The pituitary gland, or hypophysis, is the ‘master gland’ that secretes multiple hormones which regulate the functioning of other endocrine organs, such as the thyroid, adrenal cortex and gonads. Though none of the major pituitary hormones directly control the endocrine glandular components of the pancreas, there are multiple indirect interactions that alter glucose homeostasis. Bromocriptine quick-release formulation, an agent used for treatment of hyperprolactinemia, is also approved for treatment of type 2 diabetes mellitus (T2DM). Though not a very popular therapeutic choice for diabetes mellitus (DM) management, the drug, however, opens the window of possibility that hypophyseal hormones and other neurotransmitters can turn out be novel therapeutic strategies to treat DM. DM, on the other hand, has been recognised as a common cause behind development of hypophyseal disorders like hypogonadotropic hypogonadism (HH).1,2 Thus, pituitary disease can cause hypoglycaemia or hyperglycaemia, and conversely dysglycaemia can also alter pituitary function. The pituitary gland shares a multifaceted relationship with glucose metabolism. The focus of this review is to help the clinician understand and recognise these various interactions. The concept of hypophyseo-vigilance aims to encourage the clinical suspicion of pituitary disease in dysglycaemia and help recognise pituitary dysfunction in setting of altered glucose metabolism in a timely manner. It is hoped that this will help to improve medical care and optimise clinical outcome.
Glucose metabolism in anterior pituitary disease
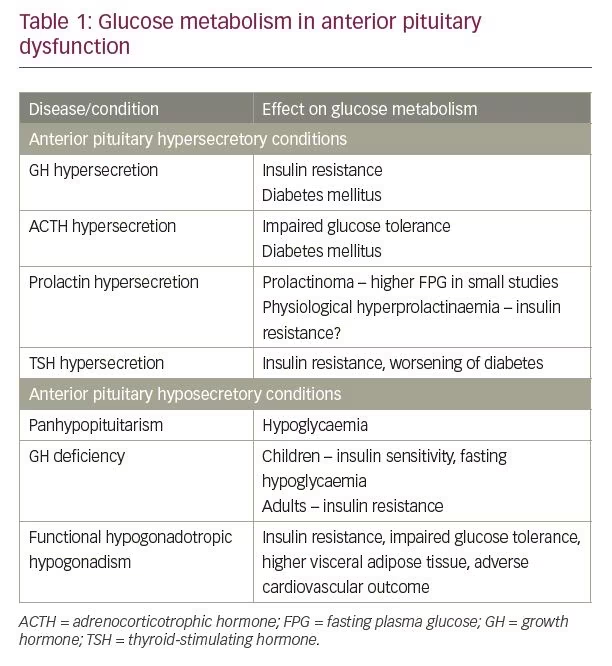
Anterior pituitary dysfunction can be either due to hypersecretion or hyposecretion of pituitary hormones. The impact of anterior pituitary dysfunction on glucose metabolism has been described in Table 1.
Hyperfunction
This encompasses four recognised conditions of hormone hypersecretion: pituitary adenoma secreting growth hormone (GH), or acromegaly; pituitary adenoma secreting adrenocorticotrophic hormone (ACTH), or Cushing’s disease; prolactin secreting pituitary adenoma; and very rarely thyroid-stimulating hormone (TSH) secreting pituitary adenoma. Gonadotrophin-secreting adenomas are usually clinically non-functioning but secretion of intact follicle-stimulating hormone (FSH) in women of reproductive age group has been associated with ovarian hyperstimulation.3
Growth hormone hypersecretion
In a database of 3,173 patients recently diagnosed with acromegaly, prevalence of DM was 27%. In the same study in non-diabetic subjects the glucose levels correlated with insulin-like growth factor-1 (IGF-1) levels, but not with GH levels.4 GH enhances hepatic glucose production by opposing the action of insulin on the liver and stimulates gluconeogenesis. GH also increases hepatic and peripheral insulin resistance. One of the mechanisms behind peripheral insulin resistance is substrate competition with glucose due to higher free fatty acid availability from GH induced lipolysis. On the other hand, IGF-1 improves peripheral insulin sensitivity but is not enough to override the diabetogenic action of chronic GH excess.5 Studies have demonstrated the association of IGF-1 levels and DM as predictors of mortality in acromegaly.6,7
Adrenocorticotrophic hormone hypersecretion
