According to the estimates of the International Diabetes Federation (IDF), one in every 11 adults of the global population has diabetes, with an overall disease prevalence of 425 million.1 This diabetes prevalence is expected to increase exponentially with time because of the global obesity epidemic related to physical inactivity and adverse food habits. Therefore, physicians and other healthcare professionals face the challenge of managing many diabetes patients in their day-to-day clinical practice, and are expected to have adequate knowledge and skills in dealing with these cases.
Two major types of diabetes cases, type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM), are often seen in clinical practice, along with a small percentage of other forms of the disease. Optimal management of diabetes primarily targets adequate control of plasma glucose levels in the normal physiological reference range as in a healthy non-diabetic subject without causing the risk of hypoglycaemia. Normal plasma glucose levels in a healthy individual depend on the dietary intake of nutrients, physical activity and hormones that control glucose homeostasis, especially insulin. Therefore, diabetes management should target dietary interventions, modifications in lifestyle including physical activities and hormonal manipulation (mainly insulin) and/or its effects. This article gives an overview of different types of non-pharmacological interventions for the effective management of patients with diabetes.
The risk of T2DM rises steadily with increasing bodyweight and those with morbid obesity have the highest risk of the disease.2 Excess bodyweight, related to physical inactivity and over-nutrition, results in the accumulation of fat mainly in the abdomen and visceral tissues. This abdominal and visceral adiposity in turn modulates several hormonal and chemical mediators in the body that result in ‘diabesity’, the term coined to interlink diabetes in relation to obesity.3 The ‘metabolically obese’ phenotype having normal bodyweight and normal or low body mass index (BMI), but increased abdominal obesity, is another underestimated risk group for the development of T2DM. Free fatty acids derived from visceral adipose tissues reduce the insulin sensitivity and impair β-cell function (lipotoxicity), resulting in the development of T2DM.
Being a disease directly related to adverse lifestyles, the first approach to the effective management of diabesity should be lifestyle interventions. Although multiple pharmacological interventions may be associated with improvements in obesity and diabesity, the durability and sustainability of these interventions are often modest. Therefore, non-pharmacological measures, including bariatric surgery, are of paramount importance in these patients. Better understanding of the pathophysiology of diabesity resulted in the development of several novel therapeutic strategies over recent years for the management of this alarming global epidemic.4
T1DM results from absolute insulin deficiency and, therefore, management is mainly with insulin. However, the treatment of T1DM should also consider dietary modifications and physical activity for optimal outcomes. Obesity affects a proportion of patients with T1DM that increases their insulin requirements and adversely affect their metabolic control.5 Therefore, lifestyle modifications are important even in cases with T1DM. Although bariatric procedures are usually done in patients with T2DM, obese patients with T1DM also have shown some beneficial effects with a significant reduction of insulin requirements and modest improvement in diabetes control.6
Dietary interventions in patients with type 2 diabetes mellitus
Nutritional interventions are important in achieving optimal glycaemic control in patients with T2DM. Most patients with T2DM are overweight or obese and, therefore, weight loss achieved through dietary restriction of energy helps diabesity management. Although results of the Look AHEAD trial did not demonstrate clear cardiovascular mortality benefits of intense lifestyle interventions including dietary restrictions aiming at weight loss, intense lifestyle interventions were associated with better glycaemic control in the participants.7 The Look AHEAD trial participants were aged >45 years with diabetes, and thus the results may not be extrapolated to younger adults and those with early onset of T2DM. However, recent studies have clearly demonstrated that different models of high-quality diets were associated with significant reductions in all-cause mortality, cardiovascular disease, cancer and T2DM by 22%, 22%, 15% and 22%, respectively.8
Diet and blood glucose response
Blood glucose response to individual food items varies according to glycaemic index (GI) and glycaemic load (GL) of the diet. GI is calculated by the blood glucose response 2 hours after the intake of 100g of a particular food item expressed in comparison to the blood glucose response after the intake of 100g of glucose. The GI value of dietary items also changes with the ripeness of food, processing and cooking methods, and the presence of other ingredients in the diet that affects digestion and absorption. GL depends on the amount of carbohydrate present in a particular food item for digestion and absorption, and is calculated by measuring the carbohydrate content of the item, multiplied by its GI and divided by 100.9,10 GL is a concept to overcome some of the drawbacks of GI. It considers the glycaemic effects of food in a normally eaten portion, a more realistic measure.10 A GI of ≤55 is considered as low, ≥70 as high and in between as medium. Similarly, a GL of ≤10 is considered low, ≥20 high and in between as medium.11
Multiple studies have shown the beneficial effects of diet with low GI on the improvement of T2DM and its complications.12–17 Diets with low GL have also shown benefits in managing T2DM and reducing complications in several studies.12,13,16,17
Medical nutrition therapy
The American Diabetes Association (ADA) uses the term medical nutrition therapy (MNT) to describe the optimal co-ordination of dietary intake with diabetic therapy (both pharmacological and non-pharmacological) to achieve a favourable outcome.18 MNT can be used as a primary, secondary or tertiary prevention measure in T2DM. Primary prevention measures of MNT are by modifying diets in high-risk individuals (i.e., pre-diabetes, obese etc.) to delay or prevent the onset of T2DM. Secondary prevention measures aim to achieve tight glycaemic control by dietary modification and, thereby, reducing diabetic complications in patients with T2DM.18 Tertiary prevention measures are to manage diabetes-related complications such as cardiovascular or renal disease in those with T2DM.
