Historical Perspective
Historical Perspective
Growth hormone deficiency (GHD) was recognized only after the discovery of GH in 1921; however, this form of proportional dwarfism had been described much earlier. In the 1950s, GH isolated from the pituitaries of humans and anthropoid apes was discovered to stimulate growth in children who had GHD—unlike insulin—bovine or porcine GH had no activity in human or other primates. The first report of GH therapy for GHD was in 1958.1 From 1958 to 1985, a limited supply of this cadaver-derived pituitary GH was used to treat about 8,000 children who had GHD in the US. The preparation was always in short supply, resulting in lower-thanideal dosing and frequent drug holidays. In order to ration the cadaveric GH, the diagnosis of GHD required that patients’ peak GH response to provocative stimuli not exceed a certain serum concentration. This limit gradually increased along with the supply of cadaveric GH, starting at 5 ng/mL, then 7 ng/mL, and finally 10 ng/mL in the early 1980s. In 1985 this preparation was linked to a risk for Creutzfeldt-Jacob disease,2,3 and its use was discontinued. Beginning in 1979, GH was produced in large quantities by expressing the human GH gene in Escherichia coli.4 Recombinant human GH (rhGH) was approved by the US Food and Drug Administration (FDA) in 1985, thus solving the GH supply problem, while replacing it with the economic burden of a very expensive treatment.
Because of new understanding of the GH–insulin-like growth factor (IGF) axis, GHD is now seen as one disorder on a continuum from failure of the pituitary gland to secrete adequate GH for growth, to problems with GH receptor binding or GH action leading to Growth Hormone Insensitivity Syndrome (GHIS, including Laron Syndrome), to problems with IGF-I secretion.5 This article is limited to a discussion of advances in the treatment of GHD.
Diagnosis of Growth Hormone Deficiency
The hallmarks of classic GHD include extreme proportional short stature with a very slow growth velocity and a delayed bone age. Children with GHD have been described as looking like cherubs from the paintings of Rubens or like Kewpie Dolls. Children with classic GHD often have highpitched voices. Hypoglycemia, especially in infancy, may result from GHD alone or from absence of GH along with absence of adrenocorticotropic hormone (ACTH) if there is wider pituitary insufficiency. Also, boys with pituitary insufficiency may present with micropenis because of the lack of gonadotropins. With increasing GH supply the severity of children diagnosed with GHD has lessened; thus, at the present time many children with the diagnosis of GHD do not appear as severely affected as children with classic GHD diagnosed in the past. Evaluation of 20,000 children treated with rhGH between 1985 to 1994 showed that 44 % had idiopathic GHD, while 13.8 % had organic GHD.6 Of those with organic GHD a small subgroup have GHD as a part of a larger complex of pituitary deficiencies. Most common is septo-optic dysplasia, which may include varying degrees of hypopituitarism. Also in this group are rare inherited syndromes of hypopituitarism caused by mutations in the transcription factors prophet of Pit-1 (POU1F1) (homologous to the mouse gene Pit- 1) and PROP1, so named because it was recognized to be related to POU1F1.7 Another rare cause of organic GHD is caused by a problem with the gene coding for GH. These are classified as “severe familial GHD”, and are discussed below.
Diagnosis of GHD should first depend upon the clinical picture. Children who should be evaluated for GHD include children who are especially short (height >3 standard deviations [SDs] below the mean for age), and/ or who have growth deceleration (growth velocity <2 SD), as well as those who are less severely short (height between –2 and –3 SD), but growth deceleration (growth velocity below –1 SD), who have a history of a brain tumor, cranial irradiation, or another organic pituitary abnormally, or who have radiologic evidence of a pituitary abnormality.8 The most common screening tests for GHD include measurement of levels of IGF-I and its binding protein, IGFBP-3. It is important to evaluate levels based on normal values for age or puberty stage. Another weakness of these measurements is disagreement in values between many of the commercially available assays. In addition, low IGF-I levels can result from other factors, such as malnutrition or chronic disease.9 Low IGFBP-3 levels are more likely to indicate GHD, although there is considerable overlap between low and normal values. Nonetheless, if both IGF-I and IGFBP-3 levels are low, GHD should be suspected, especially if consistent with the clinical picture. Although some pediatric endocrinologists have argued against GH provocative testing in the diagnosis of GHD,10 it remains a mainstay for most pediatric endocrinologists, and may help to identify those patients who have more extensive pituitary problems, as well as those who are most likely to need GH replacement as adults. At the present time, the diagnosis of GHD requires that all GH values in response to two provocative tests should be less than 10 ng/ml.
