Gestational diabetes mellitus (GDM), defined as hyperglycaemia identified after the first trimester of pregnancy that is not clearly overt diabetes, impacts approximately 7% of births in the USA; a percentage that has increased in parallel with the prevalence of both obesity and type 2 diabetes over the past 20 years.1–4 The impacts of GDM include hypertensive disorders of pregnancy, abnormal fetal growth and associated difficulties with delivery, as well as longer-term maternal and offspring morbidity.5–11
In the USA, there are significant gaps between the recommended care during and after pregnancy, and what patients actually receive. While it is difficult to estimate the percentage of patients that have morbidity attributable to uncontrolled GDM, the estimated cost of each case of GDM in pregnancy is nearly $5,800, some of which is potentially preventable with good treatment (such as the costs associated with newborn admission to the neonatal intensive care unit, inpatient and emergency department utilization).12,13 There are also well-documented gaps between what is recommended after pregnancy and what actually occurs. As an example, because a substantial proportion of women with GDM will progress to type 2 diabetes after pregnancy, screening for type 2 diabetes is both indicated and recommended.14 Nonetheless, fewer than 40% of patients are screened after pregnancy.15,16 What is at the root of the gap between what is known and what is currently being achieved for patients?
One contributor to the variability in care for GDM may be the lack of clinical consensus that is evident in differing professional society guidelines.1,17–19 While there is broad agreement that providers should screen for GDM,20–22 there is no consensus on optimal screening, diagnostic criteria, treatment, or post-delivery screening. In this review, we will focus on these ongoing controversies. We will discuss screening for GDM in detail, including the benefits and drawbacks of one- and two-step strategies. We will also review treatment options, focusing on the literature for and against oral hypoglycaemic agents as alternative treatments to insulin. Finally, we will highlight limitations in current practice after delivery, with potential solutions, as well as novel approaches to screen for type 2 diabetes after pregnancy with GDM.
Identifying gestational diabetes
GDM is defined as “diabetes diagnosed in the second or third trimester of pregnancy that was not clearly overt diabetes prior to gestation.”1 While all professional organizations agree on this general definition, there is ongoing controversy regarding specific diagnostic strategies. Both one- and two-step testing strategies have been endorsed, with the World Health Organization (WHO), the International Association of Diabetes in Pregnancy Study Groups (IADPSG), and the American Diabetes Association (ADA) endorsing the use of a 75 g oral glucose tolerance test (OGTT) in the late second trimester as the preferred strategy.1,22,23 The American College of Obstetricians and Gynecologists (ACOG) continues to prefer a two-step strategy, recommending universal screening with 50 g glucose-loading test (GLT)followed by targeted diagnostic testing with 100 g OGTT (Table 1).1,18,22,23 Both one-and two-step strategies have benefits, as well as limitations.
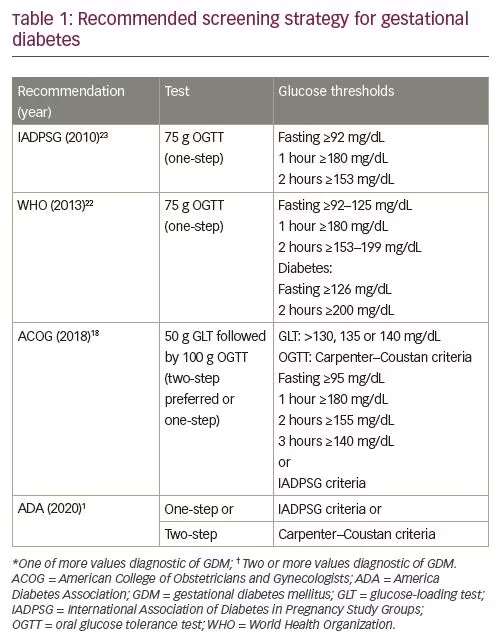
Historically, GDM was defined by risk for long-term adverse maternal outcomes.24 Following several observational studies demonstrating foetal risks associated with varying degrees of glucose intolerance, there was broad agreement that GDM should be defined by glucose thresholds at which the risk for adverse outcomes would increase.25 There was also recognition that level 1 evidence supporting treatment of ‘mild’ GDM with lifestyle counseling and insulin was evident from two randomized controlled trials which used differing diagnostic criteria for study entry. 5,6 Crowther and colleagues studied more than 900 participants in Australia and New Zealand, including participants with GDM diagnosed with a 75 g OGTT at 24–34 weeks by WHO criteria.6 A similar study, performed in the USA, included nearly 900 participants in weeks 24–31 of gestation identified using two-step testing, first screened with 50 g GLT and then using a 100 g OGTT with the Carpenter–Coustan criteria to diagnose GDM.5,26 Both studies excluded participants with elevated fasting glucose or post-load glucose levels consistent with overt diabetes.