Cushing’s disease, or ACTH secreting pituitary adenoma, is another recognised cause of DM.8 The prevalence of DM in Cushing’s disease is 40–45% and an additional 10–30% have impaired glucose tolerance.9,10 A state of chronic cortisol excess stimulates hepatic gluconeogenesis partly resulting from increased mobilisation of gluconeogenic substrate from peripheral tissues. Cortisol also has a permissive effect on glucagon and epinephrine in inducing gluconeogenesis and glycogenolysis. Insulin secretory defects coupled with increased peripheral insulin resistance have additional role in development of hyperglycaemia.9
Prolactin hypersecretion
Unlike acromegaly and Cushing’s disease, hyperprolactinemia is not a recognised cause of DM.11 Small studies have demonstrated that prolactinoma is associated with higher fasting plasma glucose (FPG) that can be corrected with treatment with cabergoline.12,13 But the relationship between DM and prolactin is complex. In a population-based study of 2,377 Chinese men and postmenopausal women, elevated serum prolactin levels were associated with lower prevalence of DM.14 In another population-based study of 370 Japanese non-diabetic men, elevated prolactin in physiological range showed an association with insulin resistance.15 Experimental studies also show conflicting results.16–19 On one hand, prolactin has been shown to induce beta-cell growth in pancreas,16,17 while on the other hand prolactin is also hypothesised to increase insulin resistance by blocking adiponectin and interleukin (IL)-6 production in fat cells.18,19
Others
Thyrotoxicosis can cause insulin resistance and predispose to diabetes.20 TSH secreting pituitary adenoma by producing thyrotoxicosis can worsen glucose intolerance like in any other thyrotoxic state, but the condition is very rare and as per our knowledge, diabetes arising from this condition has not been reported.21 Gonadotropin-secreting tumours are non-functional and in exceptional circumstances can produce manifestations related to gonadotropin excess and not usually linked to glucose homeostasis.
Hypofunction
Pituitary hypofunction or hypopituitarism implies the deficiency of one or more anterior and posterior pituitary hormones. The common causes of hypopituitarism are pituitary adenoma or other sellar and suprasellar lesions, surgery or radiation to the pituitary gland, traumatic brain injury, and Sheehan’s syndrome. In most of these situations, multiple hormones are deficient; however, in lymphocytic hypophysitis, isolated deficiency of a single hormone can occur. Hypoglycaemia can be a presenting feature of hypopituitarism.22 It results primarily from hypocortisolism, secondary to defect in ACTH secretion coupled with deficiency of GH.
GH deficiency has a different impact on glucose homeostasis at various stages of life. Children with GH deficiency have a tendency to be insulin sensitive and can develop spontaneous fasting hypoglycaemia due to reduced hepatic glucose production.23 GH deficient adults who are not receiving GH substitution, however, are insulin resistant.24
Besides overt hormonal deficiency, functional hypogonadism presumed to be due to a non-organic defect in hypothalamic-pituitary-testicular axis, is a common cause of low plasma testosterone in adult males with comorbidities like diabetes and obesity.25 Low plasma total testosterone levels have been linked with insulin resistance, impaired glucose tolerance, higher visceral adipose tissue and adverse cardiovascular (CV) outcomes.26,27 Whether metabolic syndrome is the cause behind hypogonadism or its effect remains an area of controversy.28
Glucose metabolism in posterior pituitary disease
The posterior pituitary secretes two major hormones oxytocin and vasopressin.