Magnetic resonance imaging (MRI) studies on children who are diagnosed with GHD have reported that between 12 % and 96 % have a pituitary abnormality.11 Explanations for this variation have included the heterogeneity of the GH-deficient population, inconsistency in the use of contrasted MRI (for delineation of the pituitary stalk), and variability in interpretation. The likelihood of finding an abnormality appears to increase with the severity of GHD, with the finding of ectopic neurohypophysis most associated with GHD. Failure to visualize the pituitary stalk is associated with more severe deficiency of GH as well as multiple pituitary hormone deficiencies. Generally, it appears useful to perform an MRI on any child who is diagnosed with severe GHD. In patients with suspected or partial GHD, MRI may aid in the diagnosis. When MRI is performed, the use of gadolinium contrast is recommended to better delineate the pituitary stalk. The Update of Guidelines for the Use of Growth Hormone in Children12 has suggested that GH therapy for GHD be initiated only after an MRI or a computed tomography (CT) scan has excluded an intracranial mass lesion.
Treatment
Treatment with Growth Hormone
Average adult height for untreated patients with severe isolated GHD was about 143 cm in men and 130 cm in women. Present-day commercial preparations all have the identical 191 amino acid sequence of the 22 kilodalton native human pituitary hormone13, and in the US since the late 1980s the usual dose for treating GHD has been 0.3 mg/kg/week.14 In Europe it is more usually dosed at about 0.2 mg/kg/week. Initially GH was injected intramuscularly, but in the mid-1980s (about the time of introduction of rhGH) it was shown to be as effective if given as a subcutaneous injection15, which is the practice today. Early in its use, GH was given twice weekly, but dose frequency was increased to three times weekly when more frequent dividing of the GH dose was shown to result in an increased growth response.16 At about the time of the transition from cadaveric GH to rhGH it was demonstrated that daily (six or seven injections per week) yielded an even better growth response than three times per week,17–20 and daily administration is commonly used today. It is now clear from large databases21–23 that GH-deficient children treated with GH are frequently achieving adult heights in the normal adult range.
A more recent approach has been to adjust the GH dose based on IGF-I levels, rather than relying on weight-based dosing. A study using this paradigm has shown that such an approach may result in increased growth velocities; however, the doses in some subjects were so high as to be prohibitively expensive.24 This method of dosing does provide for safety, however, since it involves frequent monitoring of IGF-I levels.
Compliance Concerns
A common problem with GH therapy has been compliance. Ideally, GH should be taken daily, but since there is little loss of growth from skipping a single dose, patients may be tempted to skip doses. One approach to this problem has been renewed interest in long-acting GH preparations. Such a preparation, which was given once or twice monthly, was in use about a decade ago,25 but for various reasons (some having to do with the logistics of manufacture), the preparation is no longer available. In addition, other problems with this preparation included inconvenience in the size of the injections (occasionally requiring dividing the dose into more than one injection) and the need for a larger needle in order to avoid clumping of the dose in the syringe as the injection was being given. Similar preparations under development have attempted to address some of these earlier problems. It is intuitive that the ideal GH preparation should result in physiologic GH secretion, which would be multiple bursts of GH each day, particularly during sleep,26 However, it should be pointed out that daily GH does not accomplish this. Subcutaneously injected daily GH peaks at about 5 hours after injection and persists for about 24 hours.27 The long-acting GH preparation that was previously available resulted in a large initial peak of GH, followed by a tail that lasted about 2 weeks.25 Careful examination of GH response to this preparation demonstrated that the growth response was most correlated not with the peak GH value or with the complete area under the curve, but with the length of the exposure to GH, that is, with the area under the curve of the tail. Therefore, since the first goal of treatment is a good GH response, the long-acting GH preparation that produces the best effect on growth may not necessarily be the one that mimics the most physiologic secretion pattern. Although there are no FDA-approved long-acting GH preparations currently available, there are several that are in various stages of development.28–31 Most of the long-acting GH preparations under study are designed to be injected weekly.