MNT not only aims to control blood glucose, but also to improve other co-morbidities such as hypertension, dyslipidaemia and obesity.19 Diets with low GI reduce postprandial blood glucose excursions that improve glycaemic control. Non-nutritive sweeteners and a low-calorie diet may also help diabetes management. Increasing intake of dietary fibre also improves glycaemic control. Routine supplementation of vitamins, antioxidants and trace minerals are not recommended, unless there is a deficiency of the same. Hypocaloric and low carbohydrate diets help weight management, along with metabolic control in T2DM. People with pre-diabetes and diabetes should receive individualised MNT from a registered dietician familiar with diabetic MNT. Steps involved in MNT planning and implementation are given in Figure 1.
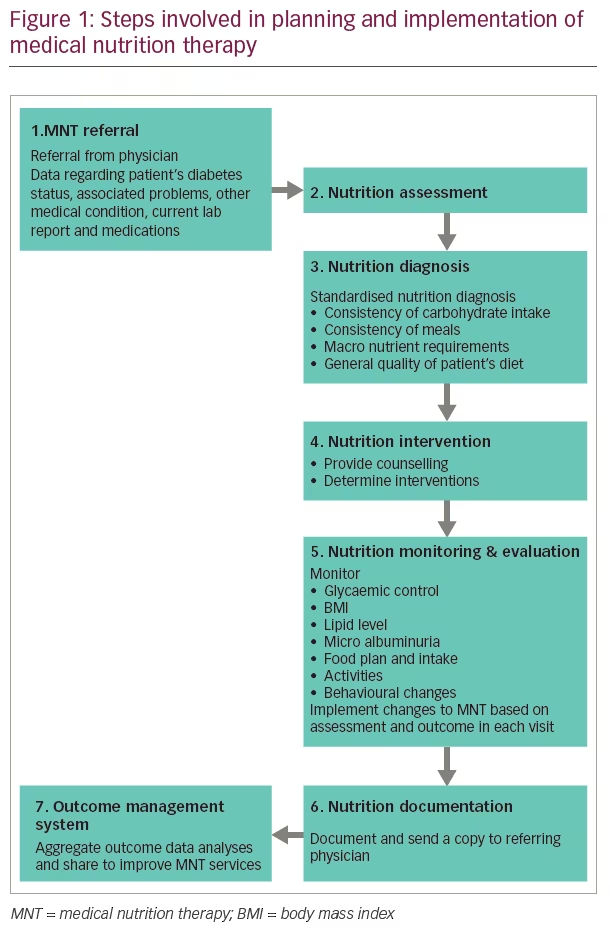
Diabetes-specific formulae adopted in MNT have resulted in a significant reduction of glycated haemoglobin (HbA1c) levels, postprandial glucose levels and insulinaemic responses in diabetes.19 A meta-analysis that included 16 studies (follow-up duration 6 months to 4 years) revealed that MNT with diets containing a low amount of carbohydrates and low GI, and with a high content of protein or Mediterranean diet (MD), resulted in a significant improvement of glycaemic control in patients with T2DM.20 Among these MNT types, the MD was associated with the largest reductions in HbA1c (-0.47%) and bodyweight (-1.84 kg on average).
Health benefits of a Mediterranean diet in diabetes
The MD is basically a plant-based diet, which is well known for its various health benefits, mainly in cardiovascular diseases and cancer.21 The MD also has a favourable effect in people with diabetes. This dietary pattern adapted in the olive-growing areas of the Mediterranean region (especially Greece and Italy) in the late 1950s and early 1960s, collectively known as MD, is used to describe the traditionally followed dietary pattern of people living in countries bordering the Mediterranean.21,22
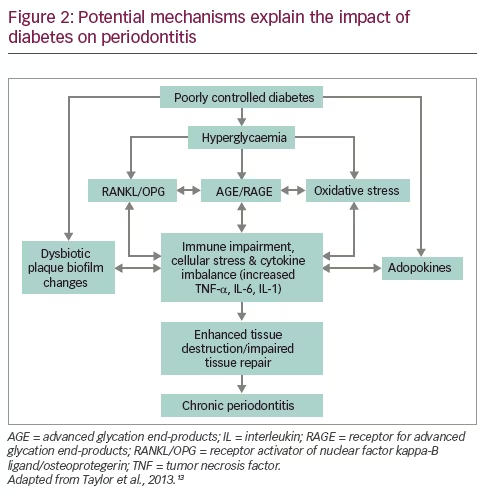
The MD consists of a high amount of olive oil, vegetables, legumes, whole grains, fruits and nuts, a moderate amount of poultry and fish, a low amount of whole fat dairy products and red meat, and low to moderate amounts of wine.23,24 The MD is found to be protective not only in healthy individuals but also in women with a history of gestational diabetes and those with cardiovascular disease.25,26 In the PREDIMED-Reus nutrition intervention trial among non-diabetic participants with high cardiovascular risk, MD was found to reduce the incidence of T2DM by 52%.27 In patients with T2DM, MD is found to be beneficial in achieving glycaemic control, reducing insulin resistance and cardiovascular risk factors (BMI, blood pressure, cholesterol, inflammatory markers and adhesion molecules) and improving liver function and sexual function.28,29 Probable mechanisms of the beneficial effects of MD in T2DM are depicted in Figure 2.
