultrasound be performed in patients with AIT when there are no nodules or abnormal lymph nodes palpated?
PTC is a common endocrine tumor and many pedigrees have at least one relative with PTC. The risk of familial, non-medullary thyroid cancer increases when two first degree relatives have DTC, approaching a 96% likelihood of an inherited disorder when three or more first degree relatives have DTC.48 However, even if PTC were limited to the mother, the probability of having children with PTC is 4.5-fold more common (OR, CI = 1.2–19.8, p=0.045).49 The risk is even greater (odds ratio [OR] = 7.4, range 1.8–30.4) if that relative is a sibling.50 In these families, ultrasound surveillance identifies tumors with smaller size (0.8 cm vs. 2.85 cm; p<0.001), a lower incidence of extrathyroidal extension (20.9% vs. 56.2%; p=0.002), and a lower incidence of lymph node metastasis (23.2% vs. 65.6%; p<0.001).51 The ATA pediatric thyroid cancer guidelines offer no clear recommendations for prospective screening (beyond routine physical examination) in most families containing a member with PTC.12 However, this may change in future versions of the guidelines and there are updated recommendations for US screening for both the PTEN Hamartoma Tumor Syndrome, DICER-1 related disorders, and APCAssociated familial adenomatous polyposis.52
The second question raised by these data is whether or not patients with autoimmune thyroid disease warrant ultrasound, even if they do not have a suspicious thyroid gland by physical exam. Corrias, et al.2 performed ultrasound on 365 young patients with autoimmune thyroid disease and found 115 thyroid nodules (31.5%), of which, only 38 (onethird) were palpable. There were 11 PTC (10% of all nodules or 3% of all patients). Growth of a nodule while the patient was taking levothyroxine and abnormal lymphadenopathy were more common in those with PTC. Kambalapalli, et al., performed ultrasound on 154 children with goiter but without any palpable nodules.3 They found nodules in 20/154 patients (13%) and PTC in 4/20 nodules (20%) or 4/154 patients (2.6%). None of the PTC were palpable, although one was >2 cm in diameter and two had microscopic regional lymph node involvement (N1). The ATA pediatric thyroid cancer guidelines suggest that a suspicious-feeling thyroid gland warrants ultrasound examination.12 This leaves open the possibility to perform ultrasound but it is not imperative.
RECOMMENDATION 4(D)
For patients with AIT, evaluation by an experienced thyroid ultrasonographer should be pursued in any patient with a suspicious thyroid examination (suspected nodule or significant gland asymmetry), especially if associated with palpable cervical lymphadenopathy.12 Recommendation rating: B
In this particular patient, there were many factors that support the use of ultrasound: obesity limiting adequate examination, autoimmune thyroid disease, and a family history of PTC in a first degree relative.
Case five
A 10-year-old Hispanic male underwent resection of a thyroglossal duct cyst that was found to contain incidental PTC. Neck US showed no other evidence for disease and the patient had no further surgery or medical therapy at that time. Five years later, PTC recurred in the submental and submandibular lymph nodes. The patient underwent a Sistrunk procedure, total thyroidectomy, and bilateral modified radical neck dissection at levels II, III, and IV. Final pathology confirmed PTC in 10/84 lymph nodes but there was no PTC in the thyroid gland. Following surgery he received 97 mCi 131I and the post-therapy whole body scan showed uptake only in the thyroid bed. The patient was started on levothyroxine and has had a declining but persistent stimulated serum Tg (4.6–1.1 ng/ml).
This case raised several different questions.
• What evaluations of the thyroid gland and neck are appropriate for PTC in a thyroglossal duct cyst?
• What is optimal treatment for PTC in a thyroglossal duct cyst?
• What should be done for a persistent low-level but detectable serum Tg?
