Unmet needs for glucose monitoring in people with diabetes mellitus using intensive insulin
Glucose monitoring is essential for people using intensive insulin to inform adjustments to the treatment and management of diabetes. Evidence suggests many people do not adhere to the recommended testing frequency with self-monitoring of blood glucose (SMBG). In the USA, it was estimated that people using intensive insulin test their glucose on average 2.6 times/day.1 This contrasts with the American Diabetes Association (ADA) Standards of Care, which recommend that people using intensive insulin regimens should test 6–10 times (or more) daily.2 Inadequate adherence to SMBG testing is associated with poor long-term outcomes,3 which in turn, is likely to have a considerable economic impact. For example, there are significant costs associated with the medical treatment of hypoglycemic events.4 Published costs of hospital admissions in the USA for severe hypoglycemia range from $2,811–17,564 per case, and tend to be higher for people with diabetic complications and comorbidities.5,6
The main reasons for poor adherence to SMBG include a lack of time, not remembering to test, being self-conscious, pain and the perceived invasiveness of SMBG, and social stigma.7,8 Recently, continuous glucose monitoring (CGM) has overcome many of the barriers associated with SMBG and it can help people increase their adherence to the recommended frequency of glucose monitoring.9–11 For people with type 1 or type 2 diabetes mellitus (T1/T2DM) using intensive insulin, CGM has demonstrated improved glycemic control compared with SMBG in randomized controlled trials (RCTs).9–14 Despite significant clinical benefits, CGM is not widely used among the population of intensive insulin users, in part because of high acquisition costs, consequently leading to limited reimbursement.15
Out-of-pocket costs of CGM can also be high for people with insurance-based healthcare.16
Flash continuous glucose monitoring
The FreeStyle Libre™ 14-day system (Abbott Diabetes Care, Alameda, CA, USA) is a new generation of CGM known as flash CGM. Flash monitoring has been available in the USA since 2017 for adults with diabetes and it typically has lower acquisition costs than other current CGM systems.17 The system wirelessly transfers data from a sensor to a handheld reader (or smartphone). The sensor is applied to the back of the upper arm and lasts for up to 14 days. The system is factory-calibrated and does not require calibration using SMBG; the glucose data can also be used to make treatment decisions.‡,18
The clinical benefit of flash monitoring for people using intensive insulin has been demonstrated in three RCTs. In two of these trials (IMPACT in people with T1DM, and REPLACE in people with T2DM), flash monitoring was associated with substantial decreases in daytime and nocturnal hypoglycemia compared with routine SMBG, without increasing glycated hemoglobin (HbA1c) or reducing the dose of insulin.9,10 During the IMPACT and REPLACE trials, people using flash monitoring reduced their use of SMBG by 90%, to 0.5 and 0.3 times/day, respectively, while scanning their sensors on average 15.1 and 8.3 times/day. All-cause health system resource use (hospital admissions, emergency room visits and ambulance use) was lower for people using flash monitoring compared with those using routine SMBG, especially in the REPLACE trial. The third RCT included people with T2DM, and demonstrated a reduction in HbA1c and improved treatment satisfaction for flash monitoring compared with SMBG.14
In real-world studies, flash monitoring has demonstrated reductions from baseline in HbA1c and hypoglycemia,19–29 and evidence from five countries shows that people who scan more frequently spend more time in range and have a lower mean glucose and estimated HbA1c.30
Evidence supports the persistence of scanning outside the clinical trial setting.31 In an analysis based on 279,446 sensors, users performed a mean of 16.3 scans/day (median 14). In the REPLACE study, 125 of 139 patients completed the 6-month open-access phase (89.9%). The average scan rate was 7.1 times/day (median 5.7) compared with 8.4 times/day (median 6.8) during the treatment phase, with 83.6% of possible glucose data collected compared with 88.7% during the treatment phase.31
Using UK National Health Service costs, flash monitoring has been shown to reduce health system costs when compared with routine SMBG.32,33 For people with T1DM using intensive insulin, the cost of flash monitoring was 19.4% less than SMBG at 10 tests/day, before allowing for potential reductions in costs associated with severe hypoglycemic events which would likely favor the flash monitoring system.32 For people with T2DM using intensive insulin, the cost of flash monitoring was 13.4% less than SMBG at 3 tests/day, once allowance was made for the costs of resource utilization reported in the REPLACE trial.33
Objective
The objective of this cost comparison was to estimate the costs associated with the flash monitoring system as a replacement for routine SMBG for people using intensive insulin to manage T1DM or T2DM. Acquisition costs of glucose monitoring and estimated costs of severe hypoglycemia have been included. The calculations were developed from the perspective of a US private payer.