Dietary interventions in patients with type 1 diabetes mellitus
Although the principles of MNT are similar for the management of T1DM, another important aspect of dietary management is to be considered in these patients. The insulin doses can be calculated as per the carbohydrate counting principles adopted for patients to optimise diabetes control.30 Carbohydrate counting is a method of calculating the grams of carbohydrate consumed in a meal expressed as grams or as servings. One carbohydrate serving is equal to 15 g of carbohydrate. Calculating the insulin-to-carbohydrate ratios helps in adjusting the mealtime dose of insulin. Significantly better HbA1c reduction has been reported with this strategy in a recent meta-analysis.31 Therefore, in an ideal situation, carbohydrate counting should be recommended to all patients with T1DM. This approach also benefits patients at risk of hypoglycaemia because appropriate insulin dosing as per patient’s metabolic requirement and food intake helps to prevent hypoglycaemia.30
Physical activity for patients with type 2 diabetes mellitus
Physical activity refers to any body movement that increases energy expenditure above the baseline, whereas exercise is a planned, structured and repetitive physical activity performed with the objective of improving physical fitness. Physical activity improves insulin sensitivity, bodyweight, cardiovascular risk factors, physical fitness, lipid level, blood pressure and overall wellbeing, and reduces the risk of cardiovascular morbidity and mortality.32
Physical activity not only improves glycaemic control in patients with T2DM, but also reduces bodyweight and blood pressure. It improves the adverse lipid profile by reducing total cholesterol and low-density lipoprotein (LDL) cholesterol and increasing high-density lipoprotein (HDL) cholesterol.33 These, in turn, reduce the risk of various cardiovascular events inherent to patients with T2DM.
Physiological changes during exercise
During the first few minutes of exercise, glycogen is broken down and used. But with continued exercise, muscle glycogen breakdown decrease and fall in blood glucose results in the suppression of insulin secretion and an increase in glucagon secretion, which stimulate hepatic glycogenolysis.34 With prolonged exercise, muscle glycogen stores get depleted and free fatty acids, derived from the breakdown of triglycerides, are used as fuel for muscle activity. Exercise increases glucose uptake and the use in skeletal muscles by increased translocation and expression of glucose transporter 4 (GLUT4, an isoform of glucose transporter) from intracellular storage depots to plasma membrane, and by increasing insulin sensitivity.34–36 Exercise also improves hepatic insulin sensitivity, reduces insulin resistance and improves glucose disposal in peripheral tissues.37,38
Medical evaluation before exercise programme
Healthcare providers should evaluate patients with T2DM before starting an exercise programme, especially in individuals leading sedentary lifestyles and at risk of cardiovascular disorders. Providers should look for the conditions which either contraindicate or predispose to increased morbidity with certain types of exercise, such as uncontrolled diabetes, uncontrolled hypertension, diabetic peripheral neuropathy, autonomic neuropathy, diabetic foot, Charcot’s arthropathy and untreated proliferative retinopathy.
Exercise increases the risk of hypoglycaemia in T2DM, especially if the patient is already on insulin/insulin secretagogues.39,40 These patients should adjust the dose of insulin/hypoglycaemia agents after appropriate discussion with the diabetes-care professional. If the blood glucose value is <100 g/dl (5.6 mmol/L) prior to exercise, additional consumption of carbohydrates may be necessary to prevent hypoglycaemia during exercise. The risk of hypoglycaemia also depends upon the duration and intensity of exercise, and the time of day exercise is done in relation to the last food intake. Increased insulin sensitivity induced by exercise can even result in delayed hypoglycaemia, several hours after the exercise.
Exercise in type 2 diabetes mellitus
Patients with T2DM should engage in moderate- to high-intensity exercise for at least 150 minutes per week, spread over a minimum of 3 days a week. They should have no more than 2 consecutive days without activities, or 75 minutes per week of vigorous-intensity physical activity.41 Adults should also perform muscle-strengthening exercises involving all major muscle groups for 2 or more days per week. All individuals should avoid prolonged sitting/sedentary activity by breaking every 30 minutes by briefly standing, walking or performing other activities.42,43 Frequency, duration, intensity and type of exercise programme, age of the patient and adherence to the exercise programme are the major factors influencing the benefits of exercise therapy in T2DM.
It was found that exercise-induced improvement in insulin sensitivity lasts between 24 and 72 hours, and hence T2DM patients should perform regular exercise with no more than 2 consecutive days without physical activity to maintain the benefits. The beneficial effect also shows a clear linear relationship with the total energy expenditure during exercise.44 The relative risk of development of T2DM in men who exercise once-weekly only was higher than those who do exercise five or more times per week in the physicians’ health study.45
Physical activity for patients with type 1 diabetes mellitus
Exercise training in the form of resistance, endurance and combined training consisting of both, has been shown to improve glycaemic control and diabetes outcomes in patients with T1DM in a recent systematic review and meta-analysis.46 However, we need to keep in mind that there is substantial risk of hypoglycaemia during and after exercise. At times, post-exercise hyperglycaemia is encountered in patients with T1DM because of a surge of counter-regulatory hormones during and after exercise. The insulin treatment may need modification in patients who do only occasional unaccustomed physical activities to prevent these complications.