There are few published series of PTC in thyroglossal duct remnants in adults53,54 and only one in children.55 Thyroid cancer is found in 0.7–1.5% of thyroglossal duct cysts in adults and 88.5% are PTC. In adults, 61.5% of patients also have PTC in the thyroid gland, 30% show extension into soft tissue, and 88.2% have regional lymph node involvement. However, the lymph node involvement often follows a different pattern depending on the location of the thyroglossal duct remnant. Lesions near or above the thyroid cartilage are associated with metastasis to level 1 and 2 rather than the central compartment (level 6) or levels 2, 3, and 4 in the lateral neck and level 6, which are more typcial for intrathyroid PTC.53 Carter, et al., recommended ultrasound examination of the thyroid gland and neck nodes, and total thyroidectomy only if there was invasion of the wall, aggressive histology, or ultrasound findings indicating disease in the gland.54 In pediatric patients, coincidental PTC appears to occur less frequently than in adults. Pfeiffer, et al. studied 26 cases in children.55 Although 7% of the population have thyroglossal duct remnants, cancer occured in only 1% of thyroglossal duct remnants. The vast majority (22/26 cases) were PTC. Although 50% had cervical lymph node metastases, none of 13 patients who had total thyroidectomy had PTC in the thyroid gland. For children, they recommended the Sistrunk procedure plus local LND, but TT only for evidence of disease in the thyroid.
In this case, initial surgery did not include the Sistrunk procedure, or dissection of the cervical nodes. Recurrence in the regional nodes appeared five years later, consistent with the data outlined above. There was no evidence for PTC in the gland, which is also consistent with the published series in children.
Following therapy, the patient has had a persistent but declining serum Tg. This indicates persistent but low level disease that has been difficult to localize (See recommendations 23 A–E in Case One above). Continued surveillance in this situation is appropriate.
Case six
A 16-year-old female had a palpable thyroid nodule and hyperthyroidism (TSH <0.01 μIU/mL, total T4 = 16.1 μg/dL, free T4 = 3.0 ng/dL and total T3 = 322 ng/dL). Thyroid ultrasound revealed a 4.2 x 1.8 cm heterogeneous partially cystic nodule in the right lobe and a 1.6 x 0.7 x 1.0 cm nodule in the left lobe. Due to the suppressed TSH, thyroid uptake and scan were obtained showing increased uptake (53%) in the right lobe and minimal uptake in the remainder of the thyroid consistent with a hyperfunctioning nodule. Right hemithyroidectomy was performed revealing a 2.2 cm encapsulated well differentiated PTC without capsular or lymphovascular invasion. During follow up, FNA of the left thyroid nodule was performed and revealed indeterminate cytology (Atypia of Undetermined Significance [AUS]—Bethesda III Category). The Afirma gene expression classifier ([GEC] Veracyte, 7000 Shoreline Court, Suite 250, South San Francisco, CA 94080) was benign. With repeat surveillance ultrasound, the left thyroid nodule (AUS) remains unchanged in size or appearance.
This case raised the following questions.
• What is the incidence of PTC in hyperfunctioning nodules in children?
• Does PTC in a hyperfunctioning nodule have the same prognosis?
• What is the risk of malignancy in lesions with AUS?
• How effective are ‘gene expression classifiers’ in pediatric tumors?
• Is there a role for completion thyroidectomy on the remaining left side?
Autonomous nodules in children may be associated with somatic activating mutations within the genes encoding the TSH receptor or the Gsalpha subunit.57 In adults, treatment options include 131I ablation, surgical resection, or ethanol injection. Because of concerns of the mutagenic effect of low-activity radioiodine on the surrounding normal thyroid, and the fact that one third of patients have an incidentally discovered DTC, surgical resection is usually recommended for pediatric patients with autonomous nodules.12 There are no data to indicate a different prognosis for children with PTC in autonomous nodules.
RECOMMENDATION 9
For pediatric patients with a suppressed TSH and a thyroid nodule, thyroid scintigraphy should be pursued. Increased uptake within the nodule is consistent with autonomous function. Surgical resection, most commonly lobectomy, is the recommended approach for most autonomous nodules in children and adolescents. Recommendation rating: A
In adults, the risk of malignancy in indeterminate nodules ranges from ~5 to 15% in the AUS/ Follicular Lesion of Undetermined Significance (FLUS) category and 15 to 30% in the follicular neoplasm or suspicious for neoplasm group.58 The limited data available suggest these indeterminate categories account for approximately 35% of pediatric FNA and that, in children, 28% of AUS/FLUS lesions and 58% of suspicious for follicular or Hürthle cell neoplasm are malignant.19,59
In adults, molecular testing aids in the management of thyroid nodules with indeterminate cytology.60 However, molecular studies have not been validated in pediatric patients, although mutational analysis has been used to examine thyroid nodules in limited numbers of children.19,61 Approximately 17% of pediatric FNAs have a mutation or rearrangement that correlates with malignancy in 100%.19 However, the cytological classification for these tumors were AUS/FLUS, suspicious for follicular or Hürthle neoplasm, suspicious for malignancy, or malignant, all of which would require surgical removal regardless of the mutational analysis.