Materials and methods
All calculations were conducted on a per patient per year (PPPY) basis.
Acquisition cost of glucose monitoring for base case and different SMBG scenarios
The acquisition cost of glucose monitoring was assessed at different frequencies of SMBG, based on the recommendation in the ADA Standards of Care, that people using intensive insulin should monitor their glucose 6–10 times (or more) daily.2 The base case compared the annual cost per person for the flash monitoring system with the cost of routine SMBG at
8 tests/day, the mid-point of the range recommended by the ADA. Scenarios 1 and 2 use the same approach as the base case, except they use the lower and upper limits of the range in the ADA Standards of Care, 6 and 10 SMBG tests/day, respectively. Scenario 3 is based on the same approach as the base case, but with routine SMBG used at 3 tests/day. Although much lower than the range recommended by the ADA, this is the frequency of SMBG observed in the REPLACE trial for people with T2DM using intensive insulin.
For the flash monitoring system, the calculations for the base case and different scenarios assumed a 14-day duration for each sensor, which is consistent with the sensor duration listed in the product labelling.18 This duration is supported by the median sensor duration of 13.92 days (interquartile range 13.61–13.95 days) observed in real-world use of the flash monitoring system, which was based on over 50,000 readers.17
Although the acquisition cost of the flash monitoring system is the same irrespective of whether people have T1DM or T2DM, separate cost calculations were created because there was a difference in the utilization of SMBG for people using flash monitoring in the IMPACT and REPLACE trials. For people with T1DM using intensive insulin, flash monitoring users were assumed to conduct a mean of 0.5 SMBG tests/day, as observed over the 6-month treatment period of the IMPACT trial.10 For people with T2DM using intensive insulin, flash monitoring users were assumed to conduct a mean of 0.3 SMBG tests/day, as observed over the 6-month treatment period of the REPLACE trial.9
The calculations used the list price for flash monitoring system sensors ($54.43 per 14-day sensor) and readers ($70.00 per reader). Based on the factory warranty, each reader is assumed to last for 3 years and the cost was therefore assumed to be the same for each year ($23.33/year). Each lancet was assumed to cost $0.08 and each test strip assumed to cost $1.42, which was the mean of leading manufacturers’ US list prices in January 2018.
Potential costs of severe hypoglycemia
For scenarios 1 and 3, evidence from the IMPACT and REPLACE trials was used to assess the potential costs of severe hypoglycemia. Scenario 1 was based on SMBG at 6 tests/day, which is close to the 5.6 SMBG tests/day observed in the IMPACT trial. Scenario 3 was based on SMBG at 3 tests/day, which is the same as the SMBG frequency observed in the REPLACE trial.
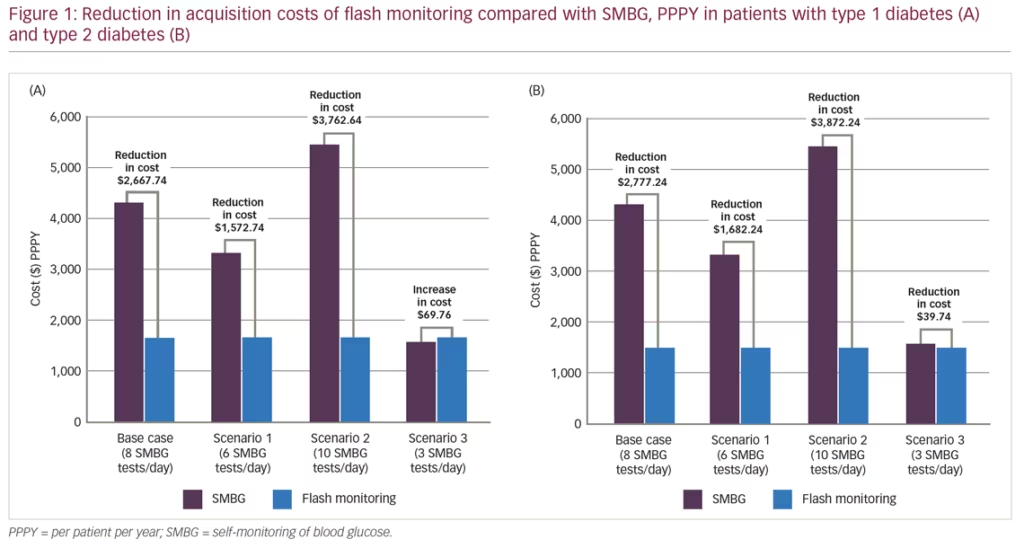
