The steadily increasing knowledge concerning selenium (Se) involvement in chronic autoimmune and viral inflammatory disease along with the growing prospect of preventive and/or therapeutic Se intervention, via diet or compounds, are the focus of this brief review. Important data on inflammation are provided, while the link between Se status and disease is described through an overview of the involvement of Se in the molecular mechanisms of inflammation.
Se is an essential element incorporated in the form of selenocysteine (Secys) in the active site of selenoproteins, which are crucially involved in regulating immune response, oxidative stress, and chronic inflammation.1 Thus, Se, being a constitutional component of deiodinases (DIO), plays a pivotal role in thyroid hormone metabolism and protection of the thyroid gland against oxidative stress-induced damage.2 In the 1980s, the observation of the development of myxoedematous cretinism and thyroid destruction following Se repletion in iodine- and Se-deficient children led to an increasing focus on Se research and thyroid pathophysiology.3,4 Though minor amounts appear to suffice for adequate activity of DIOs, the maintenance of a Se steady state via Se intake, “selenostasis,” is required to preserve the activity of glutathione peroxidase (GPX1) and thioredoxin 1 (TXRND1) in both thyroid and redox processes.5,6 Regarding more specifically TXNRD1, experimental studies showed that rats fed a Se-deficient diet exhibited decreased TXNRD1 levels in the kidney, liver, and lungs, while in the spleen and prostate the levels were unaltered compared with the rats fed a high Se diet.7 However, no increase was found in TXNRD1 protein synthesis, suggesting that the tissue-specific TXNRD1 increase was probably due to elevated Secys incorporation.
Inflammation occurs in two types, namely acute and chronic inflammation, which can be defined respectively as a regulated and a dysregulated form of the disorder.8 Pro-inflammation is a common biologic phenomenon providing first-line defense against infection or invasion of pathogenic biologic agents, such as viruses, bacteria, and parasites. On the other hand, should the balance between pro- and anti-inflammatory mechanisms be disrupted, pro-inflammation can lead to wide-ranging detrimental effects. In this line of evidence, an increased differentiation and synthesis of pro-inflammatory Th17 cytokines, as has been observed in autoimmune thyroid disease (AITD) conditions and mainly in Hashimoto thyroiditis (HT), suggests that this phenomenon may have a pathogenetic role.9 In addition, low-grade inflammation can contribute via pathogenic mechanisms to developing insulin resistance, dyslipidemia, atherogenesis, type 2 diabetes mellitus (T2DM), and hypertension in obese individuals.10
There is good evidence that Se could have a positive impact on the course and outcome of a number of etiologically inflammatory diseases.11–13 Although the data are based almost exclusively on in vitro studies, and certainly caution is advised in interpreting the results, there are strong indications that viral-, bacterial-, and/or stress-induced inflammation could be variably influenced by Se availability.
The aim of this study is to discuss, through reference to the latest relevant data, the impact of Se in health and disease, and especially in the context of viral and metabolic diseases. Discussion of Se and cancer has been avoided as it is a topic of a separate review; the same applies to Se and the thyroid since various recent studies and reviews exist that have dealt with this topic. Nevertheless, given that Se and chemoprevention and Se and the thyroid are today subjects of considerable significance, a brief report follows on the current state of knowledge of Se research in these two latter fields.