RECOMMENDATION 6
A positive mutational test appears highly likely to be associated with malignancy. Conversely, insufficient data exist in children to rely on negative genetic studies to reliably exclude malignancy. Although molecular studies hold promise for complementing the results of FNA, particularly for nodules that yield indeterminate cytology, they have not yet been sufficiently validated in children and cannot be routinely recommended in routine clinical practice until further studies are conducted.12 Recommendation rating: E
Although a proprietary multigene expression classifier has been validated to corroborate a benign diagnosis in adults with indeterminate nodules, this panel is only approved for patients >21 years of age. There are no studies determining the accuracy of the gene expression classifier in the evaluation of the indeterminate pediatric thyroid nodule. Therefore, although current molecular diagnostics might improve the diagnostic acumen for indeterminate cytology in children, additional studies are required before a formal recommendation can be offered or incorporation into clinical practice.
For pediatric patients with PTC, there is a 25% risk of bilateral disease even if undetected by thyroid US. This patient incurred damage to the recurrent laryngeal nerve during the initial lobectomy. For that reason, despite the potential risk for bilateral disease, the family agreed to purusue US surveillance of the remaining nodule with repeat FNA if indicated.
Case seven
A 10-year-old male presented with a two- to three-month history of neck mass. CT scan showed bilateral scattered enlarged lymph nodes in the anterior and posterior neck. A lymph node biopsy revealed PTC and the patient underwent total thyroidectomy with extensive bilateral LND. Pathology showed: dsvPTC with extrathyroidal invasion at both anterior and posterior margins, extensive lympho-vascular invasion, and 27/74 lymph nodes infiltrated by PTC. Five weeks after surgery, a dxWBS (123I) with rhTSH preparation showed uptake in two left neck foci and a thyroglossal duct remnant. An 18fluorodeoxy glucose positron emission tomography (FDG-PET) scan showed low-level focal uptake in the right supraclavicular and paratracheal areas. CT of the chest identified seven small (3-5 mm) lung nodules. The tumor was classified as T3 N1b M1.
The patient was treated with 131I (58.6 mCi) following two doses of recombinant TSH (0.9 mg each injection). At the time of therapy, the serum TSH was 458 μIU/mL and the serum Tg was 384 ng/ml.
Residual disease was noted seven months after the initial therapy with a right supraclavicular node and a conglomerate mass in the right paratracheal region. Due to progressive growth of this mass, he underwent a second surgery to remove the mass and 2/10 lymph nodes which were involved with PTC.
Fifteen months after the initial surgery he began treatment with the mitogen activated protein kinase (MEK) inhibitor (trametinib at a dose of 0.025 mg/kg).
This case raised the following questions:
• What is a working definition of iodine refractory disease in children?
• How do we best treat low-iodine avid disease?
• What is the role for dosimetry in patients with pulmonary metastases?
• Is there a role for empiric RAI?
• Is there a role for FDG-PET in children?
• Should additional surgery be performed to remove macroscopic neck disease prior to RAI for pulmonary metastases?
• At what point do we consider treatment with alternate therapies like tyrosine kinase inhibitors (TKIs) or MEK inhibitors?
The ATA pediatric thyroid cancer guidelines suggest that treatment of residual disease should be based on multiple factors and individualized to the patient’s age, risk stratification, and history of prior treatment and response to treatment.12
In adults, iodine refractory disease as adapted from Dadu and Cabanillas, 201262 is defined as follows.
• Disease does not take up RAI at known sites of metastatic disease.
• Disease that grows despite 131I and confirmed uptake.
• Disease that grows over a 1 year period after 131I.
• The patient has received a cumulative 131I>600 mCi and has persistent disease.