Oxytocin
An imbalance in oxytocin secretion related to a disease process is not known to cause any specific defect in glucose metabolism; however, over last few years the physiological role of oxytocin in glucose and lipid metabolism has been recognised. Peripherally, oxytocin maintains electrolyte balance, glucose metabolism and adipogenesis, and centrally it is involved in food selection, reward and satiety.29 Oxytocin preferentially suppresses intake of sweet-tasting carbohydrates. Animal studies indicate that defects in oxytocin signalling and receptor expression can lead to weight gain despite of normal food intake.30 Oxytocin levels were found to be lower in newly diagnosed T2DM and obese individuals.31 A review analysing the weight-lowering and glycaemic benefits of intranasal oxytocin in a few small studies, showed positive trend.32
Vasopressin
Diabetes insipidus, a condition caused by vasopressin or antidiuretic hormone (ADH) deficiency and syndrome of inappropriate ADH (SIADH), whereby ADH is secreted in excess, both do not have any direct effect on glucose metabolism. However other than its role in fluid balance, there is emerging evidence that vasopressin influences glucose metabolism. Murine and in vitro studies have demonstrated that the vasopressin receptor V1a is present on the liver where its activation causes glycogenolysis and gluconeogenesis.33,34 The pancreas also expresses V1b receptors on both alpha and beta cells. At higher glucose concentration, vasopressin predominantly causes insulin secretion from beta cells whereas at lower glucose concentration it primarily releases glucagon from alpha cells.35
Murine studies demonstrate that vasopressin has a predominant role in inducing insulin resistance and causing hyperglycaemia through activation of V1a receptor.36,37 These animal data have been corroborated through clinical studies that indicate that high vasopressin levels can be a risk factor for DM.38,39 Copeptin, the C‐terminal of the pre/pro‐vasopressin peptide, has been used as a surrogate indicator of vasopressin in clinical studies because it is stable and can be assayed easily.40 A recent review summarises the experimental evidence as well as the human studies, mostly epidemiological, that have demonstrated that high copeptin levels are associated with DM, insulin resistance, obesity, hypertension and metabolic syndrome.41 An interesting study which further corroborates the relationship of vasopressin with hyperglycaemia, showed that higher water intake which in turn will lower vasopressin level, was associated with lower chance of new‐onset hyperglycaemia.42
Alteration in glucose metabolism with drugs used for treatment of pituitary dysfunction
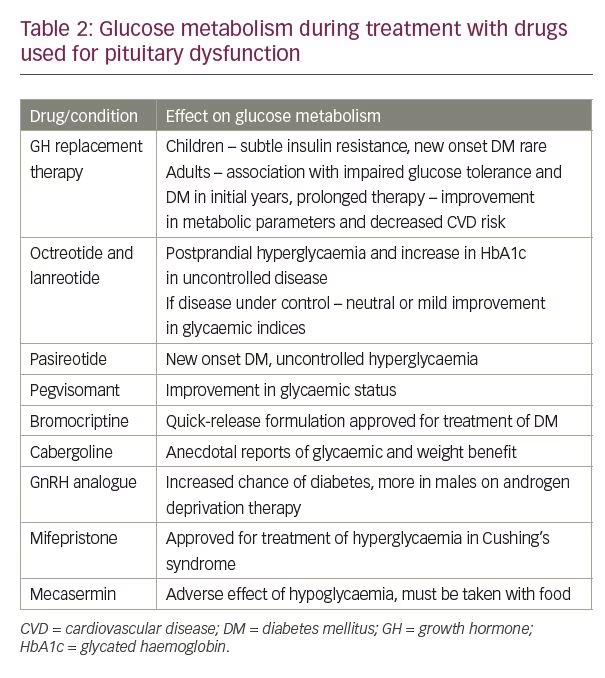
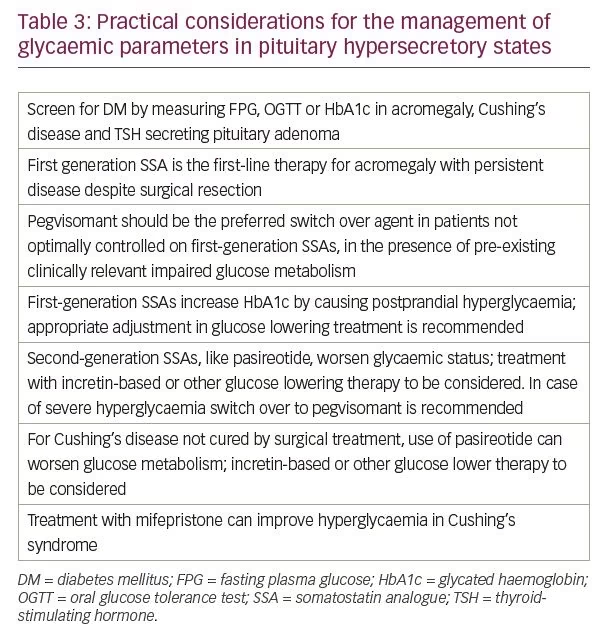
Many of the drugs that are used for treatment of pituitary dysfunction have an impact on glucose metabolism (Table 2). The clinical recommendations for screening and treatment of dysglycaemia during management of pituitary hyperfunction and hypofunction with these medications are summarised in Tables 3 and 4, respectively.