Selenium and Chemoprevention
It has been suggested that high Se levels are capable of protecting against several types of cancer. However, the Selenium and Vitamin E Cancer Prevention Trial (SELECT), a randomized, double-blind, placebo-controlled study that was conducted to assess the efficacy of Se and vitamin E, both alone and in combination, in the management of prostate cancer did not observe any reduction in the incidence of prostate cancer after 7 years.14,15 A systematic review including trials involving 3,418 prostate cancer patients from 13 countries demonstrated that trials supplementing pomegranate seed, green tea, broccoli, soy, lycopene, Se, and coenzyme Q10, and comparing a low-fat diet versus a usual diet, displayed beneficial effects of Se on prostate cancer progression.16 In addition, Se nanoparticles loaded with anisomycin, an anticancer molecule, has recently been reported to improve antihepatocarcinoma efficacy.17 It is therefore advisable that in future studies of Se chemoprevention, the specific formulation of Se compounds and the dosage, the nutritional habits of the study cohort, the study design, and the age of participants and the baseline Se levels should be carefully considered. Relevant to the concept presented in this section is the fact that protein unfolding, which is involved in cancer development, can be inhibited by diet modifications, such as Mediterranean diet.18 Most particularly, it has recently been observed that Se from sodium selenite, reacts with free sulfhydryl groups of proteins, thus preventing reductive cleavage of disulfide bonds, this followed by protein unfolding and abnormal refolding.19,20
However, no convincing evidence exists either, which should be clearly stated, of a causal relation between Se and cancer that Se supplements can prevent cancer in humans.21
Selenium, Obesity, and Diabetes
In the SELECT study, higher rates of obesity were clearly observed among African American than non-Hispanic white men, possibly suggesting that reducing obesity among African American men could decrease the racial disparity in cancer incidence.22 In a study examining the association between Se status and metabolic risk factors in men with visceral obesity, Se was positively correlated with high-density lipoprotein (HDL) and fatty acid binding protein-4, (FABP4), while GPX3 activity was negatively correlated with FABP4.23 In obese men who had at least one component of the metabolic syndrome (MS), GPX3 activity was positively correlated with HDL. These findings indicate that Se status is linked to metabolic risk factors
only in particular groups of obese men with or without MS.22 This partially accords with recent animal studies suggesting a risk that long-term high Se intake could induce insulin resistance and T2DM.24 It has meanwhile been shown that high Se intake (0.5–3 mg/kg of diet) results in insulin resistance and diabetes-like states in animals. Though the mechanism is as yet unclear, upregulation, via high Se intake of selenoproteins such as GPX1, selenoprotein S (SELS) and selenoprotein P (SEPP), β-cells, insulin synthesis, and secretion, resulting in hyperinsulinemia as well as dysfunction of major regulators of glycolysis, gluconeogenesis, and lipolysis.24
In humans, the results from observational cross-sectional studies have raised concern that high Se exposure may be associated with T2DM or insulin resistance, at least in well-nourished populations.25,26 Therefore, the interactive role of Se, and especially the deleterious role of selenosis (Se toxicity) in diabetogenesis, should be considered when long-term studies with high Se intake are implemented.
Additionally, plasma Se and GPX levels were found to be low in T2DM patients with macroalbuminuria and were related to the stage of diabetic nephropathy.27 Also, in patients with chronic kidney disease (CKD), both Se and plasma glutathione peroxidase (GSH-PX), which is synthesized in the kidney using Se as a cofactor, were reduced, especially in those who were on hemodialysis.28 However, while supplementation of Se in such cases significantly increased Se levels, its impact on GSH-PX levels was modest, the only means of adequately restoring them in these subjects being via kidney transplantation.28 In an experimental study, when Se was supplemented in the form of sodium selenite to diabetic rats, glucose uptake and its turnover, which is usually decreased in diabetic rats, showed a statistically significant increase.29
Thus, there is some evidence suggesting that Se plays an insulin-like mediating role in diabetes by tending to normalize the altered activities of glucose metabolizing enzymes and improving glucose uptake and metabolism by the liver.30
Recently, it was reported that selenocysteine-β-lyase (Scly), a protein that mediates Secys degradation and relates selenoproteins to energy metabolism, may have an effect on obesity and MS development triggered by high-fat exposure.31 Scly knock-out mice developed MS and had normal inflammatory cytokines and expression of selenoproteins, except of SEPP1.32 Thus, disruption of Scly favors obesity independent of the expression of most selenoproteins.32
Selenium and the Thyroid
While the immune system acts to protect the host from infectious agents and from other noxious insults that exist in the environment, an abnormal immune response to autoantigens that express on the thyrocytes in genetically susceptible persons can trigger AITD. The most frequent AITD is HT,33 which is more common in females and increases with age,34 with primary hypothyroidism being a frequent consequence of HT.
Se is copiously present in the thyroid gland. Inflammatory thyroid follicular cells produce a large number of cytokines, such as Th1, which play a crucial role in the pathogenesis of AITD. Se supplementation in rats is likely to regulate the expression of Th1/Th2 cytokines and alleviate AITD.36 The selenoprotein DIO comprises enzymes that convert the depot thyroid hormone thyroxine to the active triiodothyronine and inactive reversetriiodothyronine. 36 During the process of conversion, hydrogen peroxide, a toxic byproduct, is generated, which is neutralized by GPX thus preventing oxidative stress, thyroid cell damage, and disease. However, interventional clinical trials have provided somewhat controversial results regarding the efficacy of organic and inorganic Se compounds, in the form of selenomethionine (SeMet) and selenite, respectively, in patients with autoimmune thyroiditis.37,38 Se-enriched yeast (Se-yeast) is another form of Se supplementation.39 With elucidation of the manner in which speciation of Se-yeast is achieved, it was revealed that SeMet is the largest single species, making up 54–74 % of total Se. Moreover, it has been determined that Se-yeast is capable of elevating selenoenzymes activity and that its bioavailability is higher than that of inorganic Se.39 It consequently translates as an effective, safe (no toxicity has been observed), and natural source of dietary Se, though attention must be paid to the variability of Se content of various diets in accordance with food items, their geographical origin, and the corresponding soil Se content.40 In future prospective trials, the stage of disease, the iodine baseline state, and the form of the Se compound should be given particular consideration.
