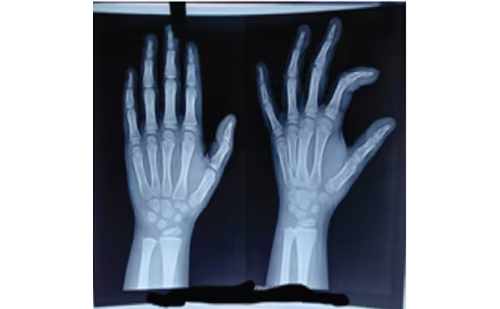
Acromegaly is an uncommon disorder characterized by excess secretion of growth hormone (GH), resulting in the classical growth of bone and soft tissues, multi-system involvement with multiple comorbidities, and heightened risk of premature mortality. GH is produced by the somatotroph cells of the pituitary gland in a pulsatile fashion. Circulating GH stimulates hepatic secretion of insulin-like growth factor-1 (IGF-1). Both GH and IGF-1 circulate, and are responsible for the exaggerated somatic growth characteristic of this disease.
Acromegaly is an uncommon disorder characterized by excess secretion of growth hormone (GH), resulting in the classical growth of bone and soft tissues, multi-system involvement with multiple comorbidities, and heightened risk of premature mortality. GH is produced by the somatotroph cells of the pituitary gland in a pulsatile fashion. Circulating GH stimulates hepatic secretion of insulin-like growth factor-1 (IGF-1). Both GH and IGF-1 circulate, and are responsible for the exaggerated somatic growth characteristic of this disease. Over 90% of cases of acromegaly are due to an adenomatous growth of the pituitary somatotroph cells. These adenomas usually occur in a sporadic fashion, but uncommonly can be part of a familial multiple endocrine neoplasia (MEN-1) syndrome associated with parathyroid and pancreatic disease, or as isolated, familial acromegaly. There are several effective treatment modalities to control this disorder and reduce or prevent the associated morbidity and mortality. This article will review the clinical approach to acromegaly and highlight the currently available therapies.
Clinical Presentation
Men and women are affected equally by this disease, and present at a mean age of 40 years. Signs and symptoms of acromegaly are attributable either to GH hypersecretion, or to localized mass effects of the tumor itself. The classic features of GH excess include frontal bossing, enlarged lips and nose, prognathic jaw, increased spacing of the teeth, enlarged tongue, changes in voice, oily skin or excess acne, and enlarged hands and feet. Other than menstrual irregularities in women, most patients with underlying acromegaly do not present with complaints or symptoms of somatic overgrowth. Rather, acromegaly is most commonly detected incidentally. Because acromegaly is an insidious disease, it can go undetected for a decade or more prior to diagnosis. Therefore, GH-secreting pituitary adenomas are generally greater than 1cm (macroadenoma) at the time of initial presentation and frequently cause multiple systemic comorbidities resulting from chronic GH excess. GH hypersecretion is associated with carpal tunnel syndrome, hypertension, type 2 diabetes mellitus, cardiomegaly, obstructive sleep apnea, and painful joint destruction. Control of GH hypersecretion and normalization of IGF-1 can dramatically improve these medical comorbidities. Signs and symptoms of local tumor invasion include headache, visual compromise due to involvement of the optic chiasm or cavernous sinuses, or hypopituitarism due to compression of the normal gland.
Some retrospective studies have suggested an increased incidence of malignancy in patients with acromegaly, particularly of the colon.1 There is more definitive evidence supporting an increased risk of pre-malignant colon polyps, detected in up to 30% of patients.2 Whether the risk of colon cancer and/or polyps is improved with biochemical disease control is unknown. Nevertheless, a baseline screening colonoscopy to exclude colon cancer may be warranted.
Untreated, acromegaly causes an approximately two- to four-fold increase in mortality due to cardiovascular and respiratory complications.3 Biochemical control, including reduction of serum GH to <2.5ng/ml and normalization of IGF-1, can negate this risk.4,5 Therefore, control of acromegaly has critical implications with regard to preventing the long-term medical and mortality consequences of the disease.
