Given the global nature of the diabetes epidemic,1 this condition is of public health importance in Sweden.2 National prevalence in Sweden is approximately 7.0%, based on a recent estimate,1 and approximately 90% of cases are type 2 diabetes (T2D).1,3 This prevalence figure, which is growing over time despite stable incidence,4 has a direct impact on overall population health. In 2015, over 3,000 deaths were attributed to diabetes in Sweden.1 Moreover, patients with diabetes are at higher risk for disabling or life-threatening health problems than non-diabetic patients.5
A high burden exists from an economic perspective, as well. Recent figures indicate that Sweden spent over US$8,000 per diabetes patient annually on managing diabetes.1 In addition to the costs of managing diabetes, poor glycaemic control is associated with higher additional healthcare costs, both to treat short-term hypoglycaemic events and to manage longer-term macro- and microvascular complications.6,7 The total cost for identified patients with T2D in the Prescribed Drug Registry in 2014 was nearly €1.3 billion,8 and this does not account for any additional societal burden associated with lost productivity or caregiver time.1,9,10
Some patients with T2D may require insulin when diet and exercise and other medications become inadequate to control blood glucose.1 The trade-off between insulin helping to reduce glycated haemoglobin (HbA1c) and an increased risk of hypoglycaemic events or related complications has been well-established.11 Frequent monitoring of glucose levels allows patients to more appropriately manage blood glucose.1,12 Self-monitoring of blood glucose (SMBG) is the current standard of care for glucose monitoring; the European Consensus Statement recommends 4–8 times daily,13 while the American Diabetes Association (ADA) suggests 6–10 times per day for patients on intensive insulin therapy.14 However, many people do not monitor glucose at these recommended rates.15–18 SMBG adherence may be negatively impacted by factors such as discomfort with obtaining a blood sample, inconvenience of carrying testing kits around, perceived social stigma, needle phobia, and difficulty in interpreting results.19–23 Furthermore, SMBG provides data for only a single point in time, conveying relatively limited information on glucose levels and variability, which can impact clinicians’ recommendations for therapy change.24 Although continuous monitoring is more informative, it is not universally reimbursed and applied due to expense.23,25
Flash glucose monitoring, an alternative to traditional blood glucose monitoring, uses a sensor worn by the patient to provide continuous collection of interstitial glucose data. Passing the system’s reader over the sensor at any time will show the current reading, a trend arrow, and the past 8 hours of data. FreeStyle Libre™ (Abbott Diabetes Care, Witney, UK), a flash glucose monitoring system, was recently tested in an intensively insulin-treated T2D population in the REPLACE study.26
The REPLACE trial was a 6-month, multicentre, randomised controlled trial of the flash monitoring system versus SMBG in adults with T2D and HbA1c 7.5–12% (58–108 mmol/mol), who were using multiple daily injection therapy or continuous subcutaneous insulin infusion for at least 6 months prior to enrolment. The study found clinical benefits such as a 27.7% reduction in measured hypoglycaemia <70 mg/dl for flash monitoring compared to SMBG, without raising HbA1c levels.26
Given potential clinical benefits of intervention and potential associated decrease in resource utilisation, this study sought to assess the value of investment in using flash monitoring instead of routine SMBG via cost-effectiveness analysis for patients with T2D receiving intensive insulin treatment in Sweden.
Research design and methods
The present study was performed using version 8.5 of the IQVIA CORE Diabetes Model (IQVIA CDM).
IQVIA CDM summary description
The IQVIA CDM is a non-product specific internet application to assess the long-term health outcomes and economic consequences of interventions for Type 1 diabetes (T1D) or T2D. The underlying mathematical engine includes diabetes complication sub-modules that combine Markov techniques with Monte Carlo simulation, running simultaneously to capture outcomes associated with the treatments of interest. These sub-modules interact, and each patient profile is updated at the end of each 1-year cycle to account for the events across all sub-modules. The model captures differences in life expectancy, quality-adjusted life years (QALYs), costs, cumulative incidences of complication events due to intervention effects on diabetes-related adverse events, as well as HbA1c levels and other physiological parameters that affect risks of major diabetes complications. The model has been published previously in detail, and it has been extensively validated against clinical and epidemiological studies.27,28
For this study, HbA1c progression reflects the Swedish National Diabetes Registry, while HbA1c-dependent adjustments reflect the United Kingdom Prospective Diabetes Study risk engine.29–31
Analyses took a Swedish societal perspective, evaluating costs and effects over a lifetime horizon (40 years, approximate lifetime from age at model entry). Swedish non-specific mortality information from the World Health Organisation was included.32 Costs and effects were discounted at 3% according to Swedish guidance.33 All analyses were run with 1,000 patients for 1,000 iterations.