While Se administration has proved efficacious in patients with mild thyroid-associated opthalmopathy,41 it has also recently been shown that low serum Se levels are associated with increased incidence of thyroid disease and that increased Se intake lowers the risk for disease.42 It therefore seems evident that Se status could well be a valuable marker of thyroid disease risk.
Selenium and Chronic Inflammation
Persistent acute inflammation may suppress hypersensitivity, heighten susceptibility to toxic agents, and increase bodily requirements of Se. Such a condition will inevitably derange the body’s “selenostasis,” creating a state of Se deficiency as well as causing chronic inflammatory disorders and the so-called “immune tsunami” that characterizes several various autoimmune diseases.43
Reactive oxygen species (ROS) formed during oxidative stress can damage normal cellular function by initiating lipid peroxidation and also cause DNA strand breaks. Routine clinical procedures, such as coronary bypass surgery and thrombolysis, are typically followed by generation of ROS, this being associated with poor cardiac recovery postischemia. Furthermore, ROS have recently been reported to be strongly involved in infarction, necrosis, apoptosis, arrhythmogenesis, and endothelial dysfunction following ischemia-reperfusion.44 Meanwhile, increased production of ROS necessitates high requirements of nutrients involved in antioxidant defenses, and notably Se and zinc. In this context, constant dietary Se supplementation may provide a safe and convenient method for increasing antioxidant protection in patients at risk for ischemic heart disease or in those undergoing clinical procedures involving transient periods of myocardial hypoxia.45
In a study of 77 patients in whom, after cardiopulmonary resuscitation (CPR), Se levels were found to be lowered and to be inversely correlated with the clinical outcome, administration of Se exerted anti-inflammatory effects and prevented microparticle-mediated endothelial dysfunction.46 Endothelial dysfunction is, moreover, a pivotal feature of postcardiac arrest syndrome a life-threatening state with high mortality, which is also driven by microparticles in resuscitated patients. Interestingly, Se administration diminished intercellular adhesion molecule-1- and vascular cell adhesion molecule-1-mediated monocyte adhesion induced by microparticles of CPR patients.46 Of note, the fact that Se declines in plasma of patients following CPR could possibly identify it as a novel predictive marker of intensive care unit mortality.
Viral and bacterial infections are often associated with low Se, among deficits of other macronutrients and micronutrients. For example, benign Coxsackie and influenza virus strains can mutate to highly pathogenic strains in the presence of Se deficiency.47 On the other hand, multimicronutrient supplements containing Se have been shown to improve a number of clinical and lifestyle variables in patients simultaneously infected with HIV and mycobacterium tuberculosis. The hypothesis in the latter case is that boosted Se status may upgrade the function of both adaptive and innate immunity cells.47
Se administration is thought to be beneficial to patients suffering from viral diseases. In an experimental study, the effects of Se supplementation on serum Se concentrations, mortality rate, lung virus, and cytokine titers were measured in mice inoculated with suspensions of the influenza virus.48 Basal Se levels were seen to be of major importance as mortality amounted to 75 % in Se-deficient mice compared with a 25 % mortality rate in the mice supplemented with 0.5 mg Se/kg in the form of sodium selenite. These results suggest that Se may represent a valid intervention approach for strengthening the immune response to lethal influenza virus infection.48
Other potential targets for intervention via Se supplementation are gut epithelial barrier dysfunction, innate immune activation, inflammation, and other factors that may independently predict mortality in individuals with treated HIV infection with a history of AIDS.49 It is important to note, however, that although single and multiple micronutrient deficiencies are common among HIV-infected adults pre-antiretroviral therapy (ART) initiation, their levels are likely to vary between countries50. Thus, in PEARLES, a ninecountry trial, it was investigated whether the prevalence and multiple micronutrient deficiencies among HIV-infected adults changed 48 weeks post-ART initiation.50 Importantly, despite increases in micronutrient concentrations, prevalence of individual deficiencies remained largely unchanged after 48 weeks on ART. Among all micronutrients, Se deficiency (53 %) has the highest prevalence among these patients. The above results suggest that ART alone is not sufficient to improve micronutrient deficiency.50 Working on the basis of these findings, a randomized clinical trial (RCT) demonstrated that 24-month Se supplementation significantly increases the rate of CD4 cell count, which is known to decline among ART-naive patients.51