Growth hormone replacement therapy
GH replacement alters insulin sensitivity in a complex fashion. In a recently published review, the authors found that in majority of studies, GH treatment in children resulted in a subtle form of insulin resistance, though FPG and glycated haemoglobin (HbA1c) mostly remained in the normal range.43 New onset T2DM rarely occurs during GH treatment in children with GH deficiency or even with idiopathic short stature.44 Turner syndrome and children who were small for gestational age, have a higher lifetime chance of developing T2DM compared to background population, but GH therapy in such children does not alter the incidence of T2DM in short term.45
In adults with a higher BMI and/or family history of T2DM, GH therapy can be associated with the development of glucose intolerance or T2DM in the initial years of treatment, though the incidence decreases with prolonged treatment.46,47 Close monitoring of glycaemic status is advisable for those on GH replacement in adults with risk factors for T2DM.45 Treatment of GH deficiency in adults decreases fat mass and improves lean body mass, lipid profile, carotid intima medial thickness, and CV inflammatory markers, and more than offsets for mild derangement in glycaemic parameters.48–50
First-generation somatostatin analogues
Octreotide and lanreotide are considered as first-generation somatostatin analogues (SSAs) having a strong affinity to somatostatin receptor (SSR)2 and SSR5, and a weaker affinity to SSR3. They are the first-line medical therapy for acromegaly. First generation SSAs exert a glucose lowering effect by decreasing GH secretion from the tumour which is counterbalanced by its action on the pancreas where somatostatin and its analogues inhibit insulin secretion.51 The net effect on glucose metabolism in acromegaly is variable. In a recently published meta-analysis, the authors showed that a mild but significant increase in HbA1c occurred with first generations SSAs, resulting from increase in post prandial glucose levels without any alteration in FPG. It was also demonstrated that first-generation SSAs significantly reduced insulin secretion which was not completely counterbalanced by the reduction in GH and IGF-I levels. The authors concluded that the overall effect of first-generation SSAs on glucose metabolism depends on the net balance between these two opposing factors and on the patient’s predisposition.52 Different trends were observed in a retrospective 1-year study comparing SSAs to surgery. SSAs caused improvement in all metabolic syndrome parameters and significantly lowered FPG and HbA1c when the disease activity was controlled, the main determinant of glycaemic improvement.53 Higher dose and higher frequency of SSAs do not have an unfavourable glycaemic impact.54,55 To summarise, octreotide and lanreotide might have a mild detrimental effect on post prandial hyperglycaemia and HbA1c, especially in the setting of uncontrolled disease activity. If the disease is under control, the first generation SSAs have either a neutral or even a slightly beneficial effect on glycaemic indices.
Pasireotide
This novel SSA has strong affinity for SSR5 and SSR1, moderate affinity for SSR3 and lower affinity for SSR2 in comparison to octreotide.56 In addition to better efficacy for treatment of acromegaly, as compared to first-generation SSAs, pasireotide, by virtue of its affinity for both SSR2 and SSR5, suppress ACTH hypersecretion in patients with corticotroph tumors.57,58 Its predominant adverse effect is hyperglycaemia in both acromegaly (57.3–67.0%) and Cushing’s disease (68.4–73.0%), and uncontrolled hyperglycaemia is a common cause of treatment discontinuation.59 Insulin release from beta cells of islets of Langerhans is mediated by SSR5 and SSR2 and glucagon release from alpha cells depend on SSR2.60 Pasireotide has a prominent inhibitory effect on insulin release as a result of its higher affinity for SSR5, whereas the inhibitory effect on glucagon secretion due to SSR2 affinity is only modest. First-generation SSAs are more potent suppressors of glucagon secretion because of stronger binding to SSR2.61 Apart from this imbalance in insulin and glucagon secretion, pasireotide also reduces incretin response and incretin-based therapy might be more effective than other forms of therapy for management of hyperglycaemia in this condition.62
Pegvisomant