The benefits of exercise depend on factors such as frequency, duration, intensity, type of exercise, age of the patient and the adherence to the exercise programme. This is collectively termed as the FITT principle: Frequency (the number of times exercise is done), Intensity (the amount of effort for the exercise), Time (the duration of exercise) and Type (aerobic or resistance).47
Bariatric surgery
Various surgical procedures collectively called bariatric surgery or metabolic surgery, initially proposed for the treatment of morbid obesity are also found to be effective in both the prevention and control of T2DM. It is interesting to note that these procedures can even result in the remission of T2DM in a good proportion of cases. The restriction in calorie intake achieved directly by surgical procedures and indirectly by self-adaptation to restrict post-prandial dumping syndrome, alterations in satiety by postprandial neuro-humoral signals and incretin effects, mal-absorption of nutrients, increase in beta cell mass and improvement in insulin production, and changes in the gut microbiome are some of the mechanisms that play important roles in control and remission of T2DM in patients undergoing bariatric surgical procedures.48–50
Types of bariatric surgical procedures
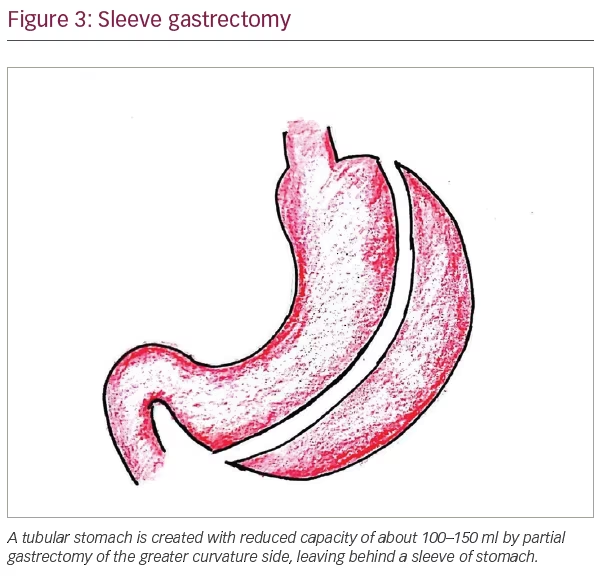
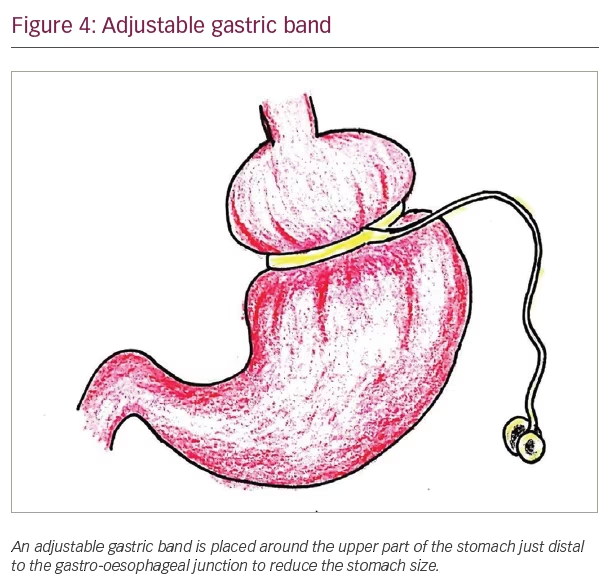
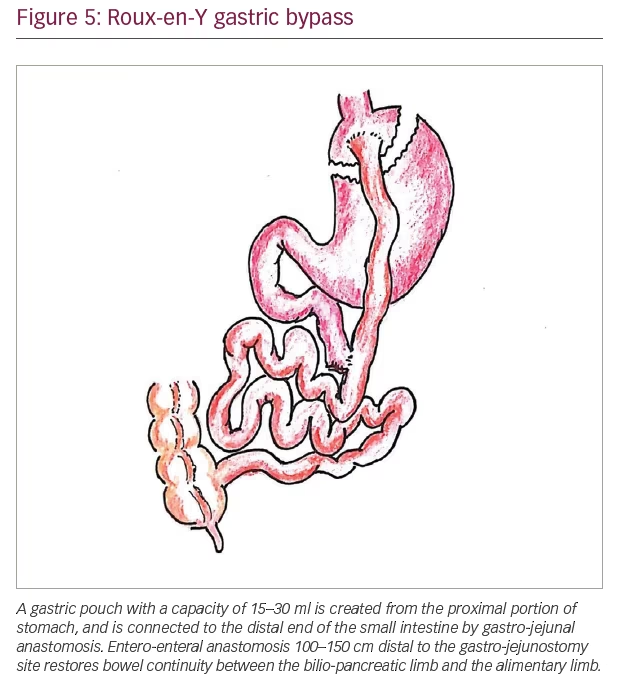
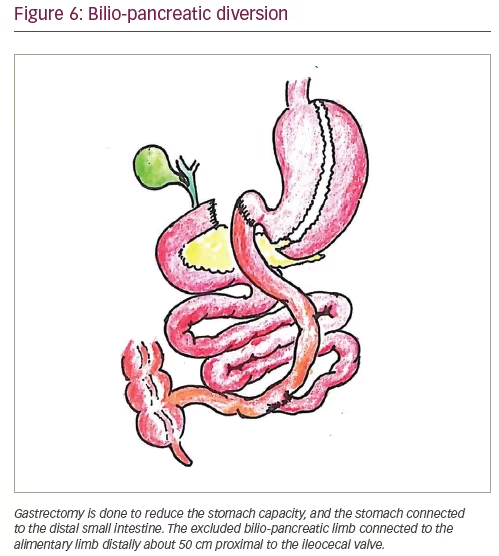
The currently accepted common metabolic surgical procedures include Roux-en-Y gastric bypass (RYGB), laparoscopic adjustable gastric banding (LAGB), bilio-pancreatic diversion (BPD) and laparoscopic sleeve gastrectomy (LSG). Laparoscopic surgeries are preferred over open surgeries, and laparoscopic sleeve gastrectomy and laparoscopic RYGB are the most commonly performed bariatric surgical procedures globally. Figures 3–6 show the diagrammatic representation of various bariatric procedures.