The Hyperglycemia and Adverse Pregnancy Outcomes (HAPO) study attempted to define glucose thresholds for adverse pregnancy outcomes.27 In this international, multicenter, observational study of more than 25,000 pregnant participants, investigators administered a 75 g OGTT between 24–32 weeks gestation and blinded clinicians to the results unless the participant was overtly hyperglycaemic (defined as fasting plasma glucose >105 mg/dL or 2-hour plasma glucose >200 mg/dL). The results demonstrated a linear association between maternal glycaemic levels with both maternal and foetal outcomes, leaving a clear diagnostic threshold for GDM uncertain. The IADPSG subsequently proposed diagnostic criteria for GDM based on the glycaemic thresholds that were associated with an increased odds of 1.75 for adverse outcomes, recommendations which were quickly endorsed by the ADA.23,28 The ACOG, however, continued to endorse a two-step strategy, based on concerns regarding the lack of data supporting intervention at these lower cut-offs, as well as healthcare delivery implications of diagnosing more cases of GDM, concerns that were validated and ultimately echoed by the National Institutes of Health (NIH) after a 2013 consensus conference, when it was concluded that there was “insufficient evidence to adopt a one-step approach.”18,29 It is important to note that while GDM prevalence varies across populations, there are consistently more patients identified with a one-step IADPSG testing strategy than with a two-step strategy, in some populations increasing the absolute incidence more than threefold.30,31
Proponents of one-step testing note that two-step testing misses a group of patients who are only identified by using a 75 g OGTT with the IADPSG thresholds and are at similar risk for adverse pregnancy outcomes as those diagnosed using the two-step method.32 A cost-effectiveness analysis estimated that adoption of universal screening using IADPSG thresholds would prevent 85 cases of shoulder dystocia, 262 cases of pre-eclampsia, and 688 cases of future diabetes for every 100,000 pregnancies that were screened.33 Screening and diagnosis can be universally implemented using a one-step approach, avoiding incomplete assessment after abnormal 50 g GLT, and expediting treatment. Those who adopted one-step testing strategies also cite broad acceptance by patients, some of whom prefer a test that is less than 3 hours.34 Of note, the majority of health systems that adopted the IADPSG guidelines are outside the USA.31
Following the NIH consensus conference, obstetric providers in the USA largely continued two-step screening and diagnosis, or returned to this strategy.1,18,29 Arguments for continuing the two-step approach are primarily centered on healthcare delivery considerations. The 50 g GLT can be administered regardless of oral intake prior to visit, while the one-step 75 g OGTT must be administered after a fast; an estimated 15% of women in the USA have inadequate prenatal care, highlighting the need for a test that can be administered at any visit.35 The impact of GDM diagnosis is also not entirely beneficial. While treatment at Carpenter–Coustan thresholds did not change the rate of caesarean delivery in a randomized controlled trial setting, observational data comparing caesarean delivery rates in patients with GDM diagnosed at differing thresholds in a single institution suggest that just the diagnosis of GDM may be associated with morbidity in real-world settings.5,36 There is a substantial burden of diagnosis and treatment to patients, with pain and social isolation identified as themes in qualitative studies.37–39 Finally, the cost of additional cases is significant; universal screening with a one-step strategy utilizing the IADPSG thresholds costs an estimated $20,336 per quality-adjusted life year gained, and is cost effective only when post-delivery care reduces the incidence of type 2 diabetes.33
Therefore, while there is agreement that treatment of GDM has benefits, the lack of a clear threshold for increased risk of adverse pregnancy outcomes, the lack of level 1 evidence supporting treatment at IADPSG thresholds, and the need to balance the additional diagnoses of GDM at IADPSG thresholds with the burden of diagnosis to both patients and the healthcare system, has left providers without consensus guidelines. A recent study compared pregnancy outcomes in over 20,000 pregnant women randomized to diagnosis of GDM by a one-step approach and criteria versus diagnosis by a two-step approach and criteria. This study showed no difference in pregnancy outcomes of the one-step versus the two-step groups, despite about twice as many more women receiving a diagnosis of GDM when the former was used.40 It is too soon to tell if this study will lead to greater consensus in the approach to the diagnosis of GDM.