Model inputs
Cohort details
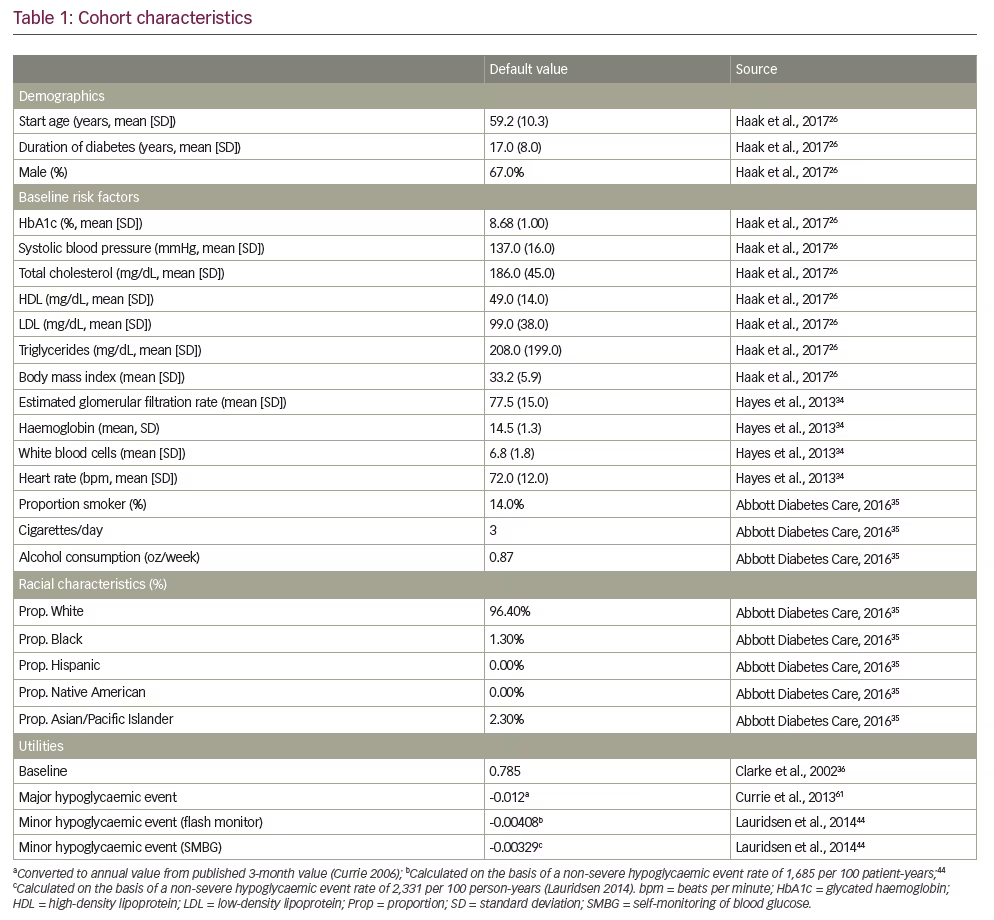
The model cohort was designed to represent the REPLACE trial population (Table 1),26,34–36,44,61 to ensure appropriate alignment with intervention effects. Some cohort characteristics were unavailable in the trial data, and therefore published sources were used to supplement as needed.
Intervention effects
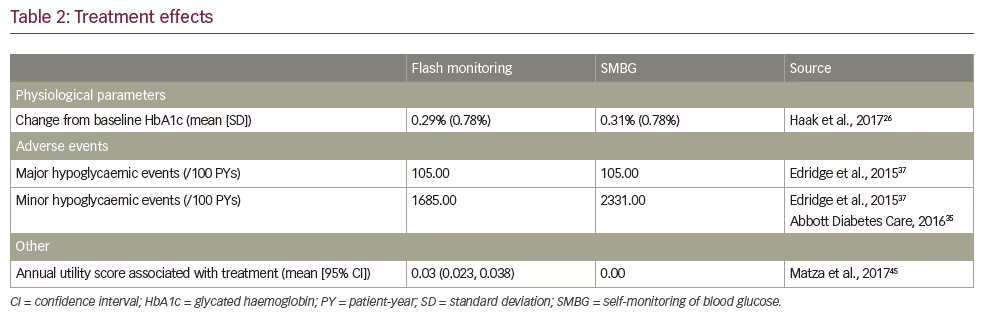
Intervention effects used in analyses are found in Table 2. Although no significant differences were seen in effect on HbA1c in the REPLACE trial, trial-based decreases by end of study of 0.29% (standard deviation [SD] 0.78%; 3.19 mmol/mol [SD 8.58]) in the flash monitoring arm and 0.31% (SD 0.78%; 3.41 mmol/mol [SD 8.58]) in the SMBG arm were implemented into the model.