The average cost of treating a severe hypoglycemic event was estimated based on the proportions of events requiring an ambulance, use of an emergency room, or hospital admission, reported by Heller.4 These proportions were different for people with T1DM or T2DM. Consequently, the estimated cost for treating a severe hypoglycemic event for a person with T1DM was used for scenario 1, while the corresponding cost for treating a severe event for a person with T2DM was used for scenario 3. The costs for an ambulance, use of an emergency room, or a hospital admission are from a US Government Accountability Office report5 and a trial by Quilliam,6 updated to 2018 values based on the Consumer Price Index for medical care.34
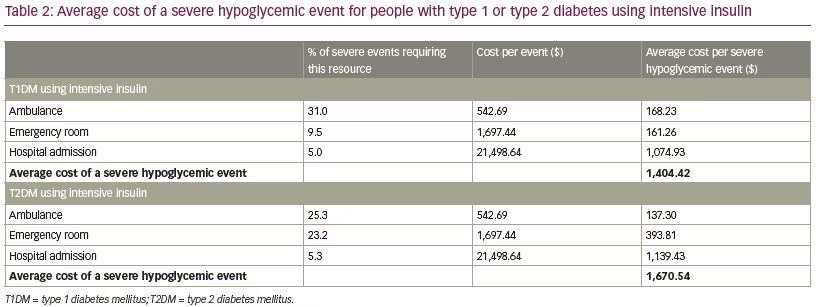
In scenario 1, the rate of severe hypoglycemic events for routine SMBG users was assumed to be 3.20 events PPPY, based on an observational study of people with T1DM for over 15 years.35 This evidence is from the UK; however, this source is considered suitable because it is based on an unselected population and hence is more relevant to the real-world setting of people with T1DM treated in general practice or by an endocrinologist.35 Although this source is from 2007, evidence from the EDIC study for 1995–2013 suggests that rates of severe hypoglycemia for people with T1DM using SMBG have been stable over time.36
Flash monitoring system users were assumed to experience a mean of 1.65 severe hypoglycemic events PPPY. This 48.5% reduction in the rate of events with the flash monitoring system was based on a proxy measure, the reduction in the rate of episodes with glucose levels below 45 mg/dL observed in the IMPACT trial. The use of changes in biochemical hypoglycemia as a proxy measure for changes in severe hypoglycemia is consistent with results reported using the data set from the Diabetes Control and Complications Trial,37 and is the same as the method used for a previous cost calculation.32
A similar approach was used for scenario 3 although in this case the rate of severe hypoglycemic events for routine SMBG users was assumed to be 1.05 events PPPY, based on a meta-analysis of trials of people with T2DM using insulin.38 Flash monitoring system users were assumed to experience a mean of 0.54 severe hypoglycemic events PPPY. This 48.8% reduction in the rate of events with flash monitoring was based on the reduction in the rate of episodes with glucose levels below 45 mg/dL observed in the REPLACE trial.9
Budget impact based on hypothetical cohorts
Hypothetical cohorts of 1,000 people using intensive insulin were used to assess the potential budget impact associated with the introduction of the flash monitoring system as a replacement for routine SMBG. Separate cohorts were considered for people with T1DM and for people with T2DM, and the base case utilization of SMBG, 8 tests/day, was assumed for people using SMBG. For both cohorts everyone used routine SMBG in the baseline year; in the first year, 15% used the flash monitoring system and 85% used SMBG; in the second year, 25% used flash monitoring and 75% used SMBG; and in the third year, 35% used flash monitoring and 65% used SMBG. The total acquisition cost of glucose monitoring was estimated for each year.