However, iodine refractory disease has not been defined for children. It is useful to remember that disease-specific mortality for DTC in children is less than 3% and long-term survival is >97%.63 Even in children with pulmonary metastasis, a combination of surgery, 131I therapy, and TSH suppression results in complete (47.32%) or partial (38.39%) response in the majority of children. Only 14.29% of children with pulmonary metastases fail to show any response to treatment.63 Approximately 30–45% of children with pulmonary metastases develop stable but persistent disease following surgery and 131I therapy.8 Based on these facts and the fact that 131I may continue to have an effect for many years after treatment in children, the ATA pediatric thyroid cancer guidelines state that an undetectable Tg level should no longer be the sole goal of treatment of children with pulmonary metastases.12
RECOMMENDATION 28(A)
The decision to treat or to observe structural cervical disease should be individualized and include considerations of age, initial ATA Pediatric Risk classification, the presence of distant metastases, and prior treatment history (including complications), in addition to the size, extent, anatomic location and iodine avidity. Recommendation rating: C
RECOMMENDATION 28(B)
Children with macroscopic cervical disease (>1 cm in size) should be assessed by a high-volume thyroid surgeon to determine the feasibility of additional surgery. Recommendation rating: B
RECOMMENDATION 28(C)
Iodine-avid cervical disease (visualized with dxWBS) could be treated with surgery or 131I depending on risks and the presence or absence of distant metastases. Surgery would be favored for disease localized to the neck, especially if located in a lymph node compartment not previously operated upon. Recommendation rating: B
RECOMMENDATION 28(D)
If repeat surgery is performed, postoperative re-staging can be utilized to determine whether additional 131I treatment is warranted, especially in the patient who has not received previous therapeutic 131I.1 Recommendation rating: C
As reviewed in case one, the dxWBS is not as sensitive for detecting disease as is the serum Tg level. A negative dxWBS does not prove absence of disease nor can it be used as evidence for iodine refractory disease. Metastatic PTC in lymph nodes typically has reduced RAI absorption. For that reason, there may be increased lymph node uptake when higheractivities of RAI are given. It is not entirely clear however, if this will achieve tumoricidal effect on bulky lymph node metastasis. Bulky lymph node uptake also reduces the efficacy of RAI to treat pulmonary metastases. For these reasons, it is prudent to re-operate (if possible) to remove all residual neck disease prior to 131I therapy.
Without a clear definition of iodine reractory disease in children, continued treatment for pulmonary metastases should be given only to those who show signs of progression (increase in tumor size as detected by anatomic imaging or serum Tg) and documented clinical response to previous 131I therapy. If the patient has no evidence of cervical disease, low urine iodine excretion, and still shows no uptake of 131I into the pulmonary lesions, then it is unlikely that a higher or additional doses of 131I would benefit. At that point, if the patient displays progressive, RAI refractory pulmonary or non-surgical disease, one may consider systemic therapy.
For adult patients with iodine refractory disease, there is an increasing number of molecular targeted therapies. The agents target protein tyrosine kinase dependent pathways and are collectively referred to as multi-kinase inhibitors (TKIs).64 TKIs have been studied in adults with advanced PTC and FTC, medullary thyroid cancer (MTC) and anaplastic thyroid caricinoma.64 For adults with an increased risk of disease-specific mortality, the benefit of extending PFS is significant. As an example, phase II trials of sorafenib have response rates up to 30% with median PFS between 58–79 weeks.64 The Double-Blind Randomized Phase III Study Evaluating the Efficacy and Safety of Sorafenib Compared to Placebo in Locally Advanced/Metastatic RAIRefractory Differentiated Thyroid Cancer (DECISION) trial reported a 12% response rate and an increase in median PFS from 5.8 to 10.8 months.65 More recently, the Multicenter, Randomized, Double-Blind, Placebo-Controlled, Phase 3 Trial of Lenvatinib (E7080) in 131I-Refractory Differentiated Thyroid Cancer (SELECT) trial reported increased PFS for FTC (28.8 months vs. 2.4 months for placebo) and PTC (16.4 months vs. 3.5 months for placebo).66 Almost 75% of patients responded within 30–days after intitiatng treatment and four of the 169 patients achieved complete remission. An increasing list of additional TKIs are currently in phase II clinical trials.67,68