Pegvisomant is a genetically engineered potent GH receptor antagonist that acts at the receptor level to decrease IGF-1 levels.63 Unlike SSAs, it doesn’t have any inhibitory action on insulin secretion on the pancreas and has a favourable effect on glycaemic status.64 Pegvisomant improves glucose metabolism by improving insulin resistance in acromegaly through blocking of excess GH action.65,66 It has a positive impact on glycaemic control in patients with acromegaly and uncontrolled DM.67 Patients with acromegaly and DM have a higher requirement for pegvisomant to control the disease and have a lower rate of IGF-1 normalisation compared to those without DM.68 A possible hypothesis is that hepatic GH receptor expression may be increased due to hyperinsulinism and higher dose of pegvisomant may be needed to saturate it.69 The recently published consensus statement by Melmed et al. recommends shifting from first generation SSAs to pegvisomant in case there is pre-existing clinically relevant impaired glucose metabolism.70
Bromocriptine
Dopamine and dopaminergic signals in the central nervous system (CNS) decrease liver gluconeogenesis via the sympathetic nervous system. In obese individuals with DM, decreased dopaminergic activity alters signalling from the CNS resulting in increased hepatic gluconeogenesis and insulin resistance.71 Antipsychotic medicines that block dopaminergic activity cause weight gain, dyslipidaemia and insulin resistance.72 Bromocriptine, a D2-dopamine agonist with sympatholytic activity, is primarily indicated for the treatment of prolactinoma. Bromocriptine quick-release tablet ingestion within 2 hours of waking is assumed to enhance low hypothalamic dopamine levels and inhibit excessive sympathetic tone within the CNS.73 This is the only approved glucose lowering medication that predominantly acts on the CNS and causes reduction of FPG, HbA1c, triglyceride and free fatty acid levels.74 In a 1-year prospective study, HbA1c was decreased by 0.6% and composite CV outcome was reduced by 40%.75 However, the mechanism behind reduction in CV events is not defined. The additional advantages of bromocriptine include absence of hypoglycaemia, no change in weight, and lack of major side effects.
Cabergoline
Cabergoline, a long-acting D2-dopamine agonist, is better tolerated than bromocriptine and considered as the first-line treatment of hyperprolactinemia. It has also demonstrated benefit in DM, obesity and insulin resistance in small studies.76–78 In a recently published trial, cabergoline did not exhibit any difference with placebo in maintaining weight loss in obese subjects who had lost at least 5% weight with an 800 kcal/day commercial meal replacement programme.79 Cabergoline, despite of being a stronger D2 receptor agonist, has minimal glycaemic benefit as compared to bromocriptine quick-release formulation. Unravelling of intricacies of dopaminergic and other CNS pathways that control metabolism, might resolve these discrepancies in future.
Gonadotropin releasing hormone analogue
Gonadotropin releasing hormone (GnRH) analogues, such as leuprolide, triptorelin, buserelin, goserelin and histrelin are used in adult females for the treatment of endometriosis, fibroids, premenopausal ovarian suppression, and for the treatment of metastatic prostate cancer in males. The metabolic changes that occur due to androgen deprivation therapy with GnRH analogues in males include increase in adipose tissue, increased low-density lipoprotein cholesterol, and insulin resistance and DM. In an observational study of a population-based cohort of 73,196 people, the unadjusted rates per 1,000 person-years for developing incident diabetes was 29 for those receiving GnRH analogue therapy versus 20.9 for those not on treatment, with an adjusted hazard ratio of incident DM of 1.44 with GnRH analogues.80 GnRH analogue-induced hyperglycaemia has been reported to respond well to pioglitazone.81 Females also may develop worsening of glycaemic control when treated with these agents.82
Mifepristone
Mifepristone is a glucocorticoid receptor blocker acting on glucocorticoid receptor-II and the progesterone receptor. It is approved for treatment of hyperglycaemia in the setting of Cushing’s syndrome. In a study of 50 patients with endogenous Cushing’s syndrome (including pituitary, adrenal and ectopic causes of hypercortisolism), 60% of patients with glucose intolerance or DM showed improvement. In patients using insulin, their dose was reduced by half and mean HbA1c came down from 7.43% at baseline to 6.29% at study conclusion.83
Mecasermin
Mecasermin or recombinant human IGF-1 is used for treatment of growth failure resulting from severe primary IGF-1 deficiency, or in cases of GH gene deletion with neutralising antibodies to GH that make GH ineffective.84 Hypoglycaemia is a recognised side effect of mecasermin, especially in small children due to inconsistent oral intake. Individuals should not undertake high-risk activities (e.g. swimming) within 2–3 hours after dosing, particularly at initiation of therapy until the dose is tolerated. It is injected subcutaneously twice daily and should be administered within 20 minutes of a meal or a snack.