Hypoglycaemia event rates were likewise implemented as intervention effects; the analysis assumes major (or severe) events require third-party intervention, and minor (or non-severe) events require no outside care. To ensure alignment with available data on costs of hypoglycaemic events, rates were obtained from the literature. The rate for major hypoglycaemic events from a recent meta-analysis was used for both the flash monitoring and SMBG arms;37 no difference was assumed for the purpose of the model because the REPLACE study was not designed to detect differences in safety outcomes such as severe hypoglycaemia. The minor hypoglycaemia rate reported in the same meta-analysis was used for SMBG. Because the REPLACE trial demonstrated a relative effect on non-severe hypoglycaemia due to flash monitoring, this relative reduction was applied to the meta-analysis minor hypoglycaemia rate to calculate a flash monitoring rate.
Intervention-related resource utilisation
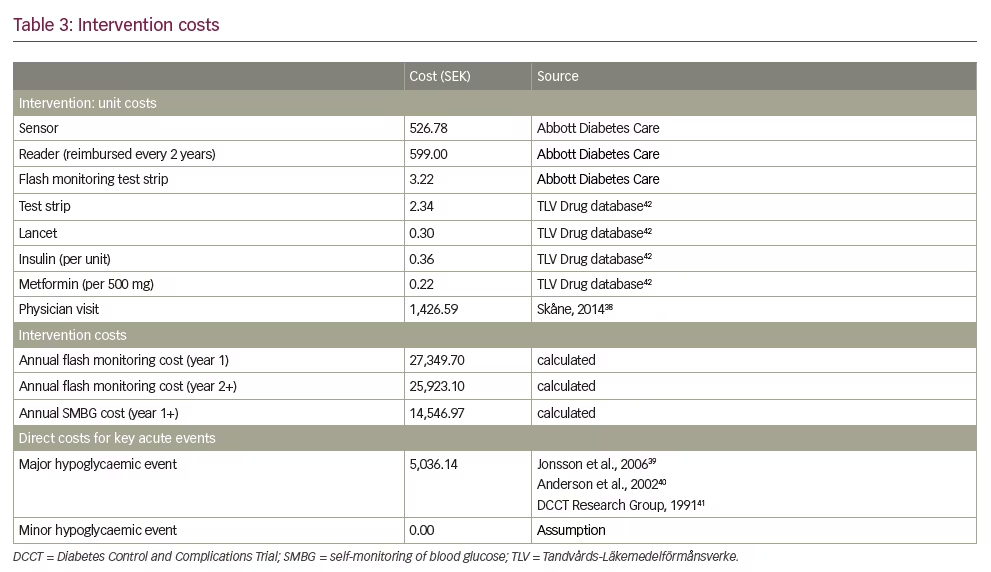
Intervention-related resource use was derived from the REPLACE trial. For patients using flash monitoring, the analysis assumed 109.5 test strips/year, 251.85 lancets/year, and 85.2 units of insulin/day, as well as 26 sensors per year and an extra physician visit in the first year. In the SMBG arm, parallel resource use assumptions included 1,095 test strips/year, 459.9 lancets/year, and 87.8 units of insulin/day. All patients received 1,500 mg of metformin daily.
Unit costs
Table 338–42 shows the key cost inputs for the analyses, including intervention-related unit costs, total intervention costs, and costs for key acute events; Appendix 1 shows the full list of costs used in the analyses. Intervention-specific consumables reflect lowest-cost items available from Tandvårds-Läkemedelförmånsverke (TLV),42 and all costs were inflated as needed to 2016 currency using the consumer price index for Sweden from the Organization for Economic Co-operation and Development.43
Utilities
Utilities and disutilities (Table 1) were based on published literature. For the minor hypoglycaemic disutility, values were separately calculated for the flash monitoring and SMBG arms using a diminishing disutilities approach from Lauridsen et al., 2014.44 The literature shows that for the first few minor hypoglycaemic events, patients experience relatively high disutilities; as the patient has more events, the disutility per event diminishes. Therefore, the average disutility per event is contingent on the total rate of minor hypoglycaemic events. A treatment-related utility benefit of 0.03 was applied to the flash monitoring arm based on a recent time trade off study.45 Sensitivity analyses explored this concept further.