Results
Acquisition cost of glucose monitoring
The acquisition cost of routine SMBG for the base case (8 tests/day), was estimated to be $4,380 PPPY compared with $1,712 PPPY for people with T1DM using the flash monitoring system, a reduction of $2,667 (61%). The cost for flash monitoring includes $274 PPPY for SMBG, based on 0.5 tests/day observed in the IMPACT trial. For people with T2DM, the acquisition cost for the flash monitoring system was $1,603 PPPY, a reduction of $2,777 (63%) compared with routine SMBG. The cost for flash monitoring includes $164 PPPY for SMBG, based on 0.3 tests/day observed in the REPLACE trial.
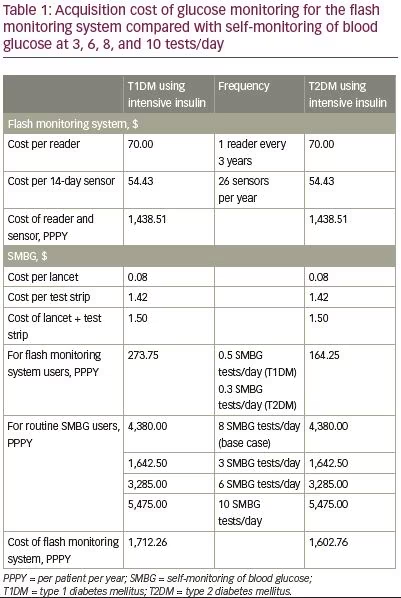
Scenarios 1 and 2 also demonstrated substantial cost savings for flash monitoring compared with SMBG at 6 and 10 tests/day for people with T1DM or T2DM, ranging from $1,573–3,872 PPPY. For scenario 3 there was a small cost reduction for flash monitoring compared with SMBG for people with T2DM. There was a small cost increase for flash monitoring in scenario 3 for people with T1DM although the additional cost in this scenario is much less than the cost savings shown in the base case or other scenarios.
Based on the results for the base case and scenarios, it is concluded that for people with diabetes who are using intensive insulin, the acquisition cost of glucose monitoring using the flash monitoring system is substantially less than that for routine SMBG testing from 3–10 or more times/day (Table 1, Figures 1A and 1B).
Estimated cost of severe hypoglycemia
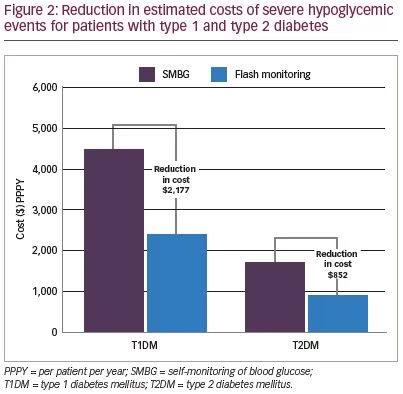
The estimated average cost of treating a severe hypoglycemic event was $1,404 for people with T1DM using intensive insulin and $1,671 for people with T2DM using intensive insulin (Table 2). For people with T1DM using intensive insulin, Table 3 and Figure 2 shows the cost of severe hypoglycemia for the flash monitoring system was estimated at $2,317 PPPY compared with $4,494 PPPY for routine SMBG. This is a reduction of $2,177 PPPY in favor of the flash monitoring system, together with a reduction in the incidence of severe hypoglycemia of almost 50%. For people with T2DM using intensive insulin, Table 3 and Figure 2 shows the cost of severe hypoglycemia for the flash monitoring system was estimated at $902 PPPY, compared with $1,754 PPPY for routine SMBG. This is a reduction of $852 PPPY in favor of the flash monitoring system together with a reduction in the incidence of severe hypoglycemia of almost 50%.
Combined cost of glucose monitoring and severe hypoglycemia
For scenario 1, the flash monitoring system was compared with SMBG at 6 tests/day, which is close to the 5.6 tests/day observed in the IMPACT trial. For people with T1DM using intensive insulin, the cost savings associated with use of flash monitoring compared with routine SMBG (at 6 tests/day) was $3,750 PPPY. These savings were based on a reduction of $1,573 PPPY in the cost of glucose monitoring and $2,177 PPPY in the cost of medical treatment of severe hypoglycemic events. For scenario 3, the flash monitoring system was compared with SMBG at 3 tests/day, which is the same as the 3.0 tests/day observed in the REPLACE trial. For people with T2DM using intensive insulin, the cost savings associated with use of flash monitoring compared with routine SMBG at 3 tests/day was $892 PPPY. These savings were based on a reduction of $40 PPPY in the cost of glucose monitoring and $852 PPPY in the cost of medical treatment of severe hypoglycemic events.