Effect of diabetes on hypophyseal function
Pituitary hormones may be altered in both T1DM and T2DM.
Diabetes and growth hormone – IGF-1 axis
Type 1 diabetes mellitus
GH and insulin appear to be metabolically antagonistic to each other, but in reality, insulin plays a permissive role in GH action. Portal insulin up-regulates hepatic GH-receptor expression, and that in turn causes downstream production of IGF-1 and IGF-binding protein-3 (IGFBP3).85 Low portal insulin levels, decreased IGF-1 levels, and elevated proinflammatory cytokines in T1DM lead to IGF-1 resistance. Low IGF-1 in T1DM has multiple deleterious effects like decreased growth, insulin resistance and poor CV outcomes.86 The imbalance in the GH/IGF-1/IGFBP3 axis, however, does not get corrected with continuous subcutaneous insulin infusion despite optimal glycaemic control, but does improve with continuous portal insulin infusion.87,88 The implication of the finding, that even optimal glycaemic control might not restore the GH/IGF-1 axis, is not clear. Whether deficient portal insulin in T1DM adversely affects the final height outcome is yet to be proven and the adult height of individuals with T1DM remains highly variable.86
Type 2 diabetes mellitus
The GH/IGF-1 axis plays a possible role in glucose homeostasis and the etiopathogenesis of T2DM and insulin resistance. In T2DM, GH levels are low and free IGF-1 levels are elevated; however, total IGF-1 levels are normal. In addition, IGFBP1 and IGFBP2 levels are low while IGFBP3 is elevated in T2DM.89 Epidemiological studies on the connection between IGF proteins and the incidence of T2DM report conflicting results and most fail to show any association.90,91 Low levels of IGFBP1, however, have been associated with future risk of T2DM development.92 Glucose metabolism and GH, IGF-1 and IGFBPs are interlinked in a complicated manner and their relationship is currently a subject of ongoing research.
Diabetes and gonadal axis
It is well established that males with T2DM have a high prevalence of decreased serum testosterone along with inappropriately low luteinizing hormone and FSH levels, a condition that has been labelled as functional HH because it is not associated with any structural disease.25 HH has a close link to insulin resistance and can be found in individuals with T2DM, obesity and metabolic syndrome.1,2 C-reactive protein levels are higher in individuals with HH and have an inverse association to testosterone levels. Inflammation secondary to insulin resistance may contribute to its pathogenesis.93 Testosterone replacement is recommended in symptomatic men with HH but there is controversy regarding the CV risk associated with testosterone therapy.94,95 An interesting observation is that the hypogonadism in these conditions respond well to clomiphene.96 Prevalence of HH in men with T1DM does not appear to be increased and risk factors in T1DM for low testosterone levels are similar to that of the general population.97
Diabetes and prolactin secretion
An early study reported that prolactin levels are elevated in T2DM.98 Subsequent findings, however, suggest that high prolactin levels reduce the prevalence of DM and insulin resistance.13,14 Additionally, a study reported that those with diabetic retinopathy have lower levels of prolactin.99 A possible hypothesis behind this is prolactin is broken down to vasoinhibins, with antiangiogenic, vasoconstrictive and anti-permeability effects that may exert protective action in diabetic retinopathy.100 However, a few other clinical studies have not confirmed the benefits of elevated levels of prolactin on retinopathy.101,102
Diabetes and cortisol axis
Animal studies suggest that hypothalamus–pituitary–adrenal (HPA) axis may be hyperfunctioning in T2DM and these changes may be mediated through the endogenous cannabinoid receptor system.103–106 The clinical implication of this finding is not known.
Diabetes and hypophysis with syndromic connection
Diabetes is a defining feature of numerous syndromes and many of them have associated hypophyseal affection (Table 5).