Enhanced recovery after surgery (ERAS) protocol is a multimodal programme that further reduces the complications, and shortens the length of hospital stay after bariatric surgery. Careful patient selection and education, well defined standard preoperative protocols, properly educated medical teams, use of laparoscopic surgery, clearly defined discharge criteria and modern perioperative care are the major components of ERAS.51 Although this approach has shown benefits in terms of reduced hospital stay, reduction of morbidity has not been observed in a recent meta-analysis.52
Complications of bariatric surgery
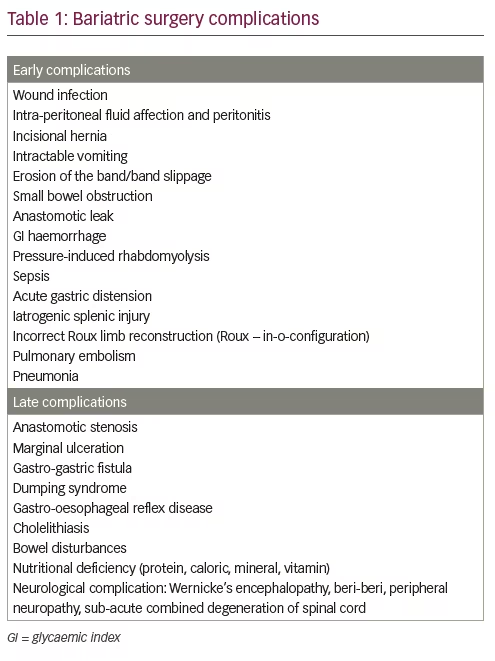
Common complications following bariatric surgery are listed in Table 1. Thirty-day mortality of bariatric surgical procedure is low (0.1–0.3%), similar to that of laparoscopic cholecystectomy.53,54 Factors which influence the risk of complications include experience and volume of surgeries in the centre, complexity of the procedure, age and BMI of the patient, male sex, limited physical activity and associated problems such as hypertension, obstructive sleep apnoea (OSA) and risk factors for pulmonary thromboembolism.55 Obesity surgery mortality risk score helps to predict surgical mortality risk after bariatric surgery.56 The variables considered are BMI >50, male sex, hypertension, risk factors for pulmonary thromboembolism and age >45 years. Obesity surgery mortality risk score assigns one point for each variable, so that the total score varies from 0–5. Higher scores are associated with increased mortality risk. The predicted mortality risks are: low-risk group A (scores 0–1) – 0.2%; intermediate group B (scores 2–3) – 1.1%; and high-risk group C (scores 4–5) – 2.4%.56
Postoperative bleeding can be from mesenteric or omental vessels or from the anastomotic site after bariatric surgery. The risk of pulmonary embolism after bariatric surgery is around 2% and is identified as a leading cause of death after surgery.54 Early ambulation in the post-operative period can reduce the risk of pulmonary embolism. Anastomotic leak is another leading cause of mortality and usually presents with severe tachycardia, respiratory failure, left shoulder pain and left pleural effusion in the early post-operative period that should be differentiated from acute pulmonary embolism.57
Another common complication is small bowel obstruction within the first month of surgery that presents with severe abdominal pain, nausea, vomiting, obstipation and tachycardia resulting from kinking at the jejuno-jejunostomy site, anastomotic site stenosis, external compression, or adhesions or herniation through the trocar site.58,59 Early dumping syndrome associated with cholinergic symptoms such as vomiting, tachycardia and dizziness (within 30–60 minutes of food intake) occurs in several cases, especially after taking carbohydrate-rich food due to the rapid exposure of undigested carbohydrates to the distal small intestine. Late dumping syndrome, the clinical features of which results from hypoglycaemia, occurs 1–3 hours after a meal due to excess insulin release. Common symptoms include hunger, sweating, tremor, difficulty in concentrating and occasionally loss of consciousness. Post-bariatric surgery patients should adhere to specific dietary recommendations such as taking food in small fractions, eating slowly, chewing well and drinking fluids only after an hour of a meal to prevent many of these complications.60 Failure to adhere to dietary recommendations after surgery results in pouch distension and vomiting. A high-protein diet and vitamin supplementation help to prevent nutritional deficiencies seen after bariatric surgery.
Acute gastric dilatation, a potentially dangerous complication of bariatric surgery, even though rare, presents with abdominal pain or left shoulder pain, abdominal distension, bloating, hiccup, nausea, vomiting, breathlessness and tachycardia.61
Infections of the excess abdominal wall skin (panniculitis) are common after bariatric surgery. If this is not controlled even after adequate skin hygiene and antibiotics, surgical excision of excess skin is required.