Diagnosis
A biochemical evaluation should be undertaken to confirm suspected acromegaly. Since GH levels are episodic throughout the day, random measurements are rarely useful. An oral glucose tolerance test (OGTT), utilizing the normal ability of glucose to suppress GH, is the gold standard laboratory test to confirm or exclude this disease. Failure to suppress GH levels to <1ng/ml after OGTT suggests the diagnosis of acromegaly. Since IGF-1 is an integrated marker of GH secretion, an elevated IGF-1 level, compared with an age and gender normative data range, is supportive of the diagnosis. Following biochemical diagnosis, a magnetic resonance imaging (MRI) scan should be performed to determine the presence of a pituitary adenoma, and to assess for local mass effects. If a macroadenoma is present, it is important to evaluate for hypopituitarism, especially adrenal insufficiency (which needs to be rapidly treated), and to assess for visual field compromise.
Treatment
Goals of Therapy
The specific goals of treatment are to:
- normalize disease markers (GH and IGF-1);
- slow or reverse the clinical signs and symptoms;
- preserve normal pituitary function; and
- restore life expectancy to that of the general population.
Although controversial, most experts agree that disease control is defined by normal serum IGF-1 levels and attainment of a safe GH level, such as mean basal serum GH <2.5ng/ml or a GH level following an OGTT of <1ng/ml.6,7 There are three major treatment modalities to achieve these end-points—surgery, medical therapy, and radiation, which are described in detail below.
Surgery
Transphenoidal surgery is the primary modality of therapy in the majority of cases because it can yield a rapid cure and correct local mass effects. Transfrontal craniotomy is uncommonly utilized as the surgical approach for more aggressive, invasive tumors. The cure rate for a well-circumscribed intrasellar microadenoma (<1cm) is approximately 90%.8 In contrast, surgical efficacy rates for macroadenomas are lower and range from 30% to 70%. Cure rates depend on tumor size and location (with lower efficacy rates in the presence of extrasellar involvement, including cavernous sinus invasion), pre-surgical GH levels, and surgical expertise.9,10 Complications of surgery are infrequent but include visual impairment,meningitis or cerebrospinal fluid leak, anterior or posterior pituitary hormone dysfunction, and local nasal complications. Following surgery, subjects should undergo repeat biochemical testing with serum IGF-1 and basal GH levels at approximately 8–12 weeks to determine surgical efficacy. Since most patients with acromegaly have a macroadenoma at the time of presentation, many do not attain a surgical cure, and in these cases additional therapeutic options may be necessary. Re-operation is considered for residual disease, but is usually ineffective in the presence of extrasellar tumor invasion. Therefore, further adjuvant therapy is recommended for such patients.
Medical Therapy
Medical therapy is usually considered in an adjuvant role for patients with residual disease following surgery. There are three classes of medical options—somatostatin analogs, dopamine agonists, and a GH receptor antagonist (see Figure 1).
Somatostatin Analogs
Somatostatin analogs (SAs) (octreotide and lanreotide, though octreotide alone is currently available in the US) bind to somatostatin receptor (SSTR) subtypes 2 and 5 on somatotroph adenomas to suppress GH release. Octreotide is most commonly administered as a long-acting release (octreotide LAR) with monthly intramuscular (IM) injections, and lanreotide is available as a monthly subcutaneous (SC) autogel formulation. SAs result in GH control and normalization of serum IGF-1 levels in approximately 50–70% of cases, although this number may be exaggerated as many studies pre-select subjects for somatostatin analog responsive tumors.11 Factors that influence GH/IGF-1 response to SAs include tumor size and GH levels; SAs are less effective with larger tumors with more GH hypersecretion.12 In the adjuvant role, SAs shrink tumors in only 20–40% of subjects, and the degree of tumor reduction is usually modest.13,14 Side effects of SAs include gastrointestinal upset (which usually improves over time), gallstones in up to 40%, hair loss, bradycardia, and hyperglycemia.