For both these scenarios, the savings associated with use of flash monitoring were even greater from reductions in severe hypoglycemia than from glucose monitoring. This confirms that on a per-patient basis, the overall cost of using the flash monitoring system will be less than for routine SMBG for people with T1DM or T2DM who are using intensive insulin.
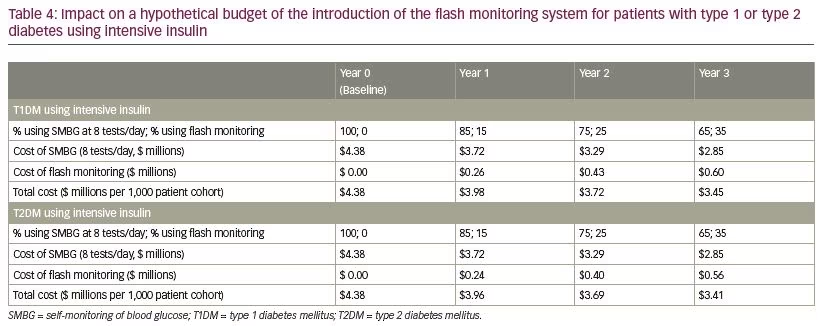
Budget impact based on hypothetical cohorts
For a cohort of 1,000 people with T1DM using intensive insulin, the total cost of glucose monitoring decreased from $4.38 million in the baseline year to $3.45 million in year 3 (Table 4). This is a reduction of 21% in the cost of glucose monitoring over the 3-year period. Similarly, for a cohort of 1,000 people with T2DM using intensive insulin, the total cost of glucose monitoring decreased from $4.38 million in the baseline year, to $3.41 million in year 3 (Table 4). This is a reduction of 22% in the cost of glucose monitoring over the 3-year period. These savings do not include additional savings from potential reductions in the rate of severe hypoglycemic events associated with the flash monitoring system.
Discussion
From a US private payer perspective for people with diabetes and who are using intensive insulin, these cost calculations demonstrate the flash CGM system provides a substantial cost-saving compared with SMBG testing from 3–10 or more times/day. In addition, the scanning rates observed in the IMPACT and REPLACE trials show that flash monitoring is associated with improved adherence to the recommended testing frequency from the ADA Standards of Care when compared with routine SMBG.9,10 Real-world evidence confirms that the scanning frequency observed in the IMPACT and REPLACE trials is maintained when flash monitoring is used by people in the regular daily management of their diabetes.32 Therefore, compared with SMBG, the flash monitoring system is expected to reduce the number of severe hypoglycemic events, reduce health system costs, and achieve greater adherence to recommended glucose monitoring frequencies for people with diabetes who are using intensive insulin.
The improvement in adherence to the recommended glucose monitoring frequency with the flash monitoring system may be higher than suggested by studies of SMBG.1 This is because those studies are typically based on prescription record data, which tends to over-estimate actual utilization of SMBG.
The possibility that use of the flash monitoring system could lead to reductions in health system costs is also supported by secondary evidence from the REPLACE trial, which showed a large reduction in utilization of health system resources (emergency room visits, ambulance callouts and hospital admissions) compared with the SMBG group, on an all-cause basis.33
The benefits of flash monitoring are supported by the observation that each scan provides more information than a single glucose reading from a SMBG test. For example, the flash monitoring system can provide a summary ambulatory glucose profile and a complete 24-hour glucose record. With flash monitoring, users are encouraged to test their glucose frequently because the incremental cost of each scan is zero, unlike SMBG where supplies are typically limited for each patient and the health system will pay more for an increased testing frequency. ADA Standards of Care2 and evidence from real-world studies3 support frequent glucose testing to achieve effective management of both HbA1c levels and to reduce the risk of hypoglycemia associated with intensive insulin use; flash monitoring appears to be more consistent with this goal than SMBG.
A recent international consensus statement endorsed by the ADA, American Association of Clinical Endocrinologists, American Association of Diabetes Educators, European Association for the Study of Diabetes, and the International Society for Pediatric and Adolescent Diabetes,39 recognizes the importance of time in glucose ranges (TIR) as “appropriate and useful as clinical targets and outcome measurements”. The flash monitoring system provides TIR in the ambulatory glucose profile report; however, SMBG does not provide this valuable measure and therefore is not consistent with the international consensus statement in this regard.