Major nutritional deficiencies associated with bariatric surgical procedure include: iron, vitamin B12, folic acid, calcium, vitamin D, thiamine, proteins, essential fatty acids and fat-soluble vitamins.62 As nutrients are diverted from the stomach and proximal small bowel, iron, vitamin B12 and other micronutrient absorption can be impaired which results in various nutritional deficiencies. The risk of development of iron deficiency is 13–52% within 2–5 years after bariatric surgery, which can be significantly reduced by iron supplementation.62 The chance of the development of calcium deficiency is about 10% and vitamin D deficiency is up to 51%, which can lead to secondary hyperparathyroidism. Routine daily supplementation of multivitamins, iron, vitamin B12 and calcium can reduce the incidence of these deficiencies.62
Gastro-gastric fistulas, an abnormal communication between the gastric pouch and the excluded stomach, is a rare complication after bariatric surgery that results in inadequate weight loss or weight gain, marginal ulceration, recurrent gastrointestinal haemorrhage and abdominal pain.63 Some surgeons perform prophylactic cholecystectomy for those with gallstones to prevent future biliary obstruction. Stenosis at the gastro-jejunostomy site is another late complication that leads to progressive epigastric pain and vomiting after food and even liquids.64 Hyperoxaluria with nephrolithiasis is another important delayed complication of mal-absorptive bariatric procedures.65
Post-bariatric surgery hypoglycaemia (PBH) is another severe late complication, where hypoglycaemia occurs 1–2 hours after food intake without any vasomotor symptoms. Differentiating PBH from late dumping syndrome can be difficult. However, the presence of neuroglycopaenic features is often associated with PBH, which helps suspicion of this condition in patients.66 Carbohydrate-restricted diet, increased consumption of dietary fibres and avoidance of alcohol helps to reduce the incidence of PBH.66–68 Drugs such as acarbose, diazoxide, octreotide and verapamil are useful in treating PBH.67,68 Restoring gastric restriction with an adjustable band, placing the gastrostomy tube and subtotal or total pancreatectomy are proposed as surgical treatment options for severe intractable PBH.68
Mechanisms of type 2 diabetes mellitus remission after bariatric surgery
Several putative mechanisms have been proposed to explain the remarkable improvements in metabolic parameters that lead onto diabetes remission in a significant proportion of patients undergoing bariatric procedures. However, there may still be other mechanisms to be elucidated. The currently identified mechanisms are discussed below.
Marked loss of bodyweight following surgery is one of the most important factors in diabetes remission.48 The Look AHEAD study has clearly demonstrated the linear association between the degree of weight loss and HbA1c reduction in the study participants.69 However, weight loss alone will not explain the improvement in glycaemic parameters and diabetes remission following surgery.
A reduction of calorie intake following the surgery is one of the important factors that leads onto glycaemic improvement in the immediate period along with long-term weight loss. A calorie-restricted diet by itself helps T2DM cases to improve insulin sensitivity, β-cell function and the glycaemic parameters.70 Long-term calorie restriction is mandatory in patients who have undergone bariatric surgery to obtain maximum health benefits including the maintenance of diabetes remission.
Marked changes in the gut hormone levels have been observed in animal models and clinical cases following bariatric procedures. Increased secretion of glucagon-like peptide-1, glucose-dependent insulinotropic peptide, peptide-YY and oxyntomodulin that alter energy balance and glucose metabolism have been reported.48,71,72 Ghrelin (a gut hormone that stimulates hunger) level was found to be low after bariatric surgery, which suppress hunger signals in these participants.73 These alterations in hormonal mechanisms positively affect the energy balance, insulin sensitivity, β-cell function and glycaemic parameters to contribute to improvement/remission of T2DM and also help weight loss in patients.
Major alterations in the intestinal microbiota is another important mechanism that helps in the improvement of T2DM in patients undergoing bariatric surgery. In recent years, gut microbiome has been proposed to influence a multitude of physiological and pathological processes in human beings.74 Marked changes in the gut microbiome after bariatric procedures have been shown to improve metabolic parameters and bodyweight in multiple studies.75–77 The intestinal microbiome seen in obese individuals are replaced by organisms seen in lean individuals after bariatric surgery.78 Germ-free mice receiving these ‘lean microbiota’ population showed significant weight loss and reduction in body fat.79 People with T2DM receiving microbiota from normal individuals showed improvement in glycaemic control, emphasising the role of gut microbiota in metabolic control.80 Germ-free mice receiving gut microbiota from post-bariatric surgery cases showed a reduction in body fat composition and lower respiratory quotient.81 Alterations in the intestinal and plasma bile acids is another postulated mechanism in the improvement of T2DM, bodyweight and other metabolic parameters after bariatric surgery.82
Type 2 diabetes mellitus and obesity outcomes following bariatric procedures
Several factors influence diabetes outcomes following bariatric surgery. The Diabetes Surgery Score system helps to predict these outcomes that consists of variables such as age, BMI, c-peptide levels and the duration of diabetes. The total score varies from 0–10, and patients with higher scores get better outcomes, including diabetes remission.83 Those with a total score of 10, the chance of remission nears 100%, whereas for those with a score of 0, it is only 33%. An increase in the total score by 1 point translates into a 6.7% increase in the chance of remission.83 Other scoring systems used for the prediction of diabetes remission include ABCD score (age, BMI, c-peptide level, duration of diabetes) and DiaRem score (age, HbA1c level, medications and insulin usage).84