Treating gestational diabetes
Insulin remains the pharmacologic agent of first choice to treat hyperglycaemia in women with GDM, as recommended by the ADA, ACOG, and Diabetes Canada, when lifestyle changes fail to achieve glycaemic goals.18,41,42 These recommendations are based on the two trials that established the value of treating mild GDM to improve perinatal outcomes using insulin when lifestyle changes were insufficient to achieve optimal glycaemic control.5,6 Insulin does not cross the placenta and has been used during pregnancy for decades without adverse effects on the neonate/offspring.17,41,43–45 Insulin analogs (including aspart, lispro, detemir, and glargine) have also been used safely in pregnancy, and they are now used more frequently in pregnant women with diabetes than human insulin.46–49 Insulin pumps have been safely used in GDM, but are rarely needed in this population.50 To date, there are no head-to-head studies demonstrating the superiority of one insulin regimen or type of insulin over another.51 Some concern has been raised about the use of glargine, which has increased affinity for the insulin-like growth factor (IGF)-1 receptor and increased mitogenicity when compared with other insulins, and therefore might be associated with greater risk of large for gestational age birthweight.52 However, several studies, including a systematic review and meta-analysis, have not supported this concern.53–56
Controversy regarding optimal insulin type or regimen results from a lack of powered head-to-head studies comparing insulin type and insulin regimen. At present, given that the data on insulin analogues support their effectiveness and safety, the type of insulin and regimen used will depend on the ordering clinician and the individual patient, with the goal of achieving optimal glucose goals for pregnancy.
There has been great interest in whether oral medications that reduce glucose levels could be used safely and effectively in women with GDM to avoid the need for insulin injections. The two oral agents used most commonly in the treatment of GDM are metformin and glyburide. In a study of medications for diabetes that looked at initiation in late pregnancy (a surrogate for GDM diagnosis), insulin was the most commonly prescribed drug in international prescription databases derived from seven countries and three continents, but metformin use grew over time. During the study period from 2006 to 2016, metformin was the most common alternative to insulin in every country but the USA, where glyburide was more commonly prescribed as an alternative to insulin.48 A study of USA commercial pharmacy claims in women with GDM found that from 2000 to 2011, glyburide use increased from 7% to 65%, surpassing insulin as the most common treatment as of 2007.57 Large, multicenter, randomized trials have demonstrated that metformin, but not glyburide, is non-inferior to insulin for pregnancy outcomes.58,59 Because of concerns for both the efficacy of and potential for long-term impact on children exposed in utero, insulin remains the recommended first-line agent for many organizations.17,18,42 Others recommend metformin, recognizing that not all women with GDM who need pharmacologic therapy will be able, or willing, to use insulin.19
Metformin
Metformin is a biguanide and decreases hepatic glucose production and promotes glucose uptake by peripheral tissues.60,61 The mechanisms of action in lowering blood glucose remains incompletely understood, but it is one of the few agents used to lower blood glucoses that does not cause hypoglycaemia. Metformin is not recommended for use in individuals with renal or hepatic impairment, due to concern for accumulation leading to lactic acidosis, which is a rare complication, but accordingly, the ADA cautions that metformin should not be used in women with placental insufficiency in such conditions as hypertension, pre-eclampsia, or at risk for intrauterine growth restriction.17,61 Metformin crosses the placenta, with levels in the foetal circulation similar to maternal levels at delivery.62,63 Nonetheless, because of the maternal safety profile, as well as its low cost, the use of metformin in GDM is increasing.