RECOMMENDATION 29(A)
Children with RAI-avid pulmonary metastases visualized with a DxWBS are good candidates for 131I therapy. Recommendation rating: A
RECOMMENDATION 29(B)
After a therapeutic activity of 131I, the TSH-suppressed Tg level and imaging studies should be monitored until the nadir clinical and biochemical (Tg) response is reached. Recommendation rating: B
RECOMMENDATION 29(C)
If the nadir clinical and biochemical (Tg) response suggests persistent disease or if there is documented disease progression >12 months after 131I therapy, further evaluation with a DxWBS and a TSH-stimulated Tg is indicated. Recommendation rating: B
RECOMMENDATION 29(D)
Re-treatment of RAI-avid pulmonary metastases should be considered in children who have demonstrated progression of disease and a previous response to 131I. Treatment with 131I should be performed by experts with experience in managing children with pulmonary metastases. Recommendation rating: B
RECOMMENDATION 29(E)
Re-treatment of pulmonary metastases with 131I is not recommended in children who do not have uptake on a dxWBS and who have not demonstrated a previous response to 131I. Recommendation rating: E
RECOMMENDATION 29(F)
Pulmonary function testing should be considered in all children with diffuse pulmonary metastases, especially if multiple 131I treatments are planned.1 Recommendation rating: C
One exciting area is redifferentiation therapy, which is designed to increase expression of the sodium-iodine symporter (NIS). Retinoic acid was initially tried but found to be ineffective.69,70 More recently, the MEK 1/MEK 2 inhibitor (selective mitogen-activated extracelluar signalregulated kinase), selumetinib (75 mg twice daily x 4 weeks), was shown to increase RAI uptake to the threshold required for 131I treatment in eight of 12 patients with NRAS or BRAF mutations.71 Trametinib, the MEK inhibitor used in this particular case has a similar target. Although trametinib appears to be effective in treating BRAF mutant malignant melanoma,72 there is only one study examining its potential effectiveness in thyroid cancer cell-lines and a xenograft mouse-model.73
Four TKIs have received FDA approval for use in adults with advanced thyroid cancer; sorafenib and lenvatinib for DTC, and vandetanib and carbozantinib for MTC.74 Phase I and II trials have also been conducted to determine the safety and efficacy of sorafenib in pediatric patients with refractory solid tumors or leukemias.75,76 However, no children with PTC were enrolled into either of the pediatric sorafenib trials75,76 and there is only limited or anectodotal experience in using molecular therapies in children.77,78 A small phase I/II trial of vandetanib in pediatric patients with locally advanced or metastatic MTC reported partial response in 12/16 patients (decrease in calcitonin) and 8/16 subjects (decrease in carcinoembryonic antigen).79
With the exception of vandetinib for treatment of advanced MTC, none of these agents have been examined in the pediatric population for the treatment of progressive, non-RAI responsive disease. All of these agents have significant toxicities and even in adults with progressive, RAI-refratory disease, none of these agents improves overall survival.64 National clinical trials are needed to determine which pediatric patients would benefit from systemic therapy as well as the best strategy for their clinical use.
RECOMMENDATION 31
Most children with asymptomatic and non-progressive 131I-refractory disease can be safely monitored while continuing TSH suppression. Systemic treatment for advanced thyroid cancer in children remains unstudied and at this time should be considered the purview of specialized centers for the treatment of children with thyroid cancer.1 Recommendation rating: C
The 18FDG-PET/CT scan is increasingly used in the evaluation of adults with persistent non iodine-avid thyroid cancer80–82 and appears to offer prognostic information that might change clinical management.83 However, there are limited data regarding the use of 18FDG-PET/CT in children, except for a case report, isolated pediatric subjects embedded within adult studies, and unpublished observations. These reports suggest low sensitivity of 18FDGPET/ CT to identify disease in children that cannot be identified with US and cross-sectional imaging (Personal communication, SGW). Whether or not the 18F-FDG PET has similar prognostic value or will change disease management in children with thyroid cancer remains to be determined.