Analyses
The base case analyses compare flash monitoring use against routine SMBG use, utilising default inputs from Table 2. Additional scenario analyses (summarised in Table 4) explored the impact of key model assumptions. These include a scenario to test the impact of incorporating non-intervention resource utilisation as observed in the trial in the first year of the model. In this scenario, all-cause trial-based use of resources such as ambulances, emergency room visits, and hospitalisation were implemented. Among intensive insulin users it is not possible to establish whether resource use is disease-specific due to older age and increase in comorbidities and complications,46–48 and thus all-cause resource use is appropriate for scenario evaluation. In the event that these differences were driven by hypoglycaemic events, the assigned hypoglycaemic event costs were removed from the model to avoid potential double-counting.
Additional scenarios examined the impact of varying the utility benefit for flash monitoring over a range of 0.023–0.038 (95% confidence interval around base case), and to explore the impact of varying discount rates and time horizon. A final scenario leveraged cross-sectional real-world evidence from 50,000 flash monitoring sensors,49 rather than trial-based values. This evidence showed that average scan frequency is 16/day for flash monitoring users, in comparison with an average of 2.7 tests/day for T2D SMBG users.12 A measured difference of 0.94% between the average HbA1c associated with 16 scans/day and that associated with typical SMBG use was used to explore this potential additional benefit of flash monitoring. This reflects an assumption that flash monitoring may lead to increased glucose testing and result in decreased HbA1c.
Results
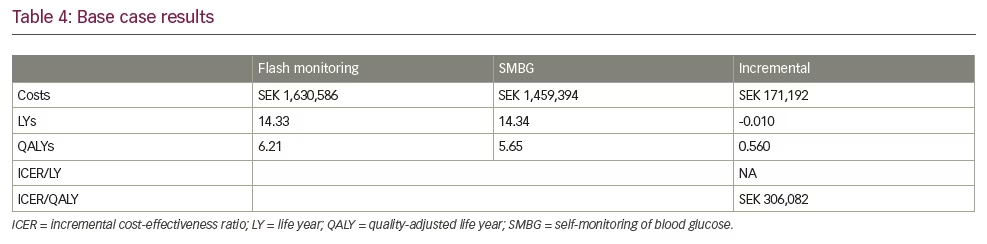
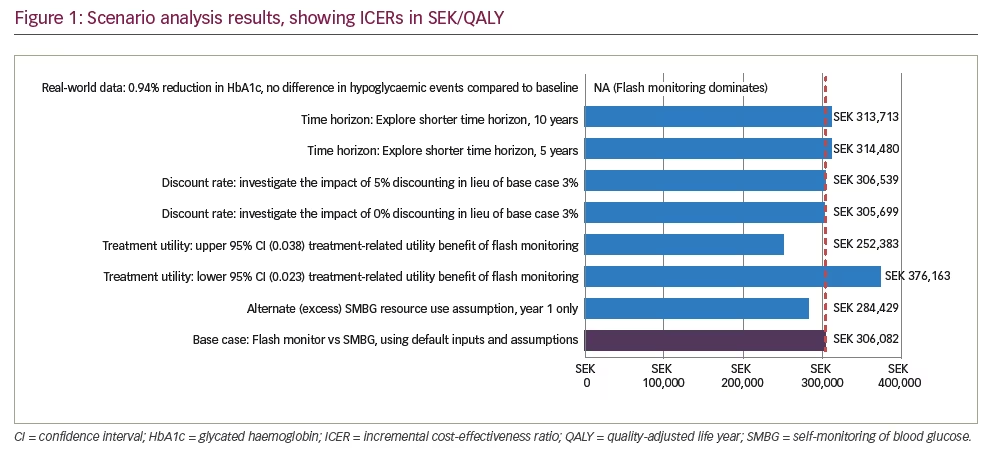
Disaggregated results, including total costs and QALYs per strategy, are reported in Table 4. In base case analysis, a difference of SEK171,192 in direct medical costs were attributable to flash monitoring (SEK1,630,586) for flash monitoring versus SEK1,459,394 [€141,902] for SMBG). Total life years (LYs) were nearly equivalent (14.33 and 14.34 LYs for flash monitoring and SMBG, respectively), with the minor difference associated with the insignificant difference in HbA1c results from the REPLACE trial. When incorporating quality-adjustment, however, flash monitoring patients experience 0.56 more QALYs (6.21 versus 5.65, respectively) for an incremental cost-effectiveness ratio (ICER) of SEK306,082/QALY (€29,762/QALY). In terms of cost per hypoglycaemic event averted, the difference of 137.22 more minor hypoglycaemic events for SMBG patients led to a cost per minor event averted of SEK1,248.