If the fasting plasma glucose is <100 mg/dL or HbA1c <6% for at least 1 year without any active pharmacological therapy, complete remission is obtained, and if these parameters are sustained for at least 5 years, prolonged remission is achieved.85 Similarly, if fasting plasma glucose is between 100–125 mg/d or HbA1c between 6 and 6.5% for at least 1 year, patients achieved partial remission. Factors identified to influence remission were: preoperative glycaemic control, history of insulin use, duration of diabetes, patient compliance, weight loss, the type of diabetes (T2DM or late onset autoimmune diabetes, or T1DM) and greater baseline visceral fat area.86,87
A sustained remission of diabetes is seen in 30–63% of T2DM patients following bariatric surgery on an average follow-up period of 1–5 years.88 Although 30–50% of patients achieved initial remission relapse after a median follow-up duration of 8.3 years,89,90 they maintained significant improvement of glucose control compared to the baseline for 5–15 years.91–93 Favourable factors associated with higher chance T2DM remission following bariatric surgery include younger age, shorter duration of T2DM (<8 years) good glycaemic control without insulin and greater baseline visceral fat area.94,95
The percentage loss of excess weight and remission of diabetes achieved by BPD is higher (73% and 95% respectively) compared to RYGB (63% and 80%) and LAGB (49% and 57%).96 A recent American RYGB follow-up data suggested that the adjusted mean change from baseline in bodyweight in the surgery group was -45 kg (mean percent change, -35.0) at 2 years, -36.3 kg (mean percent change, -28) at 6 years and -35 kg (mean percent change, -26.9) at 12 years.97 Incident T2DM was significantly low and the maintenance of diabetes remission at 12 years was much higher in the RYGB group compared to controls. Hypertension and dyslipidaemia also were significantly low in these cases at 12 years.97
Morbidity and mortality benefits of bariatric surgery in type 2 diabetes mellitus
Apart from benefits resulting from massive weight loss and diabetes remission/improvement, bariatric surgery also confers protection from micro-vascular and macro-vascular complications of T2DM including diabetic nephropathy and myocardial infarction. Marked improvements in metabolic parameters such as a reduction in LDL cholesterol and triglycerides, and increase in HDL cholesterol were seen in patients.98 Remission of abnormal kidney function in 86%, prediabetes in 76%, elevated blood pressure in 74% and dyslipidaemia in 66% of young adults with significant improvement of weight-related quality of life were reported following 3 years of bariatric surgery in adolescents recently.99
Significant reduction of all-cause and disease-specific mortality are reported in patients with T2DM undergoing bariatric procedures by multiple large cohort studies on long-term follow-up.97,99–101 A very recent meta-analysis that included one RCT and nine cohort studies examining the mortality risk with follow-up duration of >5 years showed a relative risk of 0.21 (95% confidence interval [CI] 0.20–0.21) in the surgical groups compared to non-surgical groups.100 Another recent study reporting 12-year follow-up data showed that the age- and sex-adjusted mortality was significantly lower in the RYGB group compared with the non-surgery group (odds ratio [OR] = 0.53 [95% CI 0.30–0.92]; p=0.02).97 However, this study revealed an increased suicide rate in the surgery group possibly from worsening of depression after bariatric procedures.
Bariatric surgery for type 1 diabetes mellitus
Although bariatric surgery is not a treatment option for glycaemic management in patients with T1DM, some patients with severe obesity may need surgery for the management of obesity and other associated metabolic conditions. Surgery was associated with a significant reduction in bodyweight, insulin requirements, improvement of obesity comorbidities and some diabetes complications.102 However, there was only a marginal effect on glycaemic control in treated patients on long-term follow-up.
Newer bariatric procedures
Intra-gastric balloon treatment is a procedure in which a balloon is placed inside the stomach endoscopically, inflated with saline and kept for 6 months, for promoting weight loss in obese individuals. Intra-gastric balloon has been recently approved by the US Food and Drug Administration for the treatment of obesity. A recent meta-analysis examining the benefits of this treatment revealed that a statistically significant proportion of patients had resolution of T2DM (OR 1.4; 95% CI 1.3–1.6).103 Serious complications were reported in 1.3% of cases. Oesophagitis, nausea and vomiting are the common side effects and 25–40% regain weight in 1-year follow-up.
EndoBarrier Gastrointestinal Liner (GI Dynamics®, Boston, US), duodenal-jejunal bypass sleeve, is a mechanical device that is placed in the proximal small intestine to prevent the contact of food with pancreatic and biliary secretions until it reaches the initial part of the jejunum.104 A 60 cm sleeve, made of polymer, placed endoscopically in the initial part of the small intestine that excludes proximal small gut (EndoBarrier) is effective in reducing bodyweight and HbA1c level in obese patients with T2DM. Mean differences in bodyweight, excess weight loss and HbA1c of -5.1 kg, 12.6% and -0.9% respectively, were observed in a recent meta-analysis examining five RCTs and 10 observational studies involving 446 patients treated by this technique.105
Alterations in the levels of gut hormones and changes in hepatobiliary enzymes in the bypassed proximal intestine are the proposed mechanisms in the improvement of diabesity outcomes in these patients.106 However, more research is necessary to elucidate the exact reasons for improvement. EndoBarrier treatment has been associated with an improvement of hepatic steatosis and fibrosis in patients with T2DM in a recent study.107 Common adverse events were abdominal pain, nausea and vomiting.106 Major complications are uncommon and include gastrointestinal bleeding, intestinal obstruction and spontaneous displacement of the device. The EndoBarrier device is usually removed after 1 year of placement, and long-term data on diabesity outcomes after initial improvement are still awaited.