RECOMMENDATION 26(A)
For the child with a detectable TSH-suppressed Tg but a negative ultrasound and dxWBS, contrast-enhanced cross sectional imaging of the neck and chest should be considered once iodine excess has been eliminated as a cause of a false negative dxWBS. Recommendation rating: B
RECOMMENDATION 26(B)
The utility of 18FDG-PET/CT is poorly studied in pediatric DTC and 18FDG-PET/CT cannot be routinely recommended in the care of children who have persistent evidence of DTC on follow-up. Recommendation rating: D
RECOMMENDATION 26(C)
Empiric 131I therapy and a post-treatment scan are not recommended to localize disease in the child with a negative DxWBS unless there is evidence for clinical progression (e.g. a rising Tg level) and a documented clinical response to previous 131I therapy.1 Recommendation rating: D
Case eight
A 14-year-old with Hashimoto’s thyroiditis was found to have a 3 cm PTC in the right lobe that was densely adherent to the strap muscles and trachea. He underwent TT, central LND and dissection of the right lateral neck (levels 2–4). A total of 24/71 central nodes contained tumor and 1/3 lateral nodes contained tumor. He was treated with 130.6 mCi 131I and the post-therapy scan showed multifocal areas of persistent uptake in the left neck (contralateral). Eight months later, the basal serum Tg was 11 ng/ml (negative Tg-Ab) and repeat surgery removed 41 lymph nodes from the left neck (5/41 contained tumor). The basal (Tg = 5.7 ng/ml) and TSH-stimulated Tg levels (79.6 ng/ml) remained elevated. Despite that, US, neck and chest MRI, PET scan, and dxWBS failed to identify disease. After four years, empiric RAI therapy (191.4 mCi 131I) was given. Post-therapy scan and singlephoton emission computed tomography (SPECT-CT) showed uptake in the lungs that was not visible on CT, suggesting micro-nodular metastasis. After his second RAI therapy, the basal Tg was detectable but lower (1.6 and 3.5 ng/ml).
This last case raised several unique questions.
• Why did imaging fail to show disease with a stimulated serum Tg of 79.6 ng/ml?
• Is there a role for ‘empiric’ RAI therapy and if so, at what point?
• How is it best to follow and or treat, now that basal Tg is low but still detectable?
This case demonstrates the extensive regional lymph node involvement that can occur in pediatric patients and underscores the importance of pre-operative staging, well-executed surgical resection, and the need for reassessment if the surveillance Tg does not decrease. Micrometastasis to regional lymph nodes may be difficult to visualize on pre-operative ultrasound and MRI, although this is very uncommon. Initial surgery removed central and right lateral nodes that contained tumor but surveillance Tg suggested persistent disease. This was only identified by post-therapy 131I scan and confirmed during left lateral neck dissection.
It is difficult to determine why all imaging modalities failed to identify disease with a serum Tg of 79.6 ng/ml. Highly sensitive immunometric assays for Tg are more sensitive for detecting disease in adults than dxWBS.29 However, in one pediatric study, ultrasound and dxWBS equally identified lymph node metastases in the majority of patients (35/45). However, in six patients, lymph node metastases were found only with a post-treatment WBS.84
Previous studies of children with DTC have focused on the dxWBS as the ‘gold-standard’ for disease detection, and there are few data regarding the interpretation of Tg levels in children with DTC. Children with PTC who have residual/recurrent disease are most likely to have cervical lymph node disease. US, in conjunction with serum Tg, has proven highly effective in identifying and localizing regional nodal metastases in both adults and children with PTC and may even be more sensitive than a TSHstimulated Tg to identify disease.85 (Recommendation 23 A–E in case one) As in the previous case, there are few data regarding the use of 18FDGPET/ CT in children with DTC.86
Finally, empiric treatment with 131I does not appear to be effective in adults who have a negative dxWBS.87,88 Although children are more likely to have RAI-responsive disease, empiric treatment with 131I is not generally advocated to identify disease, unless there is evidence for clinical progression (e.g. a rising Tg level) and a documented response to previous 131I therapy. In select cases such as this, with rising serum Tg (1.6–3.5 ng/ml), no evidence of surgically resectable disease, and previous response to 131I therapy (decline in serum Tg) empiric was indicated and helped identify micro-nodular pulmonary metastasis.
Conclusion
The authors of this series of cases hope that their presentation and discussion have been informative and will help to illustrate how the ATA Pediatric Thyroid Cancer Guidelines can be applied to case management. For full details of the ATA Treatment Guidelines for Children with Differentiated Thyroid Cancer, the reader is referred to the Management Guidelines for Children with Thyroid Nodules and Differentiated Thyroid Cancer.12







