Basal insulin therapy is recommended as a first-line injectable therapy in persons with type 2 diabetes who do not respond to metformin monotherapy. Insulin works to control fasting glycaemia, and it is expected that metformin (with or without other drugs) will suffice apropos post-prandial euglycaemia. In many patients, however, basal insulin is unable to achieve adequate glycaemic control. This has been termed basal insulin failure. However, in the light of newer developments, we suggest a more appropriate term, basal insulin inadequacy, and discuss how it can be used.
Basal Insulin Failure
According to the current American Diabetes Association (ADA)/ European Association for Study of Diabetes (EASD) guidelines, change of basal insulin therapy is indicated if the treatment strategy fails to achieve normal glycated haemoglobin (HbA1c) in spite of adequate fasting control, or if >0.5 μ/kg/day of basal insulin is required.1 Basal insulin failure has earlier been defined as the inability to achieve a pre-decided target glycaemic control, after optimisation of lifestyle modification measures and maximal titration of basal dose beyond which unacceptable hypoglycaemia will occur.2
Basal Insulin Inadequacy
The current nomenclature implies that basal insulin has ‘failed’, while actually it may have succeeded in achieving fasting euglycaemia. Thus a more appropriate terminology is ‘basal insulin inadequacy’. This conveys a more accurate message that basal insulin is inadequate for the particular patient’s need. Inadequacy avoids the negative connotation associated with the word ‘failure’. It does not pass judgmental opinion on either the patient’s efforts to manage lifestyle or the physician’s choice of therapeutic strategy.
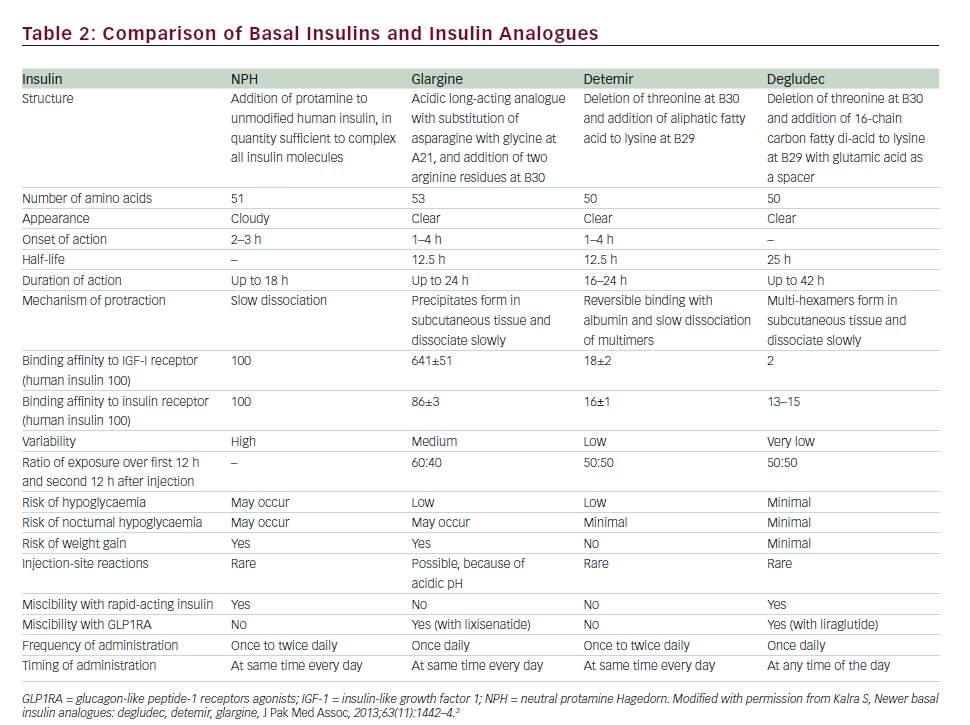
Basal Insulin Dissimilarity
All basal insulins are not alike. Each basal insulin and basal analogue has a unique structure, which contribute to specific pharmacokinetic and

pharmacodynamic characteristics.3 These properties allow a systematic listing of basal insulin as intermediate-, long- and ultra-long-acting molecules (see Tables 1 and 2). The differences in duration of action, glycaemic variability and risk of hypoglycaemia, specifically nocturnal hypoglycaemia, may allow for substitution of one basal insulin for another, in case adequate control is not achieved with a particular preparation. Thus, a new strategy for intensification of therapy is available for persons not responding to basal therapy: a switch to a longer-acting basal insulin.
Such a therapy is supported by mechanistic studies, randomised controlled trials (RCTs) and meta-analysis. Insulin degludec, for example, has been shown to have a longer half-life and duration of action, with significantly less glycaemic variability than glargine.4
RCTs and meta-analyses report a significantly lower incidence of hypoglycaemia and nocturnal hypoglycaemia, while achieving better fasting glucose control, in persons randomised to insulin degludec compared with glargine.5–8 Refractory patients, switched from glargine to degludec, have also been reported to achieve good glycaemic control in clinical practice.9,10 Cost-effectiveness of such a shift is also found to be beneficial.11,12 Thus, the clinical phenomenon of ‘basal insulin inadequacy’ may be drug-specific. Inability of a particular basal insulin to achieve adequate glycaemic control does not imply that all basal insulins will be inadequate for the purpose. Applied to a patient not responding to glargine, the phrase ‘basal insulin inadequacy’ may not be valid for all basal insulins.
We therefore suggest the following terminology and definitions:
- Basal insulin inadequacy may be defined as the inability of all
basal insulin preparations, prescribed alone or in combination
with various oral glucose-lowering drugs, to achieve pre-decided
glycaemic targets, without causing unacceptable hypoglycaemia
or weight gain, in spite of optimal lifestyle modification and
maximal dose titration. - Intermediate-, long-acting and ultra-long-acting insulin inadequacy
may be used to describe persons who do not respond to maximal
doses of NPH, glargine and detemir and degludec, respectively.