Growth Hormone Therapy During Puberty
Another challenging aspect of GH therapy is the care of the child who is in puberty.32 Patients with GHD are usually treated until they have reached an acceptable height, or until they experience epiphyseal fusion and are no longer able to respond to GH with linear growth. The usual point for stopping GH because of epiphyseal fusion is an annual growth velocity slower than 2.5 cm per year in a patient who is known to be compliant with the GH injections. The first approach addressing GH treatment during puberty was to increase doses of GH during puberty. This strategy is based on the observation that the normal pattern of GH secretion is that GH levels increase (often at least double) during puberty. Following publication of a study which demonstrated about a 5 cm increase in adult height33 with an increased in dose of GH (0.7 mg/kg/week) compared with the standard dose (0.3 mg/kg/week), the FDA approved the use of the higher dose of GH during puberty. Although not all pediatric endocrinologists use doses as high as 0.7 mg/kg/week in all pubertal patients, it is common to use doses higher than 0.3 mg/kg/week in pubertal patients who have not achieved a height in the normal range.
A second strategy has been to attempt to extend the time for responding to GH with the use of LHRH analogs. It is clear that LHRH analogs can increase adult height in children with precocious puberty, especially when used in very young children.34,35 Studies with GH-deficient patients receiving both GH and LHRH analogs have yielded somewhat mixed results. Changes in height prediction in those treated with GH and LHRH an LHRH analog for 2 to 4 years has varied from 7.9 to 14 cm.34,36,37 LHRH analogs limit progression of puberty, which may extend the time for a GH response. However, the amount of extra growth, while statistically significant, may be small, and the child with GHD is frequently already behind peers in terms of pubertal maturation. Such a child may not feel that trade-off of further pubertal delay for an extra few centimeters to be worthwhile.
A third approach has been the use of aromatase inhibitors in pubertal boys. A case report in 199438 described a 28-year-old tall man with estrogen resistance. He had gone through normal male puberty, but had not closed his epiphyses. This case, along with other laboratory data, confirmed the idea that it is estrogen and not androgen that causes epiphyseal closure. Aromatase inhibitors block conversion of testosterone to estradiol, and cause an increase testosterone levels behind the block. It is because of the increased testosterone levels that there has been reluctance to study these preparations in girls. With aromatase inhibitors there is no stopping of male puberty (male puberty may actually accelerate because of the testosterone build up). The results of these studies are just becoming available;39,40 it appears that treatment with an aromatase inhibitor significantly increases the height prediction in a pubertal male, and there do not appear to be any serious safety signals with its use.
Treatment with Insulin-like Growth Factor
Since its approval by the FDA in 2005, it has been possible to use IGF-I as a treatment for short stature.41 In general its most appropriate application is in treating GH-resistant conditions, such as GHIS or Laron Syndrome. However, there are some specific uses for this therapy in rare cases of severe familial GH deficiency. First described in 1978 in a consanguineous family,42 GHD type IA was determined to be the result of absence of the GH gene.43 Other similar patients have been described,44–57 and the extent of the GH gene deletions vary among the reported cases.
Children with type IA have the severest form of GHD.58 At birth they are short and somewhat obese with a small head circumference. Boys have micropenis. All infants have problems with hypoglycemia. These children grow very slowly, and usually more than 2 SD below the mean in height. They also have delay in motor development. They have a protruding forehead and a saddle nose, and sparse hair. These children test GH deficient and also DNA analysis demonstrates a deletion in the GH gene.