In the Metformin in GDM (MiG) trial, a multicenter study performed in New Zealand and Australia, 751 women with GDM diagnosed by 75 g OGTT who needed pharmacologic treatment between 20 and 33 weeks + 6 days gestation were randomized to insulin or metformin starting at 500 mg, titrated to a maximal dose of 2,500 mg daily.58 The primary outcome, a composite of neonatal complications, was similar in both treatment groups, although 46% of the participants in the metformin group required the addition of insulin to reach glycaemic goals. Participants in the metformin group experienced less hypoglycaemia and gained less weight (0.4 ± 2.9 kg versus 2.0 ± 3.3 kg; p<0.001) over the course of pregnancy than those on insulin. The rate of preterm birth before 37 weeks’ gestation was nearly twofold higher in the metformin group compared with the insulin group (12.1% versus 7.6%; p=0.04), driven primarily by spontaneous, not indicated, preterm delivery. The mechanism by which metformin might lead to spontaneous preterm delivery is not known.
Several meta-analyses have subsequently compared metformin to insulin. Rates of macrosomia were reported to be similar or lower with metformin, with lower rates of neonatal hypoglycaemia than insulin.64,65 In addition, metformin use has been associated with less maternal weight gain and lower risk for preeclampsia than insulin.64,66 As seen in the MiG trial, there was a higher risk for spontaneous preterm birth with metformin than insulin.64,66
A limited number of infants exposed to metformin for treatment of GDM have now been followed up to 9 years of age.67,68 At 7 years of age, there were no differences in body composition or metabolic measures in the offspring of mothers treated with metformin versus insulin. However, at 9 years of age, the metformin-exposed offspring had significantly higher weight, body mass index and waist circumference than those exposed only to insulin.68 Longitudinal studies are ongoing, with concern for adverse metabolic findings in the offspring informing recommendations regarding the use of metformin for the treatment of GDM.69
Glyburide
Glyburide, also called glibenclamide, is a second-generation sulfonylurea which lowers glucose levels by stimulation of insulin release by the beta cells in the pancreas.70 Both ACOG and ADA recognize glyburide as an alternative medication for women who cannot, or will not, use insulin and cannot tolerate metformin.17,18
In a large, multicenter, randomized trial performed in France, 914 women with GDM diagnosed by one-step testing were randomized to glyburide (starting at 2.5 mg, titrated up to 20 mg/day as needed) or insulin analogue (starting at 4 U/day, up to 20 U/day) with intermediate-acting insulin as needed, in a non-inferiority study.59 The primary endpoint, which was a composite neonatal outcome that included macrosomia, neonatal hypoglycaemia, and hyperbilirubinaemia, did not differ between the two groups. However, neonatal hypoglycaemia was more common in infants whose mothers were randomized to glyburide than in those who were randomized to insulin (12.2 % versus 7.2%; 95% confidence interval [CI] 0.5–9.5%; p=0.02). As a result, the study concluded that glyburide did not demonstrate non-inferiority to insulin for the treatment of GDM. Additionally, both efficacy and safety were not equivalent; 18% of the women randomized to glyburide had to crossover to insulin after failure to achieve glycaemic control, and maternal hypoglycaemia was more common in the glyburide group than in the insulin group (28.8% versus 3.5%; p<0.001).