Dopamine Agonists (DA)
Though DAs reduce GH levels, there is a limited therapeutic role for these drugs in the management of acromegaly. Bromocriptine normalizes IGF-1 values in approximately 8% of cases.15 In contrast, cabergoline (a non-ergot, D2 receptor-specific dopamine agonist) administration can normalize IGF-1 levels in up to 39% of cases.16 In a study by Abs et al.,16 those patients with mild biochemical disease activity and/or hyperprolactinemia responded better to therapy. These data suggest that cabergoline may be more effective than bromocriptine. Other studies have not confirmed the prognostic value of prolactin co-production by a GH-secreting adenoma in predicting successful DA response.17 Some advantages of DAs include the availability of oral formulations, and the relative low cost compared with other modalities. In summary, DAs may be considered as an adjuvant medical option, primarily in subjects with limited symptoms and modest biochemical disease. It has also been suggested that the addition of DAs to SAs may have additive effects, and may be considered in patents with limited SA responsiveness.17
Growth Hormone Receptor Antagonist
The GH receptor (GHR) antagonist pegvisomant is the newest medication approved for the treatment of acromegaly. This engineered human GH molecule has enhanced binding to the GHR and results in functional blockade of GH-mediated intracellular signaling. This results in a reduction in circulating serum IGF-1 that is long-lasting, and is associated with improvement in soft tissue enlargement and quality of life.18 In a randomized, double-blind, placebo-controlled 12-week study, pegvisomant administration resulted in a dose-responsive reduction in IGF-1, with normalization of IGF-1 levels in 89% of subjects.19 Serum GH levels can increase in response to pegvisomant, and therefore should not be measured as a disease marker in subjects receiving this medication. There is concern that the increase in GH levels may reflect growth of the pituitary tumor, and there have been rare reports of tumor growth. Longer clinical follow-up is necessary to determine the clinical ramifications of this finding, though serial MRI scanning should be performed. Pegvisomant administration has favorable effects on glucose homeostasis, including a reduction in insulin and glucose levels, and therefore may be particularly useful in the setting of acromegaly associated with type 2 diabetes mellitus.
Primary Medical Therapy—Evolving Therapeutic Paradigms
Primary medical therapy for acromegaly, either as preoperative therapy to improve surgical outcome, or as de novo therapy, has been suggested as an alternative to traditional paradigms. Although there are reports of improved surgical outcome following SA therapy, there are no controlled studies that demonstrate this. Therefore, SAs are recommended pre-operatively only to improve significant comorbidites or when surgery is delayed. For example, pre-operative SA therapy can be offered to patients with comorbidities that increase the anesthetic risk, such as retropharyngeal thickness that may complicate intubation, severe hypertension, or uncontrolled diabetes. There has been much interest in the utility of SAs as de novo acromegaly therapy, especially in treating macroadenomas without associated localized mass effects, particularly visual field loss. This option is supported by several studies showing that SAs have similar efficacy in controlling biochemical secretion whether used in a primary or adjuvant role.20 In an open-label, prospective, randomized trial, Bevan et al. showed that daily SC octreotide followed by monthly octreotide depot injections for up to 48 weeks normalized IGF-1 in 53% and GH in 79% of their previously untreated patients.14 Similar studies have also demonstrated that de novo SA therapy results in tumor shrinkage to a greater degree and in a greater percentage of subjects than noted with adjunctive use.21 Though there are no controlled studies to date showing that SA therapy is equivalent or superior to surgery in a primary treatment role, there are sufficient data to suggest that primary medical therapy may be offered safely and with benefit to selected patients. Recent consensus guidelines suggest that medical therapy may be considered in lieu of surgery for tumors not causing local mass effects, and this decision should include discussion of cost, operative risk, and patient preference.7
Radiation Therapy
Radiation therapy is considered as an adjuvant option for patients who have failed surgery and/or are unresponsive to or poorly tolerant of medical therapy. Post-operative conventional fractionated radiotherapy controls the disease in 5–78% of subjects but may take many years for effect.22,23 Additional concerns of radiation include the risk of hypopituitarism, damage to optic structures, cerebrovascular disease, and the rare occurrence of secondary tumors. Stereotactic radiosurgery (SRS) is able to deliver lower quantities of radiation more precisely than conventional radiation. Some studies suggest SRS may control disease sooner than conventional radiation.24 For example, in a prospective study of 82 subjects with active acromegaly, 63 of whom had previous transsphenoidal surgery, gamma knife SRS resulted in remission in 17% of subjects at a mean follow-up of 49.5 months. An additional 23% of subjects previously uncontrolled by SAs achieved disease control with SAs after gamma knife.25 Further studies are important to determine the overall role of SRS in the management of acromegaly.
Conclusion
Acromegaly is a disease characterized by GH hypersecretion, and is associated with multiple medical comorbidities and premature mortality. Successfully treated, life expectancy in acromegaly can be restored to normal. There are several effective treatment modalities available, with evolving paradigms in their use. With recent advances in the management of acromegaly, disease control can be reasonably expected in the majority of patients. ■