What is particularly characteristic of children with type I GHD is that some of the patients produce antibodies against GH when they are treated. These antibody titers may become quite high—high enough to attenuate their growth response to treatment.45,59 Interestingly, there are patients with type I GHD in whom these antibodies do not develop.46,50 In patients who do not develop significant antibody titers it is possible to treat them with GH. Those patients with high (growth attenuating) antibody titers to GH may be treated with IGF-I.60,61
Several other types of familial GHD have been described; however, most of these respond to GH treatment. Type IB GHD is not as well defined type IA. It appears to be autosomal recessive, and the GH is partial, not complete as in type IA. It was first described in 1994.62 Type II familial GHD is similar to type IB, except that it is autosomal dominant.63,64 Patients with type II familial GHD are not as short as those with type IA58. Type III familial GHD has X-linked inheritance due to deletions (Xq13.3-Xq21.1) or microduplications of certain X regions. Alternatively, there may be a microdeletion of contiguous genes in the midportion of the long arm of the X chromosome.58
Adult Growth Hormone Deficiency
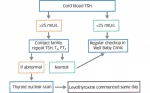
It is now recognized that some GH-deficient children will not make sufficient GH as adults to meet metabolic needs. Features of adult GH deficiency include central (intra-abdominal) obesity, high cholesterol (particularly low-density lipoprotein [LDL]), low bone mineral density, and fatigue. The fatigue is often treated as depression. Only a minority (<10 %) of patients treated for childhood GH deficiency will need GH as an adult. Those at particularly high risk are patients with familial GH deficiency, those with multiple pituitary hormone deficiencies, those with anatomic abnormalities of the pituitary gland or other organic GH deficiency, and those who had particularly low (<3–5 ng/ml) GH responses to provocative testing. It has been our practice to have patients who have complete GH therapy as children return in 6 months to one year to evaluate potential signs of adult GH deficiency. We measure IGF-I and cholesterol levels at that time. If there are signs or symptoms of adult GHD or a high cholesterol or low IGF-I, repeat GH stimulation testing can be performed. Patients with multiple pituitary hormone deficiencies or anatomical abnormalities involving the pituitary gland may not require re-testing as adults.
Safety of Growth Hormones
GH adverse events have been carefully documented in reviews of GH therapy.65,66 Most adverse events have been local injection site reactions, which rarely lead to discontinuation. Headache, nausea, and fever have been generally self-limiting and are well tolerated. Adverse events such as edema or carpal tunnel syndrome are seen more often in adults than children, and may be the result of fluid retention caused by GH.67 Adverse events seen particularly in children have included transient idiopathic intracranial hypertension (IIH, also known as pseudotumor cerebri), gynecomastia, and slipped capital femoral epiphysis.68,69 The IIH usually resolves after discontinuation of GH and re-starting at a low dose.
Overall, GH has been shown to be a safe hormone when used at recommended doses. There are excellent large databases for evaluation of possible safety signals that occur during treatment with GH. Mortality in children with GHD evaluated while they are taking GH is due almost entirely to other pituitary hormone deficiencies, the underlying cause of the GHD (e.g. a brain tumor), or treatment for the underlying condition (e.g. irradiation).70 Patients who had previous radiation are at risk for a second neoplasm if taking GH. There has been recent concern from analysis of data in French children treated with GH between 1985 and 1996, and then followed until 2009 (the Sante Adulte GH Enfant [SAGhE)] study).71 A retrospective analysis of mortality in this population suggests the possibility of increased cardiovascular disease and bone tumors in adults who received GH as children. The cardiovascular disease was primarily attributed to subarachnoid or intracerebral hemorrhages. Overall cancer mortality rates were not higher than the general population, but bone tumor-related deaths were five times higher than expected. Risk was highest in patients receiving doses >50 mcg/day. The study is flawed by not having a control group (data from those who took GH as children was compared with the population at large, which may not be an appropriate comparison). In addition, there was no apparent relationship with duration of GH therapy, which one would expect if the increase in mortality was actually related GH therapy, suggesting that the increase in mortality in this group could be related to the reason they were short and taking GH, rather than an effect of the GH itself.
Similar data from Sweden, the Netherlands, and Belgium72 have shown no increase in mortality rates and all of the deaths in this cohort were attributable to accidents or suicide, further suggesting that the French data may be misleading. Clearly, what is most needed is long-term adult follow up of those patients who received GH as children.73
Conclusions
From careful studies over the past 25 years GH appears to be an effective treatment for GHD, with adult heights of treated patients now in the normal range. The earlier treatment is started the more effective is the height gain. Compliance is a problem with GH treatment. This problem may be alleviated to some extent by us of long-acting GH preparations, now in development. Treatment during puberty may be improved by using higher doses of GH or delaying epiphyseal fusion with the use of aromatase inhibitors. GH is a relatively safe hormone, at least during the time that it is being administered. There are few data relating to long-term follow up, and this is a challenge for the future.