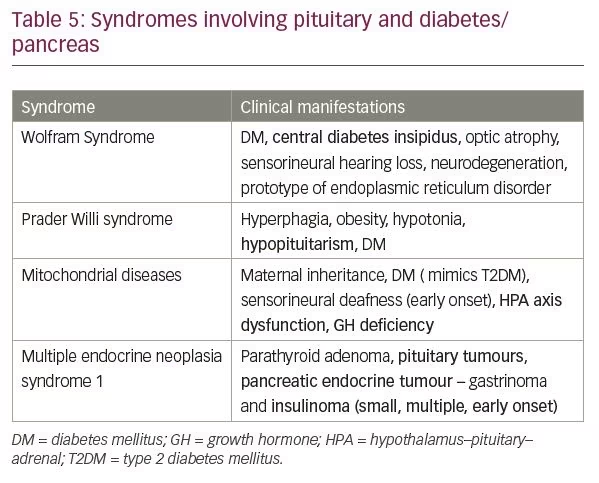
Wolfram syndrome
Wolfram syndrome is a rare genetic disease which manifests as childhood onset DM, central diabetes insipidus, optic nerve atrophy, sensorineural hearing loss, and neurodegeneration. It is a prototype of endoplasmic reticulum disorder occurring due to mutation in WFS1 gene and pancreatic beta cells, and neurons are particularly sensitive to it, most probably due to high rates of protein synthesis.107
Prader Willi syndrome
Prader Willi syndrome is one of the most common varieties of obesity syndrome. It results from the absence of expression of the paternally active genes on the long arm of chromosome 15. Excessive appetite, obesity and hypotonia are hallmark of this syndrome. Hypopituitarism is often an accompanying feature with sequential loss of GH, luteinizing hormone, FSH, TSH and ACTH secretion.108
Mitochondrial disease
Another situation where diabetes and pituitary dysfunction coexist are mitochondrial disorders. The common variants of mitochondrial diseases include mitochondrial encephalomyopathy, lactic acidosis and stroke-like episodes (MELAS); maternally-inherited diabetes and deafness; and Kearns Sayre Syndrome (KSS). Mitochondrial diabetes presents much like T2DM and should be suspected in the presence of early onset sensorineural deafness along with maternal inheritance of either condition. Other features which suggest the possibility of mitochondrial diabetes are cardiomyopathy, seizure disorder, ptosis or unusual strokes. Impairment of the HPA axis occurs in severe mitochondrial phenotypes presenting in childhood and more commonly in MELAS and KSS. GH deficiency has been described in KSS.109
Multiple endocrine neoplasia 1 syndrome
Multiple endocrine neoplasia 1 (MEN1) syndrome is an autosomal dominant condition that is characterised by predisposition to tumour formation of the parathyroid glands, anterior pituitary and islet cells of pancreas. Menin, a tumour suppressor gene located in long arm of chromosome 11 (11q13), is inactivated in majority of affected members.110 In a recently published series of patients with MEN1 syndrome, the prevalence of pituitary tumours was 42%. Prolactinoma was the most common variety of pituitary tumour, followed by GH secreting tumour, Cushing’s disease, gonadotroph adenomas and clinically non-functioning tumours.111 The other manifestation of MEN1 syndrome is tumour of pancreas and gastro-intestinal neuroendocrine glands. Zollinger Ellison syndrome, or gastrinoma, is the most common presentation among that group followed by insulinoma. Insulinomas in MEN1 syndrome are usually small and multiple and present early in second to fourth decade.112
Other syndromic associations
There are multiple other syndromes, such as Bardet-Biedl syndrome, Friedreich ataxia, Alström syndrome and Berardinelli-Seip syndrome where diabetes and hypophyseal dysfunction coexist.
Conclusion
Pituitary hormones play an integral role in controlling glucose metabolism. There are diseases like acromegaly and Cushing’s disease which are overtly associated with diabetes. Emerging evidence suggests that prolactin, vasopressin and oxytocin also exert a subtle physiological role in glucose homeostasis.15,29,41 The choice of medication in pituitary diseases such as acromegaly can be governed by the glycaemic impact of these drugs; pegvisomant being preferred in patients with uncontrolled hyperglycaemia while pasireotide is often administered in those where glycaemic control is optimal. Bromocriptine quick-release formulation, a drug principally used for hyperprolactinemia, has been used as an antihyperglycaemic agent and might open the door for future medications which target the CNS as a modality to treat DM. Diabetes is a recognised cause of HH in males and inflammation associated with T2DM has a negative impact on functioning of gonadotrophs.1,2 Whether replacement of testosterone in these individuals is beneficial is again a subject of controversy. Finally, there are syndromes which link the pituitary and the pancreas, and throw important light on mechanism of secretory function of these two major endocrine glands.