Key scenarios that were evaluated to test the impact of assumptions or input values are reported in Figure 1. Flash monitoring dominates when real-world evidence is considered in the model. The highest ICER occurs when testing a lower potential utility benefit associated with flash monitoring.
Discussion
This first cost-effectiveness analysis of flash monitoring in patients with T2D using intensive insulin demonstrates that use of the flash monitoring system is associated with a modest impact on diabetes-related costs, and can be considered cost-effective compared to current standard of care for glucose monitoring (SMBG). Although SMBG is less costly overall (by SEK171,192), flash monitoring improves QALYs for patients, leading to a favourable cost-effectiveness ratio of just over SEK300,000/QALY. Continuous glucose monitoring systems may also be available for these patients; although, on an annualised basis, the list price of these systems is typically several times greater than the flash monitoring system.50,51 Flash monitoring may be considered good value for money regardless of type 1 or type 2 status for patients using this therapeutic approach, as this analysis joins evidence on the economic value for patients with T1D using intensive insulin.52 In the clinical trial setting, use of the system substantially reduced the number of hypoglycaemic events, including at low glucose thresholds (<55 mg/dL and <40 mg/dL), without raising HbA1c across populations. The observed clinical benefits may reflect improved management by patient access to convenient glucose measurement data.53
Through additional scenario analysis, results are robust to alternate assumptions. ICERs remained under published willingness-to-pay threshold ranges for Sweden. Although Sweden does not publish an explicit threshold, interventions have been accepted with an average ICER of €36,000/QALY,54 which is approximately SEK400,000 /QALY, and an ‘informal threshold’ of SEK500,000 has been identified in publications.55 Treatments for more severe conditions have been considered acceptable up to €90,000/QALY (SEK827,000/QALY).54,56
A number of limitations for this study must be acknowledged. The analysis assumes that non-severe hypoglycaemic events are not associated with the occurrence of other more severe events like severe hypoglycaemia, myocardial infarction and mortality. However, in the PREDICTIVE study, a high frequency of non-severe events was significantly associated with the occurrence of severe events;57 this would be particularly important to consider in an older population for whom avoiding hypoglycaemia is a therapeutic goal.58
Additionally, these analyses may not capture all health outcomes. For example, the risk of cardiovascular disease in the IQVIA CDM is based on HbA1c, together with lipid levels, blood pressure, co-morbidities, and body mass index. However, recent evidence has suggested that people experiencing hypoglycaemic events may be at an increased risk of cardiovascular disease.59,60 Although the baseline characteristics in that study may differ from our analysis, it raises the possibility that our analysis underestimates the value of reducing hypoglycaemic events.
The main clinical data and patient characteristics are taken from a 6-month trial, and may not exactly represent the real-world effects of the flash monitoring system or represent the T2D patient population using the flash monitoring system in the real world. However, there were no protocol-mandated monitoring or adjustments to therapy, and therefore the results may be considered generalisable. Supplemental exploratory analysis based on cross-sectional real-world data49 from patients using flash monitoring also suggest that results may be even better than implemented in the base case due to potential HbA1c benefit in real-world use. Additionally, the trial-estimated reduction in hypoglycaemic events was based on sensor identification rather than symptoms; however, for this reason, the reduction in events was applied solely to the non-severe event rate, and that base rate was derived from the literature.
Our analysis simplified the treatment pathway faced by patients by assuming that glucose monitoring and insulin use do not change over time. Yet, in the absence of data, typical modelling practice is to assume that there is no difference associated with treatment; any insulin change applying to both strategies equally would not alter the conclusions of this study.
Additionally, current utility values may not fully represent the impact of using flash monitoring. The disutility associated with minor hypoglycaemic events is assumed to reflect the diminishing effect of each event as they become more frequent, as has been shown in recent research.44 However, the average value per event applying this technique is much smaller than that used in prior economic analyses,61 and therefore, the values used in this study are likely to be more conservative but also more realistic relative to other published values. Separately, in the treatment phase of REPLACE or during the subsequent open-access phase of 6 months’ duration,62 no safety concerns with flash monitoring were observed. Skin reactions were reported for nine (6.5%) participants during the open-access phase and six participants (4.0%) in the treatment phase (preceding 6 months). Based on this information, potential skin reactions are not considered to have a material impact on the results presented here.
Despite the limitations, this analysis provides insight into the economic value of flash monitoring use compared with SMBG in T2D intensive insulin users. With limited impact on overall costs yet substantial relative QALY improvements, as well as robust results across tested scenarios, flash monitoring may be considered cost-effective in a Swedish population among T2D patients using intensive insulin.