Several meta-analyses support the findings of the French trial, including higher risk for both macrosomia and neonatal hypoglycaemia with glyburide or glibenclamide than with insulin.64,66,71 Unlike metformin, longitudinal follow-up data in offspring of pregnancies exposed to glyburide have not been reported. To date, there are no large multicenter randomized trials of metformin versus glyburide that are adequately powered to demonstrate a difference or non-inferiority on pregnancy outcomes in the treatment of GDM. Similar to findings when compared with insulin, both small studies and meta-analyses have demonstrated higher rates of maternal hypoglycaemia and fetal macrosomia associated with glyburide or glibenclamide than with metformin.65,71,72 These studies have also demonstrated less maternal weight gain associated with metformin than with glyburide.65,66,71,73 A major limitation of both agents is the failure to achieve glycaemic control in many women with GDM.74
Other oral hypoglycaemic agents
There are other classes of agents which lower glucose levels and have been approved for use in type 2 diabetes, including dipeptidyl peptidase-4 inhibitors, glucagon-like peptide 1 agonists, and
sodium–glucose cotransporter 2 inhibitors.41 These agents have effects on many organ systems and have not been studied in pregnancy; therefore, they should not be used for the treatment of GDM. In patients with type 2 diabetes outside of pregnancy, the potential benefit of these therapies to reduce the incidence of cardiovascular and renal comorbidities of diabetes guides choice of therapy. These long-term considerations relevant for ongoing management of diabetes do not apply to GDM, in which the benefits of treatment are limited to improving pregnancy outcomes and avoiding adverse offspring effects.
Professional society recommendations for medications to treat gestational diabetes
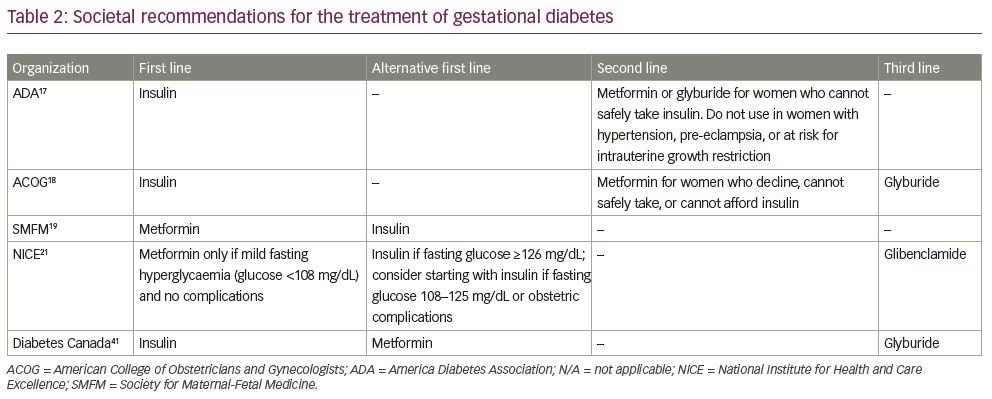
Based on available data, several professional societies have made recommendations for the treatment of GDM; however, there are differences in approach across organizations, stemming from the ongoing controversy surrounding the efficacy and safety of oral agents (Table 2).17–19,21,41 The ACOG, ADA, and Diabetes Canada all recommend insulin as the first-line agent, based on data supporting the ability to achieve glycaemic control and improve pregnancy outcomes, the long history of use without report of adverse offspring effects, and lack of placental transfer.17,18,42 The Society for Maternal-Fetal Medicine (SMFM) recommends that metformin also be considered as a first-line agent for the treatment of GDM.19 Following the SMFM guidelines in particular, a cautionary response from a group of GDM experts highlighted the lack of long-term offspring follow-up data after metformin exposure in utero.69 The National Institute for Health and Care Excellence in the UK recommends metformin as a first-line agent only in the setting of mild fasting hyperglycaemia, with insulin for more severe hyperglycaemia or in the event of obstetric complications.21 Diabetes Canada recommends metformin as an alternative first-line agent, but makes note of the lack of long-term offspring follow-up.41 Other societies recommend metformin only when a patient is unable or unwilling to take insulin.17,18
Screening for type 2 diabetes after pregnancy with gestational diabetes
Women with GDM are at high risk for diabetes, particularly in the first several years following pregnancy.14 The ADA and ACOG recommend diabetes screening at 4–12 weeks postpartum, with repeat screening every 1–3 years thereafter depending on results.17,18 In clinical practice, screening rates are poor. Several groups have identified this gap after pregnancy; despite standardized recommendations for postpartum screening, a recent meta-analysis concluded that fewer than 40% of patients are screened within the recommended time frame, with no change in rates at 6 months or 1 year after pregnancy affected by GDM.16 There have been attempts to mitigate provider barriers to screening, which include fragmented maternal care after delivery; solving the problem will require multi-pronged approaches.75–79