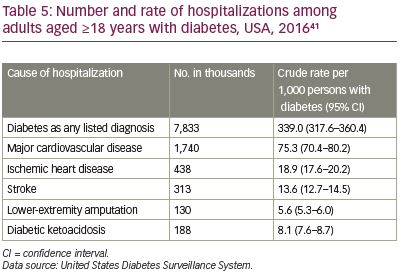
Poor glucose control is associated with an increased risk of microvascular complications, while severe hypoglycemia is associated with cardiovascular disease.40 Treating the complications of diabetes imposes a substantial burden on health systems in the USA. Table 5 presents evidence from the Centers for Disease Control and Prevention that in 2016 there were an estimated 7,833,000 hospitalizations for adults with diabetes, including 1,740,000 for major cardiovascular disease.41 Potentially, the reduction in hypoglycemia provided by the flash monitoring system may lead in the longer term to reductions in the incidence of cardiovascular events or other complications, reducing associated healthcare costs.
Possible limitations of the cost calculations in the present study include the exclusion of the cost of insulin and the focus on direct costs to the health system. Although the cost of insulin is an important consideration, it was not included in the calculations here because the IMPACT and REPLACE trials showed that insulin use was similar in the flash monitoring and SMBG arms.9,10 These cost calculations are restricted to direct treatment costs and do not include the wider costs of diabetes managed with intensive insulin. These include the possible costs, clinical consequences, and productivity losses associated with non-severe hypoglycemic events.
Concerning resources related to severe hypoglycemia, only clearly stated resource use was included.4 The authors acknowledge there was potential for under-reporting of ambulance use; also additional blood glucose measurements, costs of glucagon or intravenous dextrose, or any subsequent medical consultations that were necessary following severe hypoglycemia were not included. Given the substantial reduction in hypoglycemia observed in the IMPACT and REPLACE trials, it is possible that flash monitoring may reduce these indirect costs in the population of intensive insulin users. These cost calculations also do not take account of resource use associated with hyperglycemia, including diabetic ketoacidosis (DKA); recent real-world evidence shows flash monitoring reduces the rate of DKA compared to the period before flash monitoring was initiated,42,43 which may provide additional cost savings.
Overall, the estimated resource use in these cost calculations may be an under-estimate of the underlying total resource use for flash monitoring and SMBG, and the cost savings for flash monitoring compared with SMBG may be greater than stated here. Although these cost calculations have focused on the private payer, a previous assessment has been reported of the cost of flash monitoring versus routine SMBG in intensive insulin users with T2DM covered by Medicare and Medicaid.44 This assessment did not make allowance for the benefit of flash monitoring in reducing health system costs compared with SMBG by avoiding severe hypoglycemic events. Allowing for this factor, the flash monitoring system could potentially be cost neutral in Medicare and Medicaid populations, while increasing adherence to ADA monitoring guidelines and reducing the number of hospitalizations required for severe hypoglycemia.
The annual cost per patient of the flash monitoring system in these calculations is $1,712, which compares favorably with the annual cost reported for other CGM systems. For example, in 2016 a US model used an annual cost for CGM of $4,500–4,900,45 while a UK model included a cost of £3,74046 ($4,600) and a Canadian model used C$6,588 ($4,940).47 This cost advantage is supported by ADA Standards of Care, which state that flash monitoring “may be a more affordable alternative to real-time CGM systems for patients on intensive insulin therapy.”2 In addition, the flash monitoring system is approved by the US Food and Drug Adminstration as nonadjunctive, meaning it is designed to replace SMBG.‡ Some CGM systems, in contrast, still require the additional expense of SMBG to enable users to make insulin dosing decisions.
Conclusion
These calculations demonstrate that, from a US private payer perspective, the flash CGM system provides cost savings compared with routine SMBG for people with diabetes who are using intensive insulin. Scenarios show that cost savings with flash monitoring are achievable compared with SMBG from 3–10 or more tests/day. Cost-savings with flash glucose monitoring may be realized through a reduction in severe hypoglycemia. Further study of budget impact is needed to include changes in behavior leading to improved adherence.
‡Based on the US Food and Drug Administration (FDA) label, SMBG tests remain required for treatment decisions when: 1) the Check Blood Glucose symbol is present; 2) symptoms do not match system readings; 3) the user suspects that readings may be inaccurate; 4) the user experiences symptoms that may indicate high or low blood glucose.18