Se may provide protection against viral diseases, as was first shown in the successful treatment and prevention of Keshan disease, a form of myocarditis mainly occurring in regions of China that are characterized by very low Se levels.52 It is well known that a cofactor in the etiology of Keshan disease is Coxsackievirus B3 (CVB3), which is thought to become more virulent in a low Se environment.53–55
Furthermore, conditions of oxidative stress sustained by an Se-deficient state are likely to genetically influence the virus, causing mutations that further impair immunity, thus promoting and exacerbating chronic
Se concentrations were reduced in critically ill patients, presumably due to Se consuming oxidative stress, and particularly in patients with sepsis.57 Thus, oxidative stress might be a result of suboptimal Se and zinc concentrations.57 Se supplementation may restore Se levels and increases shedding of L-selectin from monocytes while decreasing soluble L-selectin, which has been reported to be associated with high mortality in patients with sepsis.58 However, there is currently some uncertainty regarding its efficacy, as major questions remain unanswered concerning dose, duration of supplementation, and species of Se administered.
Recently, serum Se levels were seen to be drastically decreased in alcoholic liver disease patients, this accompanied by low GPX activity.59 The serum Se/malondialdehyde (MDA) ratio, as a marker of oxidative stress, has been proposed as an indicator of hepatic damage caused by alcohol consumption and, notably, Se substitution emerged as a possible antioxidant therapy in these patients.
In the course of inflammatory bowel diseases (IBDs), absorption disorders, and consequent malnutrition results in trace element deficiencies, and especially of Se, thus impairing the body’s antioxidant protection system.60 It thus seems intuitive to propose that a nutrition therapy for IBD that would include Se, energy-protein nutrients, and other trace elements could well prove beneficial.60
In acute pancreatitis, a disease with a high mortality rate, Se support remains controversial due to several factors, such as lack of agreement as concerns mode of administration and classification of the disease.61 However, in chronic pancreatitis, as was shown in a recent meta-analysis, combined antioxidant (Se, β-carotene, vitamin C, vitamin E, methionine) therapy was seen to be associated with pain relief and thus could improve the quality of life of these patients.62
Selenium and Mechanisms of Inflammation
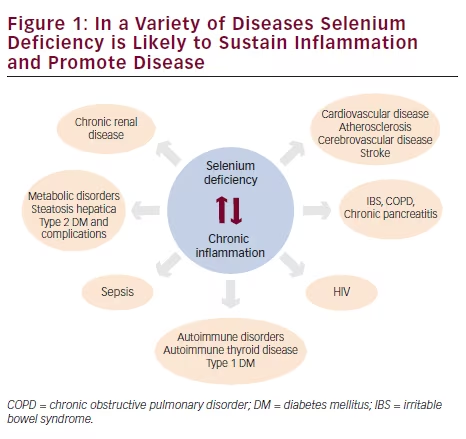
Chronic inflammation leads to Se deficiency, which intensifies the persistence of the inflammatory condition and, by creating a vicious cycle, promotes the manifestation of disease. In a state of low Se, cells create a selenoprotein hierarchy, since they utilize Se to synthesize the selenoproteins most important to them.63 The liver, the central organ for Se regulation, produces excretory Se forms to regulate whole body Se. It responds to Se deficiency by curtailing excretion and secreting SEPP into the plasma at the expense of its intracellular selenoproteins availability. Meanwhile, studies have indicated that supranutritional Se increases proliferation as well as differentiation of naive CD4-positive T lymphocytes toward T helper 1 cells, thereby enhancing acute cellular immune response (see Figure 1).64,65
Two endoplasmic reticulum (ER) transmembrane selenoproteins that have been associated with inflammation and immunity, SELK and SELS, have recently been proposed as playing a role in protecting cells during conditions that lead to ER stress.66 With regard to SELS, its role is likely to be linked to its function in retrograde translocation of misfolded proteins from the ER.67 Besides playing a pathologic role in various diseases, chronic inflammation can be caused by both genetic and environmental factors.68 Genetic variation in SELS, which is strongly involved in stress response in the ER, showed that one promoter variant, -105G-A, was associated with the cytokines interleukin (IL)-1β, IL-6, and tumor necrosis factor alpha (TNF-α) and that functional analysis of this polymorphism revealed SELS impairment following exposure to ER stress agents.67 Moreover, suppression of SELS in macrophages led to increased release of IL-6 and TNF-α, indicating that SELS is involved in mediating inflammation.67