Given the suboptimal rates of screening for diabetes after pregnancy complicated by GDM, there is controversy over which test should be performed postpartum, as well as the timing of the testing. Currently, the ADA recommends testing of women with recent GDM with a 75 g OGTT at 4–12 weeks postpartum.17 On the other hand, ACOG recommends either a fasting blood glucose or a 75 g OGTT at 4–12 weeks postpartum, recognizing that a fasting glucose is easier to complete, but that the OGTT provides more information.18 Diabetes Canada recommends a 75 g OGTT after pregnancy, as late as 6 months postpartum.42 There is consensus that although glycated haemoglobin (HbA1c) may identify some women with overt diabetes, it is an insufficient test for postpartum screening.17,18
One strategy to improve screening rates is to test immediately after pregnancy, during the delivery hospitalization. This timing may be important particularly in the USA, where as many as 40% of women do not attend a postpartum visit.80 Several groups have investigated whether screening during the delivery hospitalization might identify patients at highest risk for type 2 diabetes in the postpartum period, and thereby improve efforts to target lifestyle modification.81,82 A combined patient-level analysis of four studies with a total of 319 participants compared results of a 75 g OGTT during delivery hospitalization with results at 4–12 weeks postpartum.83 While only 52% of study subjects returned for follow-up testing, in those who did return, none of the participants with normal initial testing went on to be diagnosed with type 2 diabetes at the postpartum visit. In-hospital glucose testing had 50.0% sensitivity (95% CI 11.8–88.2%) and 95.7% specificity (95% CI 91.3–98.2%), with 98.1% negative predictive value (95% CI 94.5–99.6%). Taken together, these results suggest that in-hospital testing with 75 g OGTT after delivery has the potential for risk stratification, helping to identify those at lowest risk for type 2 diabetes in the immediate postpartum period.
There is broad recognition that a diagnosis of GDM represents a window of opportunity to target interventions aimed at reducing long-term risk for diabetes.8,84,85 Future work should focus on implementation strategies, including local process improvements to achieve optimal rates of postpartum screening.
Conclusions
GDM is one of the most common complications of pregnancy and confers lifelong risks to both women and their children. Nonetheless, there remains a lack of consensus on the best strategies to improve both short- and long-term outcomes. Because rigorous observational data demonstrate a linear association between maternal glycaemic parameters and risks for adverse pregnancy and offspring outcomes, the diagnostic criteria remain controversial. Treatment with insulin is effective, but costs and patient experiences limit use in clinical practice. Glyburide has drawbacks, including both efficacy and safety. While metformin is increasingly used for many indications in reproductive-aged women, use as a first-line agent for GDM remains controversial due to transplacental passage and limited long-term follow-up data. Finally, new approaches are needed for screening for type 2 diabetes after pregnancy to leverage the window of opportunity presented by pregnancy. Future work in the field should include studies of both clinical and implementation outcomes, examining strategies to improve the quality of care delivered to women with GDM.