Poor sleep hygiene and diabetes
Sleep is a restorative process for both mind and body. There are five stages of sleep: non-rapid eye movement sleep stages 1–4, and rapid eye movement sleep. Of these, the deeper stages of non-rapid eye movement sleep (stages 3 and 4), also called ‘slow wave sleep’, are also the most restorative. Proper sleep is important for normal metabolic and hormonal regulations of the body. Sleep deprivation and poor quality of sleep result in altered metabolic and hormonal function, leading to the development of T2DM. People who sleep less than 6 hours per night are at risk of developing diabetes, and it is advised to have 7 hours of uninterrupted sleep per night.108
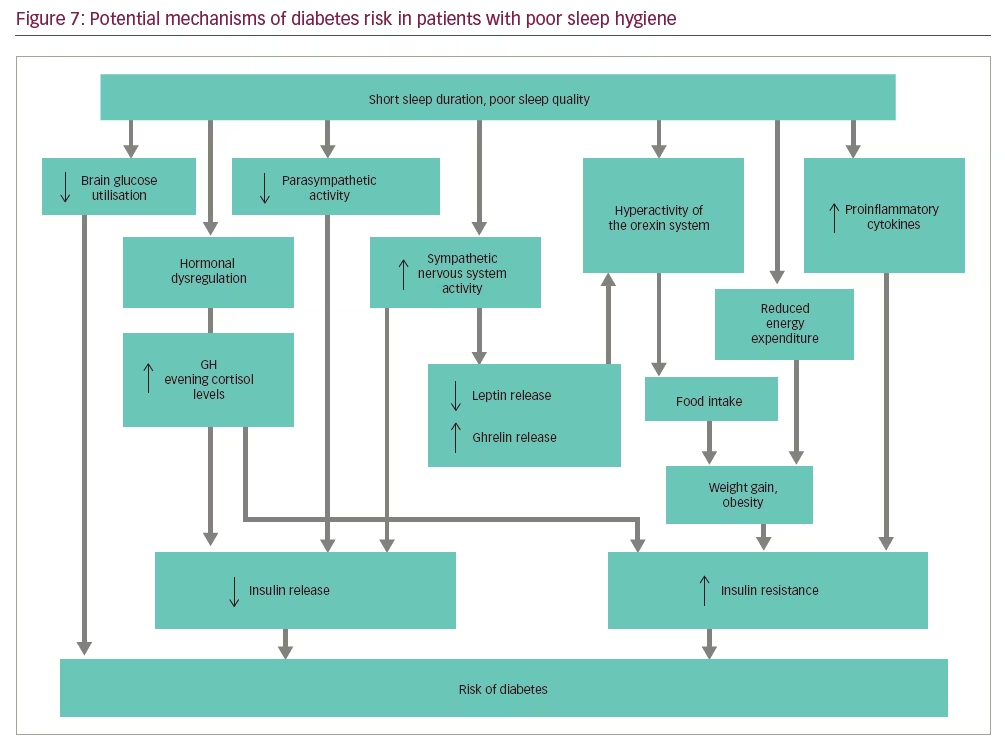
Various mechanisms are described in the development of diabetes in people with sleep deprivation (Figure 7). During sleep deprivation, brain glucose use is reduced, and there is hormonal dysregulation, resulting in overall higher GH levels and evening cortisol levels that may contribute to the development of T2DM.109 Increased sympathetic activity, reduced parasympathetic activity and increased level of inflammatory markers in these cases also may play a role. Sleep deprivation results in weight gain and obesity by multiple mechanisms such as hyperactivity of the orexin system, overeating and reduced energy expenditure.110
Different studies showed that OSA is associated with increased insulin resistance and the development of T2DM, even independent of obesity.111,112 The reported overall prevalence of OSA in T2DM varies from 58–86%, although it is largely an unrecognised problem. Similarly, the prevalence of T2DM in OSA varies from 15–30%. A significant percentage of individuals with T2DM have unrecognised OSA, which results in the worsening of glycaemic control.112 OSA also increases cardiovascular morbidity. Studies showing the effects of continuous positive airway pressure therapy on glycaemic control give mixed results. However, weight loss significantly improves both OSA and glycaemic control.113–115
Conclusions
Although there are multiple pharmacological treatment options in patients with T2DM, non-pharmacological interventions should always be considered to alter the pathophysiological mechanisms of the disease. Appropriate dietary interventions coupled with regular physical activity are very important in the management of T2DM that not only improves glycaemic outcomes, but also confer benefits for other associated diseases such as dyslipidaemia, hypertension, OSA and cardiovascular diseases. These interventions are also beneficial for patients with T1DM in appropriate clinical settings.
Bariatric surgery is the best treatment option for the management of diabesity.116 Various metabolic surgical procedures are associated with marked improvements in diabesity outcomes, lipid parameters and other comorbidities in these patients. Significant improvements in all-cause and disease-specific mortality rates have been reported in multiple studies. Although bariatric surgery may not confer significant improvements in glycaemic management in patients with T1DM, surgery may be an option in those patients with morbid obesity. Sleep hygiene is identified to be important for the prevention of T2DM and appropriate management of OSA (when co-existing) through weight loss and continuous positive airway pressure are crucial for the management of T2DM.