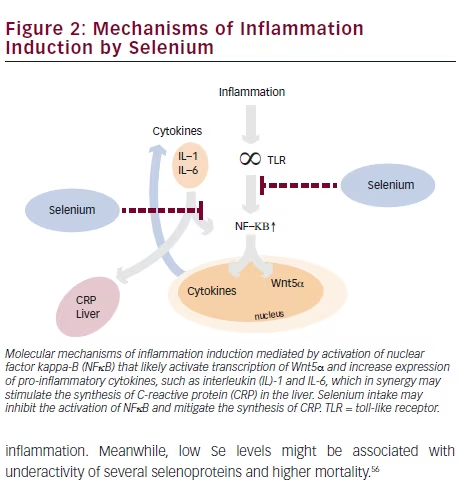
The nuclear factor kappa-B (NFκB) signaling pathway has been associated with enhanced inflammatory response, while its activation has been significantly correlated with IL-6 and TNF-α production.69 It is hypothesized that Se may be capable of inhibiting the activation of NFκΒ by modulating selenoprotein gene expression. In chronic inflammation, Se supplementation has been shown to restore the depleted hepatic and serum Se levels by increasing selenoprotein biosynthesis, this
leading to suppressed C-reactive protein (CRP) production thereby attenuating the inflammatory process.70 Moreover, Se downregulates the expression of nuclear transcription NFκB, lipoxygenase, cyclooxygenase, 5-lipoxygenase-activating protein, and receptor for leukotriene B4.71 By decreasing the production of ROS and suppressing the activation of NFκBmediated transcription of pro-inflammatory mediators, Se leads to the downregulation of the leukotriene pathway and attenuates inflammation such as in diabetic cardiac hypertrophy.71
At the epigenetic level, an investigation was made into the effect of Se supplementation in the form of selenide on histone acetylation in models of inflammation.72 Se inhibits histone H4 acetylation in macrophages, thus confirming a role of Se in downregulating inflammatory gene expression.72
These mechanisms may explain the modulatory effects of an increased Se status on the inflammatory response (see Figure 2).
The ability of Se to act as a free radical scavenger could prove useful in counteracting the generation of hydroxyl radicals and subsequent formation of insoluble parafibrin from soluble plasma fibrinogen.73 The formation of a large protease resistant polymer formed by the hydroxyl radical interaction with fibrinogen in inflammation, which, if remaining long in the circulation, promotes a state of chronic inflammation, is due to the catalytic participation of trivalent iron (Fe3+), not of divalent (Fe2+), and to the attraction of cytotoxic albeit ineffective T cells.74 It should at this point be borne in mind that any pathologic condition involving the damage of erythrocyte membranes, as for example in infections and following exposure to environmental toxins, is likely to contribute to an excess of the body’s storage of trivalent iron. Certainly, it remains to be studied whether Se supplementation, as a free radical scavenger, with or without iron-chelating agents, may prevent iron overload and persistence of fibrin-like deposits, thus preventing cancer and cardiovascular and degenerative disease.
Future Perspectives
There is a considerable amount of evidence to support Se administration, alone or in combination with other antioxidants, in chronic inflammatory diseases, such as chronic pancreatitis and AIDS. Though inflammation decreases Se levels, it is necessary to emphasize that, whereas Se supplementation may benefit patients with low Se status, it should not be recommended to those with adequate-to-high status since administration could affect them adversely, notably causing selenosis, which is characterized by a variety of symptoms, including nausea, vomiting, abdominal pain, diarrhea, hair loss, brittle nails, and peripheral neuropathy
Existing data suggest that Se administration could be beneficial in an array of metabolic and autoimmune diseases that are related to inflammation. More specifically, there is potential for future use of Se in several autoimmune diseases, applying it alone in a personalized model while utilizing more sophisticated and targeted formulations, which will offer the advantage of higher efficacy and a lower rate of side effects. However, controversies as yet exist regarding choice of Se species, dosage, and duration of administration.
Therefore, much work still needs to be carried out, with the performance of large RCTs being required in order to establish whether Se supplementation in chronic inflammatory states may have a positive impact on disease progression and